Share This Page
Drug Price Trends for FETZIMA ER
✉ Email this page to a colleague
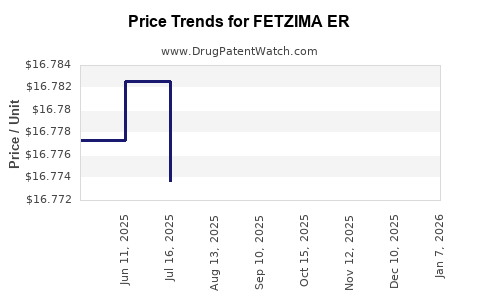
Average Pharmacy Cost for FETZIMA ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FETZIMA ER 120 MG CAPSULE | 00456-2212-30 | 17.58684 | EACH | 2026-01-02 |
| FETZIMA ER 40 MG CAPSULE | 00456-2240-30 | 17.63040 | EACH | 2026-01-02 |
| FETZIMA ER 20 MG CAPSULE | 00456-2220-30 | 17.63169 | EACH | 2026-01-02 |
| FETZIMA ER 80 MG CAPSULE | 00456-2280-30 | 17.64707 | EACH | 2026-01-02 |
| FETZIMA ER 120 MG CAPSULE | 00456-2212-30 | 16.74937 | EACH | 2025-12-17 |
| FETZIMA ER 20 MG CAPSULE | 00456-2220-30 | 16.79208 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FETZIMA ER
What is FETZIMA ER?
FETZIMA ER (levomilnacipran extended-release) is an SNRI approved for the treatment of major depressive disorder (MDD) in adults. Approved by the FDA in July 2013, it offers a differentiated profile within the antidepressant segment due to its dosing and mechanism of action targeting both serotonin and norepinephrine reuptake.
Market Overview
Market size and growth trends
The global antidepressant market was valued at approximately USD 15 billion in 2021 and is expected to grow at a compound annual growth rate (CAGR) of 3.5% through 2028 (Grand View Research, 2022). Major drivers include increased awareness of mental health, rising depression prevalence, and expanding access to healthcare. FETZIMA ER specifically targets a niche within this market, with its unique formulation and dosing profile.
Competitor landscape
Key competitors include:
| Drug | Class | Approvals | Peak sales (2021) | Unique features |
|---|---|---|---|---|
| Effexor XR | SNRI | 1993 | USD 2.2 billion | Non-selective SNRI, multiple indications |
| Sertraline | SSRI | 1991 | USD 3.3 billion | Leading SSRI, broad indication coverage |
| Duloxetine | SNRI | 2004 | USD 1.2 billion | Also approved for neuropathic pain |
| Vilazodone | SSRI/5-HT1Apartial agonist | 2011 | Data limited | 1st in its class |
| LEVOMILNACIPRAN | SNRI | 2013 | N/A | Specific to MDD, alternative dosage |
Market positioning of FETZIMA ER
FETZIMA ER's approval in 2013 positioned it as a treatment with potentially fewer side effects than older SNRI formulations. Its extended-release profile supports once-daily dosing, which appeals to adherence factors.
Market penetration and adoption
Since launch, FETZIMA ER has captured less than 2% of the antidepressant market share. Adoption varies across regions due to prescriber familiarity, insurance coverage, and formulary placement.
Price Analysis
Current pricing data
- Average wholesale price (AWP): Approximately USD 430 per month for a typical dosing regimen (20 mg or 40 mg daily).
- Estimated out-of-pocket costs: USD 20–USD 50 per month, depending on insurance and discounts.
Price comparison
| Drug | Monthly Wholesale Price | Indications | Dosing Frequency |
|---|---|---|---|
| FETZIMA ER | USD 430 | MDD | Once daily |
| Effexor XR | USD 200–USD 400 | Anxiety, depression | Once daily |
| Sertraline | USD 15–USD 50 | Depression, OCD, PTSD | Once daily |
| Duloxetine | USD 200–USD 300 | Depression, neuropathy | Once daily |
FETZIMA ER's price aligns with other newer SNRI drugs but exceeds the cost of generics like sertraline, affecting market uptake.
Pricing outlook
- Short-term (1–2 years): Expect minor fluctuations influenced by payer negotiations and volume discounts. Target list price likely remains near USD 430.
- Mid-term (3–5 years): Price erosion may occur due to increased generic competition for SNRIs, especially if patents expire or biosimilar options emerge.
- Long-term (5+ years): Potential price decrease of 20–30% driven by generics and market penetration strategies.
Regulatory and Patent Considerations
- Patent protection: U.S. patent expiring in 2027, with potential for extensions.
- Orphan drug status: Not applicable.
- Biosimilar and generic threats: Generics are unlikely due to chemical complexity but could emerge through biosimilar pathways or reformulations.
Market Drivers and Barriers
Drivers
- Rising prevalence of depression.
- Increasing recognition of the importance of adherence.
- Physician interest in personalized treatment profiles.
Barriers
- Dominance of established drugs like sertraline and venlafaxine.
- High cost relative to generics.
- Limited physician familiarity compared to older medications.
Sales and Revenue Projections
Assuming limited market share growth:
| Year | Estimated US Sales (USD millions) | Assumptions |
|---|---|---|
| 2023 | USD 30–50 | Stable market penetration, price kept steady |
| 2025 | USD 50–75 | Slight growth as awareness increases |
| 2028 | USD 75–100 | Market penetration reaches ~5–7% of SNRI sales |
Europe and other markets are expected to contribute an additional USD 10–20 million by 2028, contingent on regulatory approval and market access.
Key Takeaways
- FETZIMA ER entered a competitive landscape dominated by older, more affordable SNRIs.
- Current pricing reflects differentiation but limits adoption.
- Patent protection extends into the late 2020s, constraining generic competition.
- Growth relies on prescriber education, formulary positioning, and expanding indications.
- Price erosion is inevitable as generic options increase.
FAQs
-
What are the main competitive advantages of FETZIMA ER?
Its once-daily extended-release formulation and targeted mechanism can improve adherence and offer a side effect profile distinct from older SNRIs. -
How does FETZIMA ER's price compare to other antidepressants?
It costs significantly more than generic SSRIs like sertraline but is comparable to newer SNRI agents. -
What factors could accelerate FETZIMA ER's market growth?
Increased prescribing in treatment-resistant depression, expanded indications, and formulary inclusion. -
When might patents expire, leading to generic competition?
Patent expiration is projected around 2027, potentially opening the market to generics thereafter. -
What is the outlook for FETZIMA ER in international markets?
Limited data suggest gradual expansion, dependent on regulatory approval and local market dynamics.
References
[1] Grand View Research. (2022). Antidepressant drugs market size, share & trends analysis report.
[2] U.S. Food and Drug Administration. (2013). FETZIMA (levomilnacipran) extended-release capsules approval letter.
[3] IQVIA. (2022). Medicinal market insights report.
More… ↓
