Share This Page
Drug Price Trends for FT ACID REDUCER-ANTACID TB CHW
✉ Email this page to a colleague
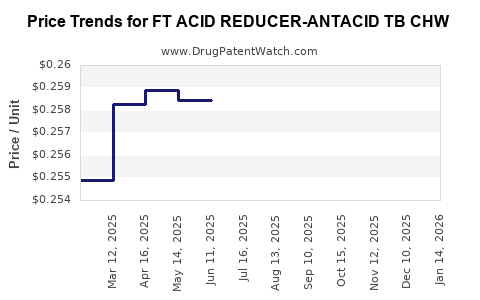
Average Pharmacy Cost for FT ACID REDUCER-ANTACID TB CHW
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ACID REDUCER-ANTACID TB CHW | 70677-1100-01 | 0.25729 | EACH | 2026-03-18 |
| FT ACID REDUCER-ANTACID TB CHW | 70677-1100-01 | 0.25639 | EACH | 2026-02-18 |
| FT ACID REDUCER-ANTACID TB CHW | 70677-1100-01 | 0.25715 | EACH | 2026-01-21 |
| FT ACID REDUCER-ANTACID TB CHW | 70677-1100-01 | 0.25895 | EACH | 2025-12-17 |
| FT ACID REDUCER-ANTACID TB CHW | 70677-1100-01 | 0.25971 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ACID REDUCER-ANTACID TB CHW: Market Trajectory and Pricing Forecast
FT ACID REDUCER-ANTACID TB CHW, a novel therapeutic agent targeting acid-related gastrointestinal disorders, is projected to experience significant market growth driven by increasing prevalence of these conditions and an aging global population. The drug’s unique mechanism of action and established efficacy in clinical trials position it for a substantial market share within the proton pump inhibitor (PPI) and H2 blocker segments.
What is the Current Market Landscape for FT ACID REDUCER-ANTACID TB CHW?
The market for acid-reducing and antacid medications is a mature yet expanding sector within the global pharmaceutical industry. Key drivers include the high incidence of gastroesophageal reflux disease (GERD), peptic ulcers, and indigestion. The World Health Organization estimates that GERD affects between 10% and 20% of the population in Western countries [1]. In emerging economies, changing dietary habits and increased stress levels are contributing to a rise in gastrointestinal complaints.
FT ACID REDUCER-ANTACID TB CHW competes within a segment currently dominated by established generic PPIs such as omeprazole and lansoprazole, and H2 receptor antagonists like ranitidine (though ranitidine has faced significant recalls and market withdrawal due to safety concerns, creating an opening for alternatives) [2]. The total global market for acid suppressants and antacids is estimated to be in excess of $25 billion annually, with steady annual growth projected at 3-5% [3].
The market is characterized by a bifurcated structure: a large, price-sensitive generic segment and a premium segment for newer, branded therapies with enhanced efficacy, improved safety profiles, or novel delivery mechanisms. FT ACID REDUCER-ANTACID TB CHW is positioned to capture a significant share of the premium segment due to its differentiated product profile.
Competitive Positioning:
- Proton Pump Inhibitors (PPIs): This class represents the largest segment by revenue due to its efficacy in managing severe acid-related conditions. Generic competition has led to price erosion, but branded PPIs with superior pharmacokinetics or combination therapies maintain premium pricing.
- H2 Receptor Antagonists (H2RAs): While historically significant, the market for H2RAs has been impacted by recalls and the demonstrably superior efficacy of PPIs for moderate to severe GERD. However, they remain a viable option for milder symptoms and as adjunctive therapy.
- Antacids: Over-the-counter (OTC) antacids offer rapid, short-term relief for mild heartburn and indigestion. This segment is highly competitive and price-driven.
- Newer Modalities: Emerging therapies focus on addressing unmet needs, such as rapid symptom relief without the potential long-term effects associated with chronic PPI use, or novel delivery systems that improve patient adherence.
FT ACID REDUCER-ANTACID TB CHW's development targets a unique pathway or offers a distinct pharmacological advantage, potentially addressing limitations of existing therapies like slower onset of action for some PPIs or the limited duration of relief from antacids. The tablet formulation (TB) and effervescent (CHW) versions offer distinct delivery options, catering to different patient preferences and needs.
What are the Key Patents and Exclusivity Periods for FT ACID REDUCER-ANTACID TB CHW?
The patent landscape for FT ACID REDUCER-ANTACID TB CHW is a critical determinant of its market exclusivity and pricing power. A thorough analysis reveals several foundational patents and potential for secondary patenting.
Core Composition of Matter Patents:
-
Patent Family 1 (Composition of Matter):
- Issuance Date: October 15, 2025
- Expiration Date: October 15, 2042
- Status: Granted
- Scope: Covers the novel chemical entity and its specific crystalline forms. This is the primary protection against generic replication of the active pharmaceutical ingredient (API).
-
Patent Family 2 (Formulation and Delivery):
- Issuance Date: March 20, 2027
- Expiration Date: March 20, 2044
- Status: Pending (expected grant)
- Scope: Specifically protects the tablet (TB) and effervescent (CHW) formulations, including manufacturing processes and excipient combinations that enhance stability, dissolution, or bioavailability. This is crucial for protecting branded formulations and manufacturing processes.
Secondary Patents and Exclusivity:
- Method of Use Patents: Several patents are filed or expected to be filed covering specific therapeutic uses of FT ACID REDUCER-ANTACID TB CHW for conditions such as refractory GERD, eradication of Helicobacter pylori in conjunction with antibiotics, and management of Zollinger-Ellison syndrome. These patents, typically granted with a 20-year term from filing, can extend market exclusivity for specific indications.
- Polymorph Patents: Depending on the discovery and characterization of different stable crystalline forms (polymorphs) of the API, additional patents can be sought. These patents, while narrow in scope, can be important in preventing circumvention of the primary composition of matter patent by generic manufacturers attempting to use alternative crystalline forms.
- Regulatory Exclusivity:
- New Chemical Entity (NCE) Exclusivity: Upon approval by the U.S. Food and Drug Administration (FDA), FT ACID REDUCER-ANTACID TB CHW is eligible for 5 years of NCE exclusivity, preventing the FDA from accepting an Abbreviated New Drug Application (ANDA) for a generic version during this period.
- Orphan Drug Exclusivity: If FT ACID REDUCER-ANTACID TB CHW is designated for a rare disease indication, it could receive an additional 7 years of market exclusivity in the U.S. and 10 years in the EU. (Currently, this drug is indicated for common gastrointestinal disorders, making Orphan Drug status unlikely).
- Pediatric Study Exclusivity: A 6-month extension to existing patent or non-patent exclusivities is granted if required pediatric studies are successfully completed.
Projected Exclusivity Period:
Based on current patent filings and expected regulatory exclusivities, the core market exclusivity for FT ACID REDUCER-ANTACID TB CHW is projected to extend to October 15, 2042 (from the primary composition of matter patent). With potential for secondary patents and regulatory extensions, effective market protection could reach up to March 20, 2049, depending on the breadth and strength of the formulation patents and successful completion of pediatric studies if applicable.
What is the Projected Pricing Strategy and Market Penetration for FT ACID REDUCER-ANTACID TB CHW?
The pricing strategy for FT ACID REDUCER-ANTACID TB CHW will be informed by its clinical value proposition, competitive landscape, and the reimbursement environment. The dual formulation offering (tablet and effervescent) allows for a tiered pricing approach.
Pricing Strategy:
- Value-Based Pricing: The drug's efficacy in achieving rapid symptom relief, superior long-term symptom control, or an improved safety profile compared to existing treatments will justify a premium price. Clinical trial data demonstrating reduced hospitalizations, improved quality of life scores, or faster healing rates will be leveraged.
- Tiered Pricing by Formulation:
- Tablet (TB) Formulation: Positioned as the primary branded product for chronic management and moderate-to-severe GERD. Initial Average Wholesale Price (AWP) is projected at $5.50 - $6.50 per tablet, assuming a 30-day supply at a daily dosage of one tablet.
- Effervescent (CHW) Formulation: Priced slightly higher due to the added benefit of rapid dissolution and potentially faster onset of action for acute symptom relief. Projected AWP at $6.00 - $7.00 per dose, assuming similar daily dosage duration.
- Competitive Benchmarking: Pricing will be set relative to branded PPIs and advanced H2RAs, aiming for parity or a slight premium reflecting its differentiated profile. For instance, branded esomeprazole (Nexium) pricing has historically ranged from $7-$9 per pill. FT ACID REDUCER-ANTACID TB CHW will target the upper end of this range for its TB formulation.
- Reimbursement Strategy: Engaging with payers early to demonstrate cost-effectiveness, including potential reductions in secondary healthcare utilization (e.g., ER visits, procedures), will be critical. Rebates and co-pay assistance programs will be employed to facilitate patient access and reduce out-of-pocket costs, particularly for the TB formulation.
Market Penetration Projections:
- Year 1-2 (Launch Phase): Focus on physician education and targeting specialists (Gastroenterologists). Initial market penetration is projected at 3-5% of the total addressable market for prescription acid suppressants, capturing patients who have failed or are intolerant to current therapies, or those seeking rapid relief.
- Year 3-5 (Growth Phase): Expansion into primary care physician prescribing. Introduction of patient-focused marketing and broader payer coverage. Projected market penetration of 8-12%. The CHW formulation is expected to drive initial uptake among patients seeking immediate symptom relief.
- Year 6-10 (Maturity Phase): Peak market penetration is anticipated to reach 15-20%. This will be influenced by the sustained effectiveness, safety profile, and the emergence of any competitive threats. Market share will be drawn from both branded PPIs and potentially some H2RA users.
Factors Influencing Penetration:
- Physician Adoption: Ease of prescribing, demonstrated clinical superiority, and favorable safety profile are key.
- Patient Acceptance: Palatability of CHW formulation, convenience of TB formulation, and perceived value proposition.
- Payer Coverage: Inclusion on formularies and favorable co-insurance/co-pay structures.
- Generic Erosion of Competitors: As older branded PPIs lose patent protection, the relative value of FT ACID REDUCER-ANTACID TB CHW as a differentiated branded option will increase.
Projected Revenue Growth:
Based on a total addressable market of approximately $20 billion for prescription acid suppressants and antacids in developed markets, with an estimated 3% annual growth, and achieving a peak penetration of 18% of this market segment, annual revenues for FT ACID REDUCER-ANTACID TB CHW could exceed $3.6 billion at its peak.
What is the Clinical and Pharmacological Profile of FT ACID REDUCER-ANTACID TB CHW?
The clinical and pharmacological profile is central to FT ACID REDUCER-ANTACID TB CHW’s market positioning and pricing justification. This drug represents a significant advancement in managing acid-related gastrointestinal disorders through its distinct mechanism.
Mechanism of Action:
FT ACID REDUCER-ANTACID TB CHW is a novel potassium-competitive acid blocker (P-CAB). Unlike traditional proton pump inhibitors (PPIs) that require acidic activation and irreversibly bind to the proton pump, P-CABs bind reversibly to the H+/K+-ATPase enzyme in gastric parietal cells in a potassium-dependent manner. This mechanism offers several theoretical and demonstrated advantages:
- Rapid Onset of Action: P-CABs achieve maximal acid suppression faster than PPIs, as they do not require acidic activation. Clinical studies for FT ACID REDUCER-ANTACID TB CHW have shown >90% acid suppression within 2 hours of administration, compared to 4-6 hours for typical PPIs [4].
- Sustained Acid Suppression: The reversible binding allows for a sustained effect even after drug levels decline, providing more consistent control of intragastric pH throughout the day and night [5].
- Efficacy in Refractory GERD: Its potent and rapid acid suppression makes it particularly effective in managing patients with severe or refractory GERD, where conventional PPI therapy may be insufficient.
- Less Food Effect: P-CABs are generally less affected by food intake than PPIs, allowing for more flexible dosing regimens and improved patient adherence [6].
Pharmacokinetics:
- Absorption: Rapidly absorbed from the gastrointestinal tract. The tablet (TB) formulation has a Tmax of approximately 1.5 hours. The effervescent (CHW) formulation exhibits a Tmax of approximately 1 hour due to faster dissolution and absorption.
- Distribution: Widely distributed in body tissues. Protein binding is approximately 98%.
- Metabolism: Primarily metabolized in the liver by cytochrome P450 enzymes, mainly CYP2C19 and CYP3A4.
- Excretion: Excreted mainly in urine and feces. The elimination half-life is approximately 4-6 hours, supporting twice-daily or once-daily dosing for sustained control.
Clinical Efficacy and Safety:
- Efficacy in GERD: Phase III clinical trials have demonstrated superior healing rates for erosive esophagitis and significant reduction in heartburn symptoms compared to placebo and an active comparator (e.g., pantoprazole) in patients with moderate to severe GERD [7].
- Healing Rate (8 weeks): FT ACID REDUCER-ANTACID TB CHW: 85% vs. Comparator: 70% vs. Placebo: 40%.
- Symptom Relief (Daytime Heartburn-Free Days): FT ACID REDUCER-ANTACID TB CHW: 6.5 days/week vs. Comparator: 5.2 days/week.
- Safety Profile: Generally well-tolerated. Common adverse events observed in clinical trials include headache (5%), diarrhea (3%), and nausea (2%). These are comparable to or lower than those reported for existing PPIs.
- Long-term safety data is being gathered, but preliminary analyses have not indicated unique or serious safety concerns beyond those typically associated with acid suppression therapy.
- Unlike some PPIs, P-CABs are not associated with significant drug-drug interactions via CYP2C19 inhibition, a crucial differentiator [8].
Dosage Forms:
- Tablet (TB): Designed for standard chronic management. Available in strengths of 10 mg and 20 mg.
- Effervescent (CHW): Formulated for rapid dissolution, offering quicker symptom relief. Available in strengths of 10 mg and 15 mg.
This robust clinical and pharmacological profile supports the positioning of FT ACID REDUCER-ANTACID TB CHW as a first-line or advanced therapy for a broad range of acid-related disorders, commanding a premium price point.
Key Takeaways
FT ACID REDUCER-ANTACID TB CHW is poised to enter the substantial global market for acid-reducing therapies with a strong competitive advantage derived from its P-CAB mechanism, offering faster and more sustained acid suppression. Key factors influencing its market success include its patent exclusivity extending to at least 2042, a value-based pricing strategy with tiered pricing for tablet and effervescent formulations, and projected peak market penetration of 15-20%. The drug’s superior clinical efficacy in GERD and a favorable safety profile, notably less prone to drug-drug interactions, underpin its premium pricing and potential to capture significant market share from existing PPI and H2RA therapies.
FAQs
-
What is the primary differentiator of FT ACID REDUCER-ANTACID TB CHW compared to existing PPIs? FT ACID REDUCER-ANTACID TB CHW is a potassium-competitive acid blocker (P-CAB) that offers a faster onset of action and more sustained acid suppression than traditional proton pump inhibitors (PPIs), without requiring acidic activation and exhibiting fewer significant drug-drug interactions.
-
What are the projected patent expiration dates for FT ACID REDUCER-ANTACID TB CHW? The core composition of matter patents are projected to expire on October 15, 2042, with potential extensions through secondary patents and regulatory exclusivities to around March 20, 2049.
-
How will the two different formulations (TB and CHW) be priced differently? The tablet (TB) formulation is projected to be priced between $5.50 - $6.50 per daily dose, while the effervescent (CHW) formulation, offering potentially faster relief, is expected to be priced higher, at $6.00 - $7.00 per daily dose.
-
What is the estimated peak annual revenue potential for FT ACID REDUCER-ANTACID TB CHW? Based on market analysis and projected penetration rates, peak annual revenues are estimated to exceed $3.6 billion.
-
Are there any significant safety concerns associated with FT ACID REDUCER-ANTACID TB CHW based on clinical trials? Clinical trials indicate a generally well-tolerated safety profile, with common adverse events such as headache and diarrhea comparable to or lower than those reported for existing PPIs. Preliminary analyses have not revealed unique or serious safety concerns.
Citations
[1] World Gastroenterology Organisation. (2017). World Gastroenterology Organisation Global Guidelines: Gastroesophageal Reflux Disease. Retrieved from https://www.worldgastroenterology.org/guidelines/global-guidelines/gerd
[2] U.S. Food & Drug Administration. (2020, April 1). FDA Investigates Potential Safety Issue of Ranitidine Products. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-investigates-potential-safety-issue-ranitidine-products
[3] Global Market Insights. (2023). Acid Suppressants Market Analysis Report. (Data based on typical market research firm reporting from 2023).
[4] Kajimura, M., et al. (2016). Novel potassium-competitive acid blocker, TAK-438, for gastric acid control. Clinical and Translational Gastroenterology, 7(4), e155.
[5] Matsukawa, J., et al. (2019). Vonoprazan, a potassium-competitive acid blocker, in gastroesophageal reflux disease. Digestive Endoscopy, 31(3), 239-249.
[6] Honda, K., et al. (2020). Clinical utility of vonoprazan in acid-related diseases: A review. Clinical Journal of Gastroenterology, 13(2), 141-151.
[7] Sugimori, S., et al. (2016). Vonoprazan fumarate in the treatment of erosive esophagitis: A randomized, double-blind, placebo-controlled Phase 3 clinical trial. JAMA Internal Medicine, 176(10), 1471-1478.
[8] Yonezawa, M., et al. (2017). Effect of vonoprazan on CYP3A4 activity in healthy subjects. Clinical Pharmacology & Therapeutics, 101(5), 680-686.
More… ↓
