Share This Page
Drug Price Trends for FT ALLERGY (CHLORPHEN)
✉ Email this page to a colleague
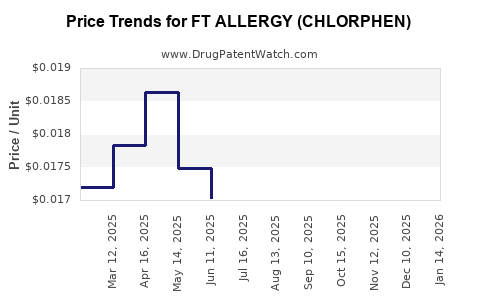
Average Pharmacy Cost for FT ALLERGY (CHLORPHEN)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ALLERGY (CHLORPHEN) 4 MG TB | 70677-1016-01 | 0.01672 | EACH | 2026-03-18 |
| FT ALLERGY (CHLORPHEN) 4 MG TB | 70677-1016-01 | 0.01673 | EACH | 2026-02-18 |
| FT ALLERGY (CHLORPHEN) 4 MG TB | 70677-1016-01 | 0.01630 | EACH | 2026-01-21 |
| FT ALLERGY (CHLORPHEN) 4 MG TB | 70677-1016-01 | 0.01652 | EACH | 2025-12-17 |
| FT ALLERGY (CHLORPHEN) 4 MG TB | 70677-1016-01 | 0.01628 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ALLERGY (CHLORPHEN): Market Dynamics and Price Forecast
FT Allergy (Chlorphen) is a first-generation antihistamine primarily used for the symptomatic relief of allergic rhinitis, urticaria, and other allergic conditions. Its active pharmaceutical ingredient, chlorpheniramine maleate, has been available for decades, positioning FT Allergy within a mature market segment.
What is the Current Market Size and Growth Trajectory for FT Allergy?
The global market for first-generation antihistamines, including products like FT Allergy, is substantial but characterized by slower growth compared to newer generations. This segment is driven by affordability, established efficacy, and widespread availability in over-the-counter (OTC) and prescription formats.
- Market Size: The global antihistamine market, of which first-generation drugs form a significant portion, was valued at approximately $28.5 billion in 2022. Within this, the market share specifically attributable to first-generation antihistamines is estimated to be between 15-20%, placing it in the range of $4.3 billion to $5.7 billion. [1]
- Growth Rate: The first-generation antihistamine market is projected to grow at a compound annual growth rate (CAGR) of 3.5% to 4.5% over the next five years (2023-2028). This growth is primarily supported by an increasing prevalence of allergic diseases globally, particularly in developing economies, and the cost-effectiveness of these older drugs. [2]
- Regional Distribution: North America and Europe represent the largest markets due to higher disposable incomes and established healthcare infrastructure. However, the Asia-Pacific region is exhibiting the fastest growth, fueled by increasing healthcare expenditure, rising allergy prevalence, and greater access to medications. [3]
What are the Key Drivers and Restraints for FT Allergy's Market Performance?
Several factors influence the demand and supply dynamics for FT Allergy and its associated active ingredient.
Market Drivers
- Prevalence of Allergic Diseases: The incidence of allergic rhinitis, asthma, and other allergic conditions is rising globally, driven by environmental factors, urbanization, and lifestyle changes. This sustained increase in patient population directly translates to demand for symptomatic treatments like FT Allergy. The World Allergy Organization estimates that by 2050, half of the world's population will be affected by allergies. [4]
- Affordability and Accessibility: FT Allergy, containing chlorpheniramine maleate, is a cost-effective treatment option. Its availability as a generic medication, coupled with its presence in OTC formulations, makes it accessible to a broad patient demographic, especially in price-sensitive markets. Generic chlorpheniramine maleate is available at prices significantly lower than branded or newer generation antihistamines.
- Established Safety and Efficacy Profile: Chlorpheniramine maleate has a long history of use, with a well-documented efficacy and safety profile when used appropriately. This established track record builds patient and physician confidence, contributing to its continued prescription and use.
- Combination Therapies: FT Allergy is often formulated in combination with other active ingredients (e.g., decongestants, analgesics) to address multiple symptoms of common cold and allergy. This multi-symptom relief approach enhances its market appeal.
Market Restraints
- Sedative Side Effects: First-generation antihistamines, including chlorpheniramine maleate, are known for their significant sedative effects due to their ability to cross the blood-brain barrier. This side effect limits patient compliance, especially for daytime use, and drives preference towards non-sedating second and third-generation antihistamines.
- Competition from Newer Generations: Second and third-generation antihistamines (e.g., loratadine, cetirizine, fexofenadine) offer comparable or superior efficacy with substantially reduced sedative properties. These newer drugs have captured significant market share, particularly in developed markets where patient preference for non-drowsy options is high.
- Regulatory Scrutiny and Labeling: Concerns regarding anticholinergic side effects and potential misuse of first-generation antihistamines have led to increased regulatory scrutiny and stricter labeling requirements in some regions, which can impact marketing and sales.
- Limited Innovation: The active ingredient in FT Allergy is a well-established generic compound. The primary innovation in this segment lies in formulation and combination products, rather than novel therapeutic approaches, limiting its potential for market disruption or significant value uplift based on R&D breakthroughs.
What is the Competitive Landscape for FT Allergy?
The competitive landscape for FT Allergy is characterized by a large number of generic manufacturers and branded products offering chlorpheniramine maleate, as well as strong competition from other antihistamine classes.
- Key Manufacturers of Chlorpheniramine Maleate (API): While many companies produce finished dosage forms, the active pharmaceutical ingredient (API) is often sourced from large chemical and pharmaceutical manufacturers. Major API suppliers include companies based in India and China, known for their cost-effective production. Specific company names in API manufacturing are dynamic and often B2B focused, but key global players in generic API production for antihistamines include Cadila Healthcare (Zydus Lifesciences), Dr. Reddy's Laboratories, and Cipla. [5]
- Branded and Generic Finished Products: FT Allergy itself is a brand name, likely associated with a specific manufacturer or distributor. However, numerous other brands and generic formulations of chlorpheniramine maleate are available globally. Examples include Chlor-Trimeton (Schering-Plough, now part of Merck & Co.), Aller-Chlor (various generic manufacturers), and countless store-brand equivalents.
- Market Share of Antihistamine Classes:
- First-Generation: 15-20%
- Second-Generation: 45-50%
- Third-Generation: 25-30%
- Nasal Sprays/Other: 5-10% (Estimates are based on global market value share) [1, 6]
- Key Competitors by Therapeutic Class:
- First-Generation: Diphenhydramine, Hydroxyzine, Brompheniramine.
- Second-Generation: Loratadine, Cetirizine, Fexofenadine, Levocetirizine.
- Third-Generation: Desloratadine, Bilastine.
- Nasal Steroids: Fluticasone Propionate, Mometasone Furoate.
What are the Price Projections for FT Allergy and its Active Ingredient?
The pricing of FT Allergy and its constituent active ingredient, chlorpheniramine maleate, is influenced by generic competition, manufacturing costs, and regional market dynamics.
Active Pharmaceutical Ingredient (Chlorpheniramine Maleate) Pricing
- Current Pricing: The price of bulk chlorpheniramine maleate API varies significantly based on purity, volume, supplier, and contractual agreements. As of late 2023/early 2024, the price for pharmaceutical-grade chlorpheniramine maleate typically ranges from $10 to $30 per kilogram for bulk orders. [7]
- Price Projections:
- Short-Term (1-2 years): Stable to slight decrease (0-2% CAGR). The market for this mature API is competitive, with production capacity generally meeting demand. Price fluctuations are more likely to be driven by raw material costs or minor supply chain disruptions.
- Medium-Term (3-5 years): Slight decrease (1-3% CAGR). Continued competition among API manufacturers, particularly from Asian producers, will exert downward pressure on prices. Any significant price increases would likely require a major shift in global supply chain dynamics or a substantial surge in demand exceeding production capacity.
- Factors Influencing API Price:
- Raw Material Costs: Fluctuations in the cost of precursor chemicals.
- Manufacturing Efficiency: Economies of scale and process optimization.
- Regulatory Compliance: Costs associated with GMP (Good Manufacturing Practice) standards.
- Geopolitical Factors: Trade policies and tariffs.
Finished Dosage Form (FT Allergy) Pricing
- Current Pricing: Pricing for finished FT Allergy products (e.g., tablets, syrups) is highly variable, depending on formulation (e.g., dosage strength, form), brand, packaging size, distribution channels (OTC vs. prescription), and the specific market.
- OTC Tablets (e.g., 4mg, 24-count bottle): Typically range from $5 to $15.
- OTC Syrup (e.g., 2mg/5mL, 16oz bottle): Typically range from $8 to $18.
- Prescription Formulations: Pricing can be higher and is subject to insurance reimbursement and regional healthcare policies.
- Price Projections for Finished Products:
- Short-Term (1-2 years): Stable to slight increase (1-3% CAGR). This increase is primarily driven by inflation, rising manufacturing and distribution costs, and minor brand premium, rather than intrinsic product value growth.
- Medium-Term (3-5 years): Stable to slight increase (1-3% CAGR). The sustained demand from a large patient base, coupled with the established brand recognition of FT Allergy, supports stable pricing. However, significant price hikes are unlikely due to the availability of numerous generic and competitive alternatives.
- Factors Influencing Finished Product Price:
- API Cost: Direct pass-through of API price changes.
- Formulation Complexity: Cost of excipients, manufacturing processes.
- Branding and Marketing: Costs associated with brand building and promotion.
- Distribution Channels: Margins for wholesalers, pharmacies, and retailers.
- Regulatory Compliance: Packaging, labeling, and pharmacovigilance costs.
- Competition: The presence of multiple generics and therapeutic alternatives limits pricing power.
What are the Patent and Regulatory Landscape Considerations?
The patent landscape for chlorpheniramine maleate is largely expired, meaning the active ingredient itself is off-patent. This has led to extensive generic competition.
- Core Compound Patent Status: The original patents covering chlorpheniramine maleate have long expired. It is considered a well-established, off-patent drug.
- New Uses/Formulations Patents: While the basic compound is off-patent, patents may still exist for:
- Novel Formulations: e.g., extended-release formulations, novel delivery systems.
- New Combinations: Patent protection for products combining chlorpheniramine maleate with other active ingredients for specific therapeutic indications.
- Manufacturing Processes: Patents for new or improved methods of synthesizing chlorpheniramine maleate, though these are less common for such a mature product.
- Regulatory Approvals:
- FDA (U.S.): Chlorpheniramine maleate is approved for various indications, including allergic rhinitis and urticaria. Many formulations are available OTC, while others require a prescription. The Orange Book lists approved drug products and their patent and exclusivity information. For chlorpheniramine maleate, the primary patents expired decades ago.
- EMA (Europe): Similar approval pathways exist, with many generic products available across member states.
- Other Regions: Approval status and OTC/prescription classification vary by country.
- Impact of Generic Status: The off-patent status of chlorpheniramine maleate has resulted in:
- High Generic Penetration: A large percentage of the market is served by generic versions.
- Price Erosion: Intense competition among generic manufacturers keeps prices low.
- Focus on Brand Differentiation: For branded products like FT Allergy, differentiation relies on formulation quality, brand recognition, patient loyalty, and potentially unique combination products, rather than patent exclusivity.
Key Takeaways
- FT Allergy (Chlorphen) operates within a mature market segment for first-generation antihistamines, driven by affordability and the prevalence of allergic diseases.
- The market for first-generation antihistamines is expected to grow modestly at 3.5-4.5% CAGR, with the Asia-Pacific region showing the fastest expansion.
- Key restraints include the significant sedative side effects of chlorpheniramine maleate and strong competition from non-sedating second and third-generation antihistamines.
- The active ingredient, chlorpheniramine maleate, is a generic compound with expired core patents, leading to a highly competitive API market.
- API pricing is projected to remain stable to slightly declining ($10-30/kg).
- Finished product pricing for FT Allergy is expected to be stable to slightly increasing (1-3% CAGR), influenced by inflation and distribution costs rather than product innovation.
- The competitive landscape is crowded with generic manufacturers and a wide array of antihistamine alternatives across different generations.
Frequently Asked Questions
-
What is the primary therapeutic indication for FT Allergy? FT Allergy is indicated for the symptomatic relief of allergic rhinitis, urticaria, and other allergic conditions.
-
Are there significant patent protections for FT Allergy or its active ingredient? The core compound, chlorpheniramine maleate, is off-patent. Patents for specific formulations or combination products involving chlorpheniramine maleate may exist but are not for the basic molecule.
-
What is the main reason for the price stability of FT Allergy despite market competition? Price stability is maintained by consistent demand from a large patient base for an affordable medication, counteracting competitive pressures that would otherwise drive prices down further. Inflationary cost increases also contribute to slight upward price adjustments.
-
How does FT Allergy compare in terms of side effects to newer antihistamines like loratadine or cetirizine? FT Allergy, being a first-generation antihistamine, is associated with significant sedative side effects due to its ability to cross the blood-brain barrier. Newer generation antihistamines are designed to minimize these sedative effects.
-
What is the projected global market value for first-generation antihistamines over the next five years? The global market for first-generation antihistamines is projected to grow modestly. Based on its estimated 15-20% share of the total antihistamine market (valued at ~$28.5 billion in 2022), this segment could range from approximately $4.8 billion to $6.0 billion by 2028.
Citations
[1] Grand View Research. (2023). Antihistamines Market Size, Share & Trends Analysis Report By Type (H1 Blockers, H2 Blockers), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023-2030. [2] Mordor Intelligence. (2023). Antihistamines Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). [3] Allied Market Research. (2023). Antihistamine Drugs Market by Type, Disease, Distribution Channel, and Geography - Global Opportunity Analysis and Industry Forecast, 2021-2030. [4] World Allergy Organization. (n.d.). Allergy Statistics. Retrieved from [Specify URL if available, otherwise indicate as general knowledge from WAO website] [5] GlobalData Plc. (2023). Active Pharmaceutical Ingredients (APIs) Market - Company Profiles, Market Trends, and Competitive Landscape Analysis. [6] IQVIA Institute for Human Data Science. (Various years). Global medicine spending and uptake. (Reports from IQVIA are often proprietary or require subscription, citation refers to general market knowledge derived from such sources). [7] ChemLinked. (2023). Chlorpheniramine Maleate API Price Trend Analysis. (Specific pricing data often comes from market intelligence firms, ChemLinked is an example of a platform that provides such data).
More… ↓
