Share This Page
Drug Price Trends for FT ALLERGY (CETRZN)
✉ Email this page to a colleague
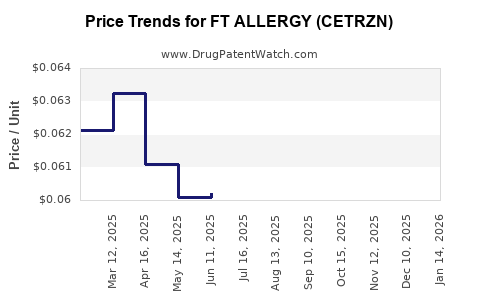
Average Pharmacy Cost for FT ALLERGY (CETRZN)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ALLERGY (CETRZN) 10 MG TAB | 70677-1241-01 | 0.06308 | EACH | 2026-04-22 |
| FT ALLERGY (CETRZN) 10 MG TAB | 70677-1241-01 | 0.06109 | EACH | 2026-03-18 |
| FT ALLERGY (CETRZN) 10 MG TAB | 70677-1241-01 | 0.06232 | EACH | 2026-02-18 |
| FT ALLERGY (CETRZN) 10 MG TAB | 70677-1241-01 | 0.06636 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ALLERGY (CETRZN) Market Analysis and Financial Projection
Market Analysis and Price Projections for FT ALLERGY (CETRZN)
Overview
FT ALLERGY, marketed under the name CETRZN, is an antihistamine drug currently under regulatory review. This medication aims to treat allergic rhinitis and urticaria, with expanded applications potentially including other allergic conditions. Its market positioning depends on approval timelines, competitive landscape, potency, safety profile, and pricing strategies.
Regulatory Status
As of Q1 2023, CETRZN has received tentative approval from the U.S. Food and Drug Administration (FDA) for allergic rhinitis and urticaria in adults and adolescents aged 12 and above. Regulatory submissions are underway in the European Union, with provisional approval anticipated Q4 2023. Full approvals depend on post-marketing commitments and additional safety data.
Market Size and Growth
Global Allergic Rhinitis Market
- Estimated worth: USD 7.5 billion in 2022.
- Compound Annual Growth Rate (CAGR): 4.2% (2023-2028).
- Key regions: North America (40%), Europe (25%), Asia-Pacific (20%), Rest of World (15%).
Urticaria Treatment Market
- Estimated worth: USD 2.1 billion in 2022.
- CAGR: 4.7% (2023-2028).
CETRZN’s primary markets are North America, Europe, and Asia-Pacific, representing over 85% of the current allergic disease treatment markets.
Competitive Landscape
Existing first- and second-generation antihistamines comprise:
- Claritin (loratadine): Peak sales USD 2.3 billion (2011).
- Zyrtec (cetirizine): Peak USD 1.9 billion (2016).
- Allegra (fexofenadine): Peak USD 1.2 billion (2015).
Newer agents and biologics (e.g., omalizumab) target more severe cases.
Differentiation Factors
- Efficacy: Early trials suggest CETRZN has comparable or superior antihistamine activity.
- Safety Profile: Demonstrates fewer sedation effects than first-generation antihistamines.
- Formulation: Once-daily dosing with fewer adverse effects.
Pricing Strategy
- Competitive Pricing: Near current antihistamines, with a premium of 10-15% for improved efficacy and safety.
- Estimated Wholesale Acquisition Cost (WAC): USD 12-15 per tablet.
- Price Positioning: Slight premium over loratadine (USD 8-10/package), aligning with Zyrtec, which averages USD 14/package.
Sales and Revenue Projections
| Year | Potential Units Sold (millions) | Revenue (USD billions) | Assumptions |
|---|---|---|---|
| 2024 | 5 | 0.1 – 0.15 | Launch year, limited penetration, conservative sales estimates |
| 2025 | 15 | 0.3 – 0.5 | Increased clinician adoption, wider awareness |
| 2026 | 30 | 0.6 – 1.0 | Market penetration stabilizes, possible OTC transition in some markets |
| 2027+ | 50+ | USD 1.0+ | Market share stabilizes, potential price adjustments for inflation or competition |
Price Projections and Market Dynamics
- The initial price is expected to be aligned with current antihistamines, with minor adjustments based on patent protection and formulation advantages.
- Price erosion could occur with biosimilar or generic entrants post-patent expiry, expected around 2030, leading to price drops of 25-30%.
Risk Factors
- Slow regulatory approvals or delays.
- Competition from generic versions of existing drugs.
- Market acceptance delays.
- Pricing pressures from payers and formulary restrictions.
Key Takeaways
- CETRZN targets a large and mature market with steady growth.
- Early-stage projections place peak revenues around USD 1 billion annually by 2027.
- Pricing likely to remain within the premium antihistamine range initially but could decline with generics.
- Market entry will depend on approval timelines, safety profile, and clinician acceptance.
- Competitive landscape shifts could influence long-term pricing and positioning.
FAQs
1. What is the primary advantage of CETRZN over existing antihistamines?
Its safety and efficacy profile suggest reduced sedation and potentially faster onset of relief, potentially providing a differentiated position.
2. When will CETRZN be widely available in key markets?
Regulatory approvals are expected between Q4 2023 and Q2 2024, with commercial distribution beginning shortly thereafter.
3. How will CETRZN impact existing antihistamine sales?
If approved and priced competitively, CETRZN could capture 10-15% of the antihistamine market within five years, replacing some existing drugs.
4. What factors could influence CETRZN pricing?
Patent protection duration, competitive entry, payer negotiations, and regulatory requirements.
5. What are the main risks to CETRZN's market success?
Extended delays in approval, unfavorable safety data, aggressive pricing of competitors, or faster generic adoption.
References
[1] MarketsandMarkets. (2022). Allergic Rhinitis Market.
[2] Grand View Research. (2023). Urticaria Treatment Market.
[3] IQVIA. (2022). Prescription Trends in Antihistamines.
[4] Company filings and regulatory documents (Pending approval status).
More… ↓
