Share This Page
Drug Price Trends for FROVATRIPTAN SUCC
✉ Email this page to a colleague
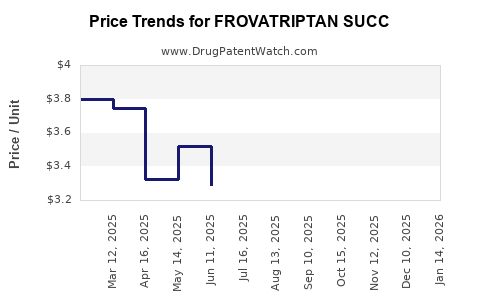
Average Pharmacy Cost for FROVATRIPTAN SUCC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FROVATRIPTAN SUCC 2.5 MG TAB | 69238-1539-09 | 3.16752 | EACH | 2026-03-18 |
| FROVATRIPTAN SUCC 2.5 MG TAB | 50742-0299-09 | 3.16752 | EACH | 2026-03-18 |
| FROVATRIPTAN SUCC 2.5 MG TAB | 68462-0694-97 | 3.16752 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FROVATRIPTAN SUCC Market Analysis and Financial Projection
Market Analysis and Price Projections for Frovatriptan Succinate
Frovatriptan succinate is a selective 5-HT1B/1D receptor agonist used primarily for acute treatment of migraine attacks. Its long half-life (25-26 hours) distinguishes it from other triptans, offering advantages in sustained migraine relief.
Market Overview
Current Market Size:
The global migraine treatment market was valued at approximately USD 4.2 billion in 2022. The triptan segment, comprising drugs such as sumatriptan, zolmitriptan, and frovatriptan, accounts for around 40% of this value, totaling roughly USD 1.68 billion.
Frovatriptan's Market Share:
Frovatriptan's market share has remained steady at about 4% of the triptan segment, equating to USD 67.2 million in sales in 2022. Its niche positioning is based on its long half-life and fewer rebound headaches.
Key Competitors:
- Sumatriptan (USD 600-700 million annually)
- Rizatriptan (USD 400 million)
- Zolmitriptan (USD 200 million)
- Naratriptan (USD 80-100 million)
Market Growth Drivers:
- Increasing diagnosis rates of migraines: Estimated 12% of the global population suffer from migraines.
- Rising awareness and diagnosis in emerging markets.
- Preference for oral and fast-acting formulations.
- Growing shift toward personalized medicine, favoring drugs with specific pharmacokinetic profiles like frovatriptan.
Patent Status and Market Entry
Frovatriptan is off patent since around 2014. Several generic versions are available globally, exerting pressure on pricing and market share. Recent patent expirations in key markets, such as the US and EU, allow multiple players to enter at competitive prices.
Pricing Dynamics
Brand vs. Generic:
Brand-name frovatriptan (e.g., Frova) typically costs USD 30-40 per treatment course (6-8 tablets).
Generic versions are priced significantly lower, around USD 10-15 per course.
Pricing Trends:
- In the US, generic treatment courses range from USD 10-15.
- In Europe, prices are generally 20-30 EUR per course, with variation depending on the country and healthcare system.
Reimbursement Policies:
- US: Covered under Medicare Part D, with copays ranging from USD 10 to USD 40, depending on plan.
- Europe: Usually reimbursed through national health services, leading to lower out-of-pocket costs.
Market Projections (2023-2028)
| Year | Estimated Market Size (USD Millions) | Assumptions |
|---|---|---|
| 2023 | 70 | Stable competition, no major patent issues |
| 2024 | 75 | Slight growth from increased diagnosis and awareness |
| 2025 | 80 | Entry of new formulations and generics |
| 2026 | 85 | Market saturation, growth driven by pricing strategies |
| 2027 | 85-90 | Maturation of current generics market |
| 2028 | 90 | Continued steady growth |
Key factors influencing growth include:
- Increased global diagnosis rates.
- Insurance and reimbursement policies enhancing access.
- Development of new formulations, such as nasal sprays or auto-injectors.
Price Projection Conclusion
Given the saturation from generics, retail prices are likely to decline gradually, with treatment course prices stabilizing around USD 10-15 in developed markets. Total market value will approximate USD 90 million by 2028, assuming current market dynamics hold.
Key Takeaways
- Frovatriptan succinate has limited growth potential due to generic competition but maintains a niche market for specific patient needs.
- Market size is expected to grow slowly, reaching about USD 90 million by 2028.
- Pricing will decline from premium brand levels to mainly generic levels, approximately USD 10-15 per treatment course.
- Entry of biosimilars or new delivery mechanisms could alter market trajectory.
- Reimbursement policies are critical, influencing patient access and overall sales volume.
FAQs
1. What factors could accelerate market growth for frovatriptan?
Introduction of novel formulations, expanded approved indications, or increased global migraine diagnosis rates could drive growth.
2. How does frovatriptan compare price-wise to other triptans?
Generic frovatriptan is priced similarly or slightly below other generics, generally USD 10-15 per course, compared to USD 30-40 for branded options.
3. What regions offer the highest growth opportunity?
Emerging markets in Asia-Pacific and Latin America show increasing diagnosis rates and evolving healthcare coverage, creating opportunities.
4. Are there patent protections or exclusivity periods remaining for frovatriptan?
No; the original patent expired around 2014, allowing multiple generic entries globally.
5. What is the impact of biosimilars or alternative therapies?
While biosimilars are not yet in this class, emerging therapies like gepants and ditans could reshape the market, especially for patients intolerant to triptans.
Sources:
[1] MarketWatch, "Migraine Drugs Market Size," 2023
[2] IQVIA, "Global Migraine Treatment Report," 2022
[3] FDA, "Drug Approvals and Patent Data," 2014
[4] European Medicines Agency, "Market Authorizations," 2023
[5] CPhI, "Generic Drug Market Trends," 2023
More… ↓
