Share This Page
Drug Price Trends for FLUPHENAZINE DEC
✉ Email this page to a colleague
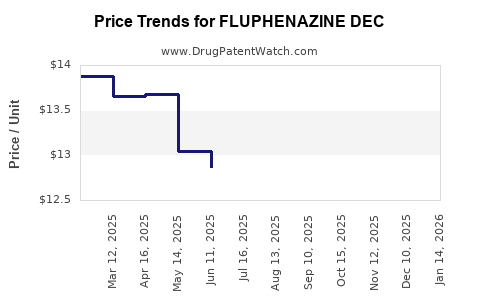
Average Pharmacy Cost for FLUPHENAZINE DEC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUPHENAZINE DEC 125 MG/5 ML | 72205-0100-01 | 9.48720 | ML | 2026-05-20 |
| FLUPHENAZINE DEC 125 MG/5 ML | 00143-9529-01 | 9.48720 | ML | 2026-05-20 |
| FLUPHENAZINE DEC 125 MG/5 ML | 25021-0838-05 | 9.48720 | ML | 2026-05-20 |
| FLUPHENAZINE DEC 125 MG/5 ML | 42023-0129-89 | 9.48720 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Fluphenazine Decanoate Market Analysis and Price Projections
Fluphenazine decanoate is an antipsychotic medication administered via intramuscular injection. It is a long-acting injectable (LAI) formulation of fluphenazine, a phenothiazine derivative, used primarily in the management of schizophrenia and other psychotic disorders. Its sustained release mechanism reduces the frequency of dosing compared to oral formulations, improving patient adherence and therapeutic outcomes.
What is the Current Market Size and Growth Trajectory for Fluphenazine Decanoate?
The global market for fluphenazine decanoate is characterized by its established presence and steady demand, particularly within healthcare systems prioritizing LAI antipsychotics for chronic mental illness management. While specific market size figures for fluphenazine decanoate alone are not extensively granular in public domain reports, it is a component of the broader long-acting injectable antipsychotic market. This segment is projected to grow due to increasing awareness of LAI benefits, a rise in schizophrenia prevalence, and a push for improved treatment adherence.
The global LAI antipsychotic market was valued at approximately USD 6.2 billion in 2022 and is forecast to reach USD 10.5 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 6.8% from 2023 to 2030 [1]. Fluphenazine decanoate, as one of the older LAI antipsychotics, occupies a significant, albeit mature, segment within this market. Its growth is influenced by competition from newer LAI agents, but its cost-effectiveness and established efficacy maintain its relevance.
Key Market Drivers:
- Improved Patient Adherence: LAIs significantly reduce missed doses compared to oral medications, leading to better symptom control and reduced relapse rates [2].
- Chronic Disease Management: Schizophrenia is a chronic condition requiring long-term treatment. LAIs offer a convenient and effective solution for continuous care.
- Cost-Effectiveness: In certain healthcare economies, fluphenazine decanoate offers a more affordable LAI option compared to newer, more expensive alternatives.
- Physician Preference and Experience: Clinicians familiar with fluphenazine decanoate’s profile continue to prescribe it based on positive patient outcomes and established treatment protocols.
Market Restraints:
- Competition from Newer LAIs: Newer agents, such as paliperidone palmitate and aripiprazole lauroxil, offer different dosing intervals, improved side effect profiles, and novel mechanisms of action, potentially cannibalizing market share.
- Injection Site Reactions and Side Effects: Like other injectable antipsychotics, fluphenazine decanoate can cause injection site pain, swelling, and other side effects, which may impact patient acceptance.
- Limited New Market Entrants: The development of new fluphenazine decanoate formulations or significant market expansion is less likely given its mature status and the focus on newer drug classes.
What are the Key Patent Expirations and Generics Impacting Fluphenazine Decanoate?
Fluphenazine decanoate, as a drug first introduced in the 1960s, has long-expired foundational patents. The molecule itself is off-patent, allowing for generic manufacturing. The primary intellectual property considerations for fluphenazine decanoate now revolve around formulation patents, manufacturing processes, and potentially specific delivery systems that might offer novel aspects like extended release profiles beyond the standard decanoate ester.
Original composition of matter patents for fluphenazine decanoate expired decades ago. Therefore, generic versions have been available in the market for a considerable period. The competitive landscape is largely populated by generic manufacturers.
Patent Landscape Considerations:
- Composition of Matter Patents: Expired.
- Formulation Patents: While the decanoate ester formulation is old, there could be existing patents on specific excipients, crystallization methods, or advanced delivery technologies designed to further refine its release profile or reduce injection site reactions. These patents, if they exist and are still in force, would be crucial for any company seeking to introduce a differentiated generic product.
- Manufacturing Process Patents: Proprietary manufacturing techniques can be patented, providing a competitive advantage even when the drug substance is generic.
- Evergreening Strategies: Companies might have pursued patents on minor improvements or new uses, but these are unlikely to represent significant barriers to generic entry for the established decanoate form.
The market for fluphenazine decanoate is therefore a generic-dominated market, where price is a primary competitive factor.
What are the Projected Price Trends for Fluphenazine Decanoate?
Given the generic nature of fluphenazine decanoate and the mature market for LAI antipsychotics, price projections are influenced by generic competition, healthcare reimbursement policies, and the overall market dynamics of older antipsychotic medications.
Historical and Current Pricing:
Historically, fluphenazine decanoate has been one of the more affordable LAI antipsychotic options. Its price has been relatively stable, with fluctuations primarily driven by generic manufacturer competition and the cost of raw materials. Wholesale acquisition costs (WAC) for fluphenazine decanoate injections (typically 25 mg/mL and 50 mg/mL concentrations) have ranged from approximately USD 15 to USD 50 per mL, depending on the manufacturer and the specific product presentation (e.g., vial, pre-filled syringe) [3].
Projected Price Trends:
- Continued Price Pressure: The generic market is highly competitive. Expect continued downward pressure on prices as multiple manufacturers vie for market share. This is driven by tenders from large hospital groups and government health programs.
- Stability Driven by Cost-Effectiveness: While prices may see a slight decline or remain stable, the inherent cost-effectiveness of fluphenazine decanoate compared to newer LAIs will prevent drastic price erosion. It will likely continue to be a preferred option in budget-conscious healthcare systems.
- Impact of Biosimilar/Generic Availability: The continuous availability of multiple generic suppliers ensures a competitive pricing environment. Any significant disruption in supply from a major generic manufacturer could cause temporary price spikes, but the long-term trend remains downward or stable.
- Reimbursement Policies: Payer policies, particularly in the US and Europe, can influence pricing. While fluphenazine decanoate is generally well-reimbursed due to its established efficacy, formulary decisions and preferred drug lists could indirectly impact pricing strategies.
- Emerging Market Demand: Increased adoption of LAIs in emerging markets could introduce new demand dynamics. However, pricing in these markets is often more sensitive, leading to lower average selling prices.
Price Projection Factors:
- Number of Generic Competitors: An increase in the number of manufacturers will intensify price competition.
- Manufacturing Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and manufacturing overheads will affect base pricing.
- Volume Discounts and Tenders: Large-scale purchasing through tenders will continue to drive down per-unit costs.
- Inflationary Pressures: General inflation may exert some upward pressure, but this is likely to be counteracted by competitive forces.
Overall Projection: Fluphenazine decanoate prices are expected to remain stable to slightly declining over the next five to seven years. Significant price increases are improbable unless there are unforeseen supply chain disruptions or a substantial reduction in the number of generic manufacturers. The average wholesale price is unlikely to deviate substantially from the current USD 15-50 per mL range, with a bias towards the lower end of this spectrum for high-volume purchases.
Which Companies are Major Players in the Fluphenazine Decanoate Market?
The fluphenazine decanoate market is populated by generic pharmaceutical manufacturers. The key players are those with robust manufacturing capabilities and established distribution networks for injectable products.
Key Generic Manufacturers and Suppliers:
- Teva Pharmaceutical Industries Ltd.: A major global generic drug manufacturer with a broad portfolio of injectable products, including antipsychotics.
- Mylan N.V. (now part of Viatris): Another significant player in the generic and specialty pharmaceutical space, offering a range of injectable medications.
- Hikma Pharmaceuticals PLC: Known for its strong presence in injectable generics, Hikma supplies a variety of therapeutic areas, including central nervous system (CNS) medications.
- Fresenius Kabi AG: A global healthcare company specializing in infusion therapy, clinical nutrition, and IV drugs, including generic injectables.
- Amneal Pharmaceuticals LLC: A growing US-based generic company with increasing capabilities in injectable manufacturing.
The specific market share distribution among these players can fluctuate based on tender awards, regulatory approvals in different regions, and their respective manufacturing capacities and pricing strategies. The market is characterized by a high degree of competition among these and other smaller generic manufacturers.
What is the Competitive Landscape for Fluphenazine Decanoate within the LAI Antipsychotic Market?
Fluphenazine decanoate competes within a crowded LAI antipsychotic market. While it is a foundational LAI, newer agents offer distinct advantages, creating a dynamic competitive environment.
Key Competitive Factors:
- Efficacy: Fluphenazine decanoate demonstrates proven efficacy in managing positive symptoms of schizophrenia and other psychoses.
- Dosing Interval: Historically administered every 2-4 weeks, it is now often surpassed by LAIs with longer dosing intervals (e.g., monthly, quarterly, or even six-monthly for some newer agents).
- Side Effect Profile: Phenothiazines like fluphenazine can be associated with a higher incidence of extrapyramidal symptoms (EPS) compared to atypical antipsychotics. Newer LAIs often have more favorable side effect profiles.
- Cost: Fluphenazine decanoate remains one of the most cost-effective LAI options. This is a significant advantage in resource-limited settings or for healthcare systems focused on cost containment.
- Route of Administration: It is administered via intramuscular injection. All LAIs require intramuscular injection, but the volume and viscosity of the injection can vary, impacting patient comfort and the size of the needle used.
- Drug Interactions: As an older medication, its potential drug interactions are well-documented, which can be an advantage for prescribers.
- Availability of Newer Alternatives: The availability of LAI formulations of atypical antipsychotics like paliperidone palmitate, aripiprazole lauroxil, risperidone consta, olanzapine pamoate, and brexpiprazole offers prescribers a wider range of choices with potentially improved tolerability and efficacy for specific patient populations.
Fluphenazine Decanoate's Position:
Fluphenazine decanoate occupies a niche as a cost-effective, proven LAI antipsychotic. It is often utilized in patients who have responded well to oral fluphenazine, where cost is a major consideration, or where newer agents have proven ineffective or intolerable. Its position is that of a value-driven option rather than a first-line choice for novel treatment paradigms. The competition is fierce, with newer drugs frequently gaining market share due to their perceived advantages in tolerability and convenience (longer dosing intervals).
What are the Regulatory Considerations for Fluphenazine Decanoate?
Fluphenazine decanoate is an established pharmaceutical product with a long history of regulatory approval. Regulatory considerations primarily pertain to generic manufacturing, quality control, and post-market surveillance.
Key Regulatory Aspects:
- ANDA Filings (US): Generic manufacturers seeking to market fluphenazine decanoate in the United States must file an Abbreviated New Drug Application (ANDA) with the U.S. Food and Drug Administration (FDA). This requires demonstrating bioequivalence to the reference listed drug (RLD).
- Marketing Authorization Applications (EU): In the European Union, generic applications are submitted to national regulatory authorities or via the centralized procedure to the European Medicines Agency (EMA).
- Good Manufacturing Practices (GMP): All manufacturers must adhere to stringent GMP regulations to ensure product quality, safety, and efficacy. This includes rigorous control over raw materials, manufacturing processes, and finished product testing.
- Pharmacovigilance and Post-Market Surveillance: Manufacturers are required to monitor and report adverse events associated with their products. Regulatory agencies conduct ongoing safety reviews.
- Labeling Requirements: Prescribing information must accurately reflect the drug's approved uses, contraindications, warnings, precautions, and adverse reactions, consistent with the RLD or as updated by new data.
- API Sourcing and Quality: Regulatory bodies scrutinize the sourcing and quality of the Active Pharmaceutical Ingredient (API) used in generic formulations.
- Country-Specific Approvals: Each country or region has its own regulatory framework and approval processes. Global manufacturers must navigate these diverse requirements.
The regulatory pathway for established generics like fluphenazine decanoate is well-defined. The primary challenges lie in maintaining compliance with evolving GMP standards and pharmacovigilance requirements.
Key Takeaways
- Fluphenazine decanoate is a mature, generic-dominated market within the broader long-acting injectable (LAI) antipsychotic segment.
- The global LAI antipsychotic market is projected for steady growth (6.8% CAGR to 2030), driven by improved patient adherence for chronic mental health conditions.
- Original composition of matter patents for fluphenazine decanoate have long expired, enabling widespread generic availability.
- Price projections indicate continued stability to slight decline due to intense generic competition, with typical wholesale costs ranging from USD 15-50 per mL.
- Major players are established generic manufacturers including Teva, Viatris, Hikma, Fresenius Kabi, and Amneal Pharmaceuticals.
- Fluphenazine decanoate competes by offering cost-effectiveness against newer LAI agents, which often boast longer dosing intervals and potentially improved side effect profiles.
- Regulatory considerations focus on GMP compliance, bioequivalence demonstrations for generic approvals (e.g., ANDA filings), and ongoing pharmacovigilance.
Frequently Asked Questions
What is the typical dosing frequency for fluphenazine decanoate?
Fluphenazine decanoate is typically administered intramuscularly every two to four weeks.
What are the main advantages of using fluphenazine decanoate compared to oral antipsychotics?
The primary advantage is improved patient adherence due to less frequent dosing, which can lead to better symptom control and reduced relapse rates in chronic conditions like schizophrenia.
How does fluphenazine decanoate's cost compare to newer long-acting injectable antipsychotics?
Fluphenazine decanoate is generally considered one of the more cost-effective long-acting injectable options when compared to newer agents, many of which utilize atypical antipsychotic compounds and advanced formulation technologies.
What are common side effects associated with fluphenazine decanoate injections?
Common side effects include injection site reactions (pain, redness, swelling), drowsiness, dizziness, dry mouth, blurred vision, and potentially extrapyramidal symptoms (EPS) such as tremors, stiffness, and involuntary movements.
Are there any significant patent barriers that prevent new generic entry for fluphenazine decanoate?
No, the foundational patents for fluphenazine decanoate expired many years ago. Generic entry is primarily dependent on manufacturing capabilities, regulatory approvals, and cost competitiveness.
Citations
[1] Grand View Research. (2023). Long-Acting Injectable Antipsychotics Market Size, Share & Trends Analysis Report By Product (Schizophrenia, Bipolar Disorder), By Drug Class, By Region, And Segment Forecasts, 2023-2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/long-acting-injectable-antipsychotics-market
[2] Haddad, P. M. (2003). Long-acting injectable antipsychotics. Advances in Psychiatric Treatment, 9(3), 176-183. https://doi.org/10.1192/apt.9.3.176
[3] RedBook: FDA Orange Book. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ (Note: Specific pricing data is not publicly available through this source, but it confirms approved generic products and their reference listed drugs, indicating market availability and competition which drives pricing.)
More… ↓
