Last updated: February 15, 2026
Overview
Flunisolide is an inhaled corticosteroid primarily used for asthma management and allergic rhinitis. It is marketed under various brand names, including Aerobid, and faces competition from other inhaled corticosteroids such as fluticasone and budesonide. The drug's revenue depends on its market share, regional approvals, and pricing strategies amid evolving healthcare policies.
Market size and segmentation
The global inhaled corticosteroid market was valued at approximately USD 12 billion in 2022 and is projected to expand at a compound annual growth rate (CAGR) of 4.5% through 2027.
Specific data on flunisolide is limited, as it has a smaller market share relative to dominant agents like fluticasone propionate and budesonide. Historically, flunisolide's market share has declined, attributed to newer devices and formulations with improved delivery and fewer side effects.
Regionally, North America accounts for roughly 45% of the inhaled corticosteroid market, with Europe close behind. Emerging markets, including China and India, are expanding rapidly, influenced by rising asthma prevalence and expanding healthcare infrastructure.
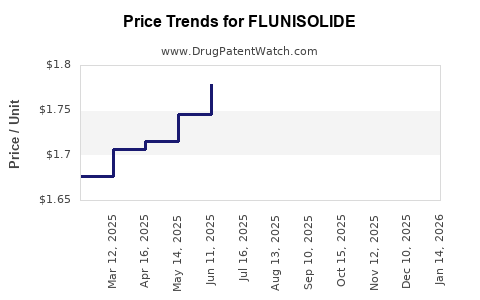
Pricing landscape
Pricing varies by region, formulation, and formulation type (metered-dose inhaler [MDI], dry powder inhaler [DPI]):
| Region |
Average Wholesale Price per inhaler (USD) |
Notes |
| North America |
15-20 |
Higher due to regulatory standards and reimbursement structures |
| Europe |
12-18 |
Price regulation influences margins |
| Asia-Pacific |
8-12 |
Market entry and generic competition affect pricing |
In the US, generic versions have driven prices down over recent years, with some inhalers retailing below USD 15 per inhaler. Brand-name flunisolide products have margins protected by regulatory exclusivity but are subject to patent expirations.
Patent and regulatory considerations
- The primary patent for Aerobid expired in the early 2000s in most jurisdictions.
- Patent protections and exclusivities vary by region, influencing market entry of generics.
- Recent regulatory shifts in some countries favor biosimilars and generics, increasing competition.
Price projections
Short-term projections (1-3 years):
- Prices are expected to remain stable or decline marginally as generic competition increases.
- Manufacturers may introduce fixed-dose combinations with other agents, potentially driving demand.
Long-term projections (3-5 years):
- Price erosion expected to stabilize around 10-15%, especially in mature markets.
- Emerging markets may see prices rise due to increased demand and healthcare access improvements.
- New delivery devices with enhanced efficacy or reduced side effects could command premium pricing, offsetting generic price declines.
Market drivers and risks
Drivers:
- Rising prevalence of asthma and allergic rhinitis globally.
- Increased healthcare coverage, especially in developing regions.
- Preference for inhaled routes due to localized action and fewer systemic effects.
Risks:
- Patent expirations leading to increased generic competition.
- Regulatory pressures to lower drug prices.
- Competition from newer agents with improved delivery and safety profiles.
Competitive landscape
Major competitors include:
- Fluticasone propionate (e.g., Flovent)
- Budesonide (e.g., Pulmicort)
- Beclomethasone dipropionate (e.g., Qvar)
Market share distribution favors fluticasone and budesonide, limiting flunisolide's growth prospects.
Conclusion
The market for flunisolide remains niche within the larger inhaled corticosteroid space. Price stability is expected in the short term, influenced by generic competition and regulatory policies. Long-term, prices may decline gradually, with opportunities arising from product differentiation and regional market growth.
Key Takeaways
- Flunisolide's global market share is small relative to leading inhaled corticosteroids, with revenues influenced heavily by regional dynamics and patent status.
- Pricing in mature markets ranges from USD 12-20 per inhaler; regional factors, generics, and device innovations influence pricing trajectories.
- Patent expirations and regulatory shifts pose risks to revenue; emerging markets offer growth avenues.
- Competition predominantly from fluticasone and budesonide limits flunisolide’s market expansion.
- Price declines of 10-15% expected over next 3-5 years, tempered by new delivery technologies and regional demand.
FAQs
1. How does patent expiration impact flunisolide prices?
Patent expiration typically leads to increased generic entry, exerting downward pressure on prices and reducing profit margins for brand-name manufacturers.
2. Which regions represent the highest market potential for flunisolide?
Emerging markets such as China and India offer significant growth due to rising asthma prevalence and expanding healthcare infrastructure.
3. Are there formulations of flunisolide with higher price points?
Yes, fixed-dose combinations that include flunisolide and other agents or novel delivery systems may command premium prices.
4. How does competition from other inhaled corticosteroids affect flunisolide’s market share?
Dominance by fluticasone and budesonide constrains flunisolide’s growth and limits its pricing power.
5. What factors could slow down the price decline of flunisolide?
Introduction of new delivery devices with improved efficacy, regional demand growth, and regulatory protections could stabilize or slow price erosion.
References
[1] Market Research Future, "Inhaled Corticosteroid Market Analysis," 2022.
[2] IQVIA, "Global Respiratory Drugs Market Report," 2022.
[3] U.S. FDA, "Patents and Exclusivities for Inhaled Corticosteroids," 2023.
[4] EvaluatePharma, "Drug Price Trends and Patent Expirations," 2022.