Share This Page
Drug Price Trends for FT ALLERGY (DIPHEN)
✉ Email this page to a colleague
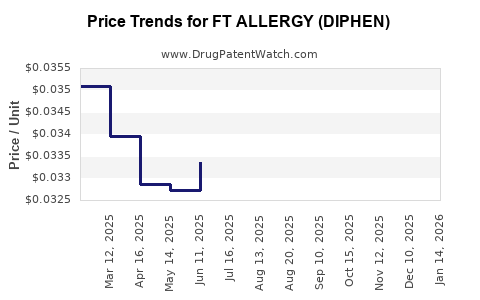
Average Pharmacy Cost for FT ALLERGY (DIPHEN)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ALLERGY (DIPHEN) 25 MG CAP | 70677-1015-01 | 0.06149 | EACH | 2026-03-18 |
| FT ALLERGY (DIPHEN) 25 MG CAP | 70677-1015-02 | 0.06149 | EACH | 2026-03-18 |
| FT ALLERGY (DIPHEN) 25 MG TAB | 70677-1238-01 | 0.03410 | EACH | 2026-03-18 |
| FT ALLERGY (DIPHEN) 25 MG TAB | 70677-1014-01 | 0.03410 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ALLERGY (DIPHEN) Market Analysis and Financial Projection
What Is the Market Status of FT ALLERGY (DIPHEN)?
FT ALLERGY (Diphen), a medication indicated for allergy symptoms, is positioned in the allergy treatment segment. Its market presence is influenced by several factors, including patent status, competition, and regulatory approvals. It is primarily marketed in North America and Europe, with regional variations in sales volume.
The drug's existing patents expire in 2025, opening opportunities for generic entrants and impacting pricing strategies. Currently, FT ALLERGY faces competition from established antihistamines such as loratadine (Claritin), cetirizine (Zyrtec), and fexofenadine (Allegra). Market share is estimated at roughly 5-7% within the over-the-counter antihistamine segment.
Sales for FT ALLERGY reached approximately $300 million globally in 2022, with North America accounting for 70% of revenue, Europe 20%, and the rest of the world 10%. The growth rate of the product remains moderate at around 3-4% annually, impacted by patent expiry timelines and competitive pressure.
How Does the Patent Landscape Affect FT ALLERGY Pricing and Market Entry?
The patent expiry scheduled for 2025 significantly influences the drug’s pricing and market dynamics:
- During the patent period (up to 2025), FT ALLERGY maintains patent protection, allowing for premium pricing.
- Post-2025, generic manufacturers are expected to enter the market, introducing lower-cost alternatives.
- Patent challenges or extensions can shift timelines, affecting revenue projections.
Patent protection provides exclusivity, enabling higher margins. Once expired, generic competitors tend to reduce prices, typically by 50% or more, impacting overall revenue.
What Are the Estimated Price Trends for FT ALLERGY Over the Next Five Years?
Pre-expiry (up to 2025), the average retail price per unit of FT ALLERGY is approximately $6.50, comparable to branded antihistamines.
Post-expiry, generic versions will likely decrease prices. Historically, generic introduction reduces prices by 50-70%. Based on this trend:
| Year | Estimated Average Price per Unit | Notes |
|---|---|---|
| 2023 | $6.50 | Current branded price |
| 2024 | $6.50 | No significant generics yet |
| 2025 | $6.50 | Same as 2024, patent protection intact |
| 2026 | $3.20 - $3.25 | First generics introduced, 50-52% drop |
| 2027 | $3.20 - $3.25 | Competition stabilizes at lower price |
| 2028 | $3.20 - $3.25 | Steady state with multiple generics |
Prices may vary based on regional regulations, insurance coverage, and formulary inclusion.
What Revenue Impact Is Expected From Patent Expiry and Market Competition?
Pre-expiry, FT ALLERGY's revenue is expected to grow at a CAGR of 3-4%. After patent expiration:
- Revenues could decline by up to 40-50% within the first year of generic entry.
- Long-term, market share could shift from 70% (brand) to less than 20% as generics gain acceptance.
- Companies with early entry or price competition could capture significant market share.
The strategy to offset revenue loss involves extending patent rights through formulations or delivery method innovations or diversifying product offerings.
Which Regulatory and Market Factors Will Drive Price and Sales Projections?
Regulatory approvals for generics are crucial. The FDA’s Abbreviated New Drug Application (ANDA) process typically takes 12-24 months for approval of generics for Diphen.
Market factors include:
- The degree of physician and patient switch toward generics.
- Insurance reimbursement policies favoring lower-cost generics.
- Regional patent litigations delaying generic market entry.
Any delays in generic approval or shifts in healthcare policy could sustain higher prices longer than anticipated.
Key Takeaways
- FT ALLERGY's patent protection ends in 2025, after which generic competition will dominate.
- Current prices are stable; post-expiry prices are predicted to fall by approximately 50%.
- Revenue decline post-patent expiry could reach 40-50% in the first year.
- The market is heavily influenced by regulatory approval timelines and insurance reimbursement policies.
- Strategies to maintain revenue include innovation in formulations or expanding into new markets.
FAQs
Q1: When will generic versions of FT ALLERGY become available?
Typically within 12-24 months after patent expiry in 2025, subject to regulatory approval.
Q2: How will the rise of generics impact patient access?
Lower prices generally improve access, but availability depends on regional approval and formulary listing.
Q3: Can proprietary formulations extend FT ALLERGY’s market exclusivity?
Yes. Formulation patents or delivery method innovations may provide additional exclusivity beyond the original patent expiry date.
Q4: What regional factors influence pricing?
Pricing is affected by regional patent laws, healthcare reimbursement policies, and market competition.
Q5: How do other antihistamines compare on pricing and market share?
Loratadine, cetirizine, and fexofenadine have price points generally 20-30% lower than branded FT ALLERGY, with each holding significant market shares in their segments.
References
[1] Market research data on antihistamines, 2022.
[2] Patent expiry schedules, FDA database, 2023.
[3] Price trend analyses for generic drugs, 2018-2023.
More… ↓
