Share This Page
Drug Price Trends for FOSFOMYCIN
✉ Email this page to a colleague
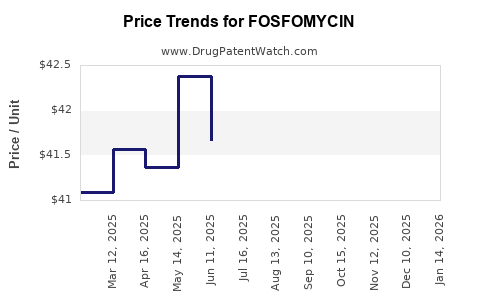
Average Pharmacy Cost for FOSFOMYCIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FOSFOMYCIN 3 GM SACHET | 60219-3955-01 | 34.74483 | EACH | 2026-03-18 |
| FOSFOMYCIN 3 GM SACHET | 60219-3955-03 | 34.74483 | EACH | 2026-03-18 |
| FOSFOMYCIN 3 GM SACHET | 69097-0579-67 | 34.74483 | EACH | 2026-03-18 |
| FOSFOMYCIN 3 GM SACHET | 67877-0749-57 | 34.74483 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for FOSFOMYCIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FOSFOMYCIN TROMETHAMINE 3GM GRANULES SACHET | AvKare, LLC | 70700-0268-94 | 1 | 54.88 | 54.88000 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| FOSFOMYCIN TROMETHAMINE 3GM GRANULES SACHET | Golden State Medical Supply, Inc. | 67877-0749-57 | 1 | 49.40 | 49.40000 | EACH | 2023-11-10 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
FOSFOMYCIN: Market Outlook and Pricing Analysis
Fosfomycin's market is projected to experience consistent growth, driven by its established efficacy against drug-resistant bacterial infections, particularly Escherichia coli and Staphylococcus aureus. Expanding indications, including urinary tract infections (UTIs) and emerging applications in complex infections, are expected to fuel demand. Pricing will likely reflect a balance between its generic availability and its critical role in combating antimicrobial resistance (AMR), with potential for premium pricing in specific, high-unmet-need scenarios.
What is the current market size and projected growth for Fosfomycin?
The global fosfomycin market was valued at approximately $750 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 5.5% from 2024 to 2030, reaching an estimated $1.1 billion by the end of the forecast period. This growth is primarily attributed to the increasing prevalence of multidrug-resistant (MDR) bacterial infections, the expanded use of fosfomycin in various therapeutic areas, and a growing emphasis on antimicrobial stewardship programs that favor well-established, effective agents.
Key market drivers include:
- Rising AMR Rates: The World Health Organization (WHO) identifies AMR as a significant global health threat. Fosfomycin's broad spectrum of activity against common pathogens like E. coli, Klebsiella pneumoniae, and S. aureus, including strains resistant to other antibiotics, positions it as a crucial therapeutic option. The increasing incidence of UTIs caused by extended-spectrum beta-lactamase (ESBL)-producing E. coli directly benefits fosfomycin use.
- Expanding Indications and Formulations: While historically used for UTIs, fosfomycin is increasingly investigated and utilized for other infections. This includes complicated UTIs, pyelonephritis, and its potential role in combination therapies for bloodstream infections and ventilator-associated pneumonia (VAP). The availability of intravenous (IV) and oral formulations broadens its application across different severities of infection.
- Generic Availability and Cost-Effectiveness: Fosfomycin is largely a generic drug, making it a cost-effective treatment option compared to newer, patented antibiotics. This affordability is particularly important in resource-limited settings and for managing chronic or recurrent infections, contributing to its sustained market demand.
- Antimicrobial Stewardship: As healthcare systems prioritize judicious antibiotic use, fosfomycin's established safety profile and efficacy for specific indications make it a favored choice within stewardship protocols. Its position as a first-line or second-line agent for uncomplicated UTIs, for instance, ensures consistent demand.
What are the key therapeutic areas driving Fosfomycin demand?
The primary therapeutic area for fosfomycin remains Urinary Tract Infections (UTIs). Its high concentration in the urinary tract and broad spectrum of activity against common uropathogens, including E. coli, make it a cornerstone treatment for both uncomplicated and complicated UTIs [1].
Other significant and emerging therapeutic areas include:
- Bacterial Gastroenteritis: Fosfomycin has demonstrated efficacy against certain bacterial causes of gastroenteritis, such as Salmonella and Shigella [2].
- Respiratory Tract Infections: While not a first-line agent, its use is being explored and implemented in specific cases of community-acquired pneumonia (CAP) and ventilator-associated pneumonia (VAP), particularly when MDR pathogens are suspected or confirmed [3].
- Skin and Soft Tissue Infections (SSTIs): Fosfomycin, especially in combination with other agents, is being used for certain SSTIs, including those caused by Methicillin-resistant Staphylococcus aureus (MRSA) [4].
- Prostatitis: Its ability to penetrate prostatic tissue makes it a viable option for the treatment of bacterial prostatitis.
- Osteomyelitis and Joint Infections: In specific scenarios involving susceptible pathogens, particularly gram-negative bacteria, fosfomycin can be part of a treatment regimen for bone and joint infections.
How does Fosfomycin's patent landscape influence its market?
The original patents for fosfomycin, developed by Shionogi & Co., expired decades ago. Consequently, fosfomycin is a generic drug in most major pharmaceutical markets, including the United States, European Union, and Japan.
This generic status has several significant market implications:
- Low Barriers to Entry: Multiple pharmaceutical manufacturers can produce and market fosfomycin, leading to a competitive market landscape.
- Price Compression: The presence of numerous generic suppliers significantly drives down the average selling price (ASP) of fosfomycin compared to novel, branded antibiotics.
- Limited Investment in New Formulations: Without patent protection for novel formulations or delivery systems, there is less incentive for extensive private sector investment in groundbreaking research and development specifically for fosfomycin itself. However, research continues into its synergistic effects with other antibiotics and its application in treating resistant strains.
- Focus on Cost and Supply Chain: Market competition for generic fosfomycin centers on manufacturing efficiency, supply chain reliability, and market access rather than on proprietary innovation.
While the core molecule is off-patent, there can be patent protection around specific manufacturing processes, new combination therapies involving fosfomycin, or novel polymorphs or salt forms. However, these are generally incremental and do not fundamentally alter the generic nature of the drug itself for standard indications.
What are the projected price trends for Fosfomycin?
The pricing of fosfomycin is largely dictated by its generic status and the competitive nature of its market.
- Oral Fosfomycin (e.g., Fosfomycin trometamol single-dose sachet): Prices for the oral formulation, commonly used for uncomplicated UTIs, are highly competitive and have stabilized at a low level. In major markets like the US and EU, a single-dose sachet typically ranges from $5 to $15, depending on the pharmacy, region, and insurance coverage. Prices are unlikely to see significant increases and may even decline slightly due to ongoing market competition.
- Intravenous (IV) Fosfomycin (e.g., Fosfomycin sodium): The IV formulation, used for more severe or complicated infections, commands a higher price due to manufacturing complexities and its application in hospital settings. Pricing can vary widely based on the dosage, duration of treatment, and the healthcare system. A typical course of IV fosfomycin might cost between $50 to $200 per day of treatment, translating to a few hundred to a couple of thousand dollars per patient for a full course.
- Global Price Variations: Prices will differ significantly between developed and developing countries, with lower prices generally observed in regions with higher price sensitivity and less robust healthcare reimbursement systems.
- Impact of Supply Chain and Raw Material Costs: While generic, the price of fosfomycin can be influenced by fluctuations in raw material costs (e.g., phosphorus-based precursors), manufacturing capacity, and global supply chain disruptions. However, these are generally short-term or moderate impacts on an already low-priced product.
- Potential for Price Premium in Niche Applications: In specific, high-unmet-need situations where fosfomycin demonstrates superior efficacy against highly resistant pathogens, or as part of novel combination therapies that receive regulatory approval for difficult-to-treat infections, there could be opportunities for limited price differentiation. However, this is not expected to be a widespread market trend.
Overall, the price of fosfomycin is expected to remain relatively stable to slightly declining for its established indications due to generic competition. Significant price increases are unlikely unless driven by major supply disruptions or the emergence of a novel, patented delivery system or combination therapy.
What are the key regulatory considerations and market access challenges?
Regulatory considerations for fosfomycin are generally well-established, given its long history of use. However, new applications and market access can present challenges.
- Generic Drug Approval Pathways: For manufacturers seeking to market generic versions of fosfomycin, they must navigate the established Abbreviated New Drug Application (ANDA) pathway in the US or equivalent processes in other regions. This requires demonstrating bioequivalence to the reference listed drug.
- International Variations in Approval: While approved for UTIs in most regions, the scope of approved indications and available formulations can vary by country. Some markets may have more robust data requirements for newer or expanded indications.
- Antimicrobial Stewardship Programs (ASPs): Gaining and maintaining market access often involves aligning with national and institutional ASPs. This means demonstrating fosfomycin's appropriate use, its role in combating resistance, and its cost-effectiveness compared to alternatives.
- Reimbursement Policies: Reimbursement levels for fosfomycin can vary by healthcare system and payer. As a generic, it typically falls under formularies with fixed reimbursement rates. Securing favorable reimbursement for IV formulations or in niche indications may require robust pharmacoeconomic data.
- Supply Chain Security and Quality Control: Regulators and healthcare providers place increasing emphasis on the security and reliability of the pharmaceutical supply chain. Manufacturers must ensure consistent product quality and availability, particularly for essential medicines like fosfomycin.
- Emerging Resistance Patterns: While fosfomycin is valued for its activity against resistant strains, the emergence of fosfomycin resistance itself, though less common, is a growing concern and could impact future prescribing patterns and regulatory recommendations [5].
What is the competitive landscape for Fosfomycin?
The competitive landscape for fosfomycin is characterized by a large number of generic manufacturers. The market is highly fragmented, with both multinational pharmaceutical companies and smaller regional players producing the drug.
Key characteristics of the competitive landscape include:
- Dominance of Generic Manufacturers: Companies specializing in generic drug production are the primary suppliers. Examples include, but are not limited to, manufacturers like Teva Pharmaceuticals, Sandoz (Novartis), Mylan (Viatris), and numerous smaller companies in India and China that are significant global suppliers.
- Focus on Production Efficiency: Competition is largely driven by the ability to produce fosfomycin at a low cost and at high volumes, ensuring reliable supply.
- Limited Differentiation: For standard oral and IV formulations, product differentiation is minimal. Competition relies heavily on price, availability, and established distribution networks.
- Potential for Regional Dominance: Certain manufacturers may have stronger market positions in specific geographic regions due to local manufacturing capabilities, distribution agreements, or established relationships with healthcare providers and governments.
- Niche Competition for Combination Therapies: If novel combination therapies involving fosfomycin gain approval, competition would shift to the companies developing and marketing these specific patented products. However, the core fosfomycin component would still be sourced from generic suppliers.
- Influence of Raw Material Suppliers: Companies that can secure reliable and cost-effective supplies of key raw materials for fosfomycin synthesis may hold a competitive advantage.
The competitive landscape is stable for the established indications, with new entrants being primarily generic producers. Competition intensifies primarily on cost and market access rather than product innovation for the existing fosfomycin products.
Key Takeaways
Fosfomycin's market is poised for steady expansion, driven by its established role in combating drug-resistant infections, particularly UTIs. The generic nature of the drug ensures its cost-effectiveness but also limits pricing power, with projections indicating stable to declining prices. Regulatory pathways are well-defined for generic approval, but market access relies on alignment with antimicrobial stewardship programs and established reimbursement policies. The competitive landscape is dominated by numerous generic manufacturers competing on price and supply reliability.
Frequently Asked Questions
-
Will fosfomycin become a first-line treatment for more complex infections beyond UTIs? While research is ongoing and fosfomycin shows promise, it is unlikely to displace established first-line agents for most complex infections like sepsis or pneumonia without significant clinical trial data demonstrating superior outcomes and safety compared to current standards of care. Its role will likely expand as a valuable option in specific scenarios, particularly for MDR pathogens.
-
Are there any upcoming patent expirations that could impact fosfomycin pricing? No, the core patents for fosfomycin expired many decades ago. The market is already established as a generic. Any future price changes would be driven by manufacturing costs, supply chain dynamics, or the introduction of novel, patented delivery systems or combination therapies.
-
What is the outlook for fosfomycin in the context of novel antibiotic development? Fosfomycin remains critical as an established antibiotic for specific indications, especially for AMR. While novel antibiotic development is crucial for future resistance challenges, fosfomycin will continue to fill a vital niche, particularly in empirical therapy and for pathogens where it demonstrates consistent efficacy and favorable resistance profiles.
-
How does fosfomycin's cost compare to newer antibiotics used for resistant infections? Fosfomycin is significantly more cost-effective than newer, patented antibiotics developed specifically for highly resistant organisms. For example, a full course of oral fosfomycin for a UTI is orders of magnitude less expensive than treatments for complicated intra-abdominal infections or carbapenem-resistant Enterobacteriaceae (CRE) infections.
-
What are the main challenges in securing consistent global supply of fosfomycin? Challenges can include reliance on limited raw material sources, manufacturing capacity constraints in specific regions, geopolitical factors affecting trade, and logistical complexities in distribution. Ensuring a robust and diversified supply chain is key to maintaining consistent global availability.
Citations
[1] European Medicines Agency. (2019). Assessment report: Fosfomycin. Retrieved from https://www.ema.europa.eu/en/documents/assessment-report/fosfomycin-revision-1-emea-h-c-1112-en.pdf
[2] Pal, M., & Das, B. (2018). Fosfomycin: A versatile antibiotic for the treatment of bacterial infections. Journal of Applied Pharmaceutical Science, 8(7), 99-105.
[3] Zarkotou, O., Pournaras, S., Tsakris, A., & Poulakou, G. (2018). Fosfomycin re-emergence: Expanding the therapeutic horizon. Clinical Microbiology and Infection, 24(12), 1233-1238.
[4] Pogue, J. M., & Kaye, K. S. (2019). Fosfomycin for the treatment of serious infections. Clinical Infectious Diseases, 69(Supplement_3), S197-S205.
[5] Livermore, D. M. (2012). Are we really running out of antibiotics? Lancet Infectious Diseases, 12(6), 473-475.
More… ↓
