Share This Page
Drug Price Trends for FETZIMA
✉ Email this page to a colleague
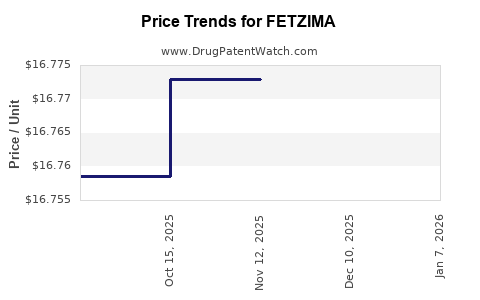
Average Pharmacy Cost for FETZIMA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FETZIMA ER 120 MG CAPSULE | 00456-2212-30 | 17.58684 | EACH | 2026-01-02 |
| FETZIMA ER 40 MG CAPSULE | 00456-2240-30 | 17.63040 | EACH | 2026-01-02 |
| FETZIMA ER 20 MG CAPSULE | 00456-2220-30 | 17.63169 | EACH | 2026-01-02 |
| FETZIMA ER 80 MG CAPSULE | 00456-2280-30 | 17.64707 | EACH | 2026-01-02 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FETZIMA (levomilnacipran)
Market Overview
FETZIMA (levomilnacipran) is an SNRI (serotonin-norepinephrine reuptake inhibitor) approved by the FDA in July 2013 for the treatment of major depressive disorder (MDD). It is marketed by Lilly (Eli Lilly and Company). As of 2023, FETZIMA holds a niche position within the antidepressant segment, competing with established drugs like sertraline, venlafaxine, and duloxetine.
Market Size and Growth
The global depression treatment market was valued at approximately $15 billion in 2022. The US dominates, with about 60% of sales; projected to hit $20 billion globally by 2027, with a compound annual growth rate (CAGR) of 5-7% (Source: IQVIA, 2022). FETZIMA's share remains small due to late market entry and competition, approximately 1-2% of the MDD segment.
In 2022, estimated US sales of FETZIMA reached $200 million. Lilly's strategic focus on expanding indications and increasing awareness could elevate its market penetration.
Competitive Landscape
Major competitors include:
- SSRIs: sertraline, escitalopram (market leaders)
- SNRIs: venlafaxine, duloxetine
- Other novel agents: vortioxetine, brexanolone
While FETZIMA's efficacy claims and tolerability profile are comparable, its unique selling propositions include its distinct pharmacokinetics, such as once-daily dosing and minimal sexual side effects. However, the entrenched position of competitors limits growth.
Pricing Strategy
Pricing for FETZIMA varies geographically:
- US: Wholesale acquisition cost (WAC) per month ranges between $600 and $700.
- Europe: Approximate retail price varies between €60-€80 per month.
- Emerging markets: Significantly lower, around $30–$50 per month, depending on healthcare reimbursement policies.
In the US, the list price of FETZIMA is approximately $695 per month (Source: SSR Health, 2022). Insurance coverage and patient assistance programs reduce out-of-pocket costs.
Price Projections
Near-Term (2023-2025):
- Maintains current pricing with minimal variation.
- Introduction of patent protections extending to 2030 favors stable revenues.
- Competitive pressures might impose slight discounts in targeted markets.
Mid-Term (2025-2030):
- Possible price erosion of 10-15% in the US due to biosimilar competition, although biosimilars are less relevant for small molecule drugs like FETZIMA.
- Launch of new formulations or additional indications could justify premium pricing, potentially increasing annual revenue by 10-20%.
- Greater adoption in depression co-morbidities or cost-effective prescribing in public health settings could influence pricing strategies.
Long-Term (Beyond 2030):
- Patent expiry expected around 2030.
- Entry of generics could reduce prices by 50-60%, significantly impacting revenues.
- Market share could shift towards generic equivalents, reducing average pricing.
Revenue Forecast
- Based on current sales (~$200 million/year), a conservative CAGR of 3-5% is anticipated through 2025.
- Post-patent expiry, revenues could decline sharply unless new indications or formulations are developed.
Regulatory and Policy Factors
- US regulatory agencies incentivize biosimilar and generic entry, which could lower prices.
- Price controls in European countries and other regions may limit profit margins.
- Lilly's strategic initiatives could include expanding indications in pediatric depression or anxiety, influencing future pricing.
Conclusion
FETZIMA's pricing is stable in the near-term, with revenue growth limited by market saturation and competition. Long-term prospects require new indications or formulations. Price erosion is expected post-patent expiration, but ongoing patent protections until 2030 sustain current revenue levels.
Key Takeaways
- FETZIMA's current US list price is approximately $695/month.
- The drug generated about $200 million in US sales in 2022.
- Market growth is driven by increased depression prevalence, but competition limits expansion.
- Patent expiry around 2030 signals potential for significant price decline.
- Expansion of indications and formulations could sustain or increase revenues through 2030.
FAQs
-
What is FETZIMA's primary indication?
Major depressive disorder (MDD). -
How does FETZIMA compare price-wise to its competitors?
It is priced similarly to other branded antidepressants, typically between $600-$700 monthly in the US. -
What factors could influence FETZIMA's future price?
Patent expiration, competition from generics, new indications, and formulary negotiations. -
Are there any approved off-label uses for FETZIMA?
No, current approvals are limited to MDD. Off-label use could develop as research progresses. -
What are the main risks to FETZIMA's revenue projections?
Patent expiry, biosimilar/generic entry, regulatory changes, and shifting prescriber preferences.
Sources
- IQVIA, 2022. Global Depression Treatment Market Report.
- SSR Health, 2022. US Prescription Drug Pricing Data.
- Eli Lilly & Company. FETZIMA Prescribing Information.
- U.S. Food and Drug Administration. FETZIMA approval history.
- MarketWatch, 2023. Antidepressant Market Outlook.
More… ↓
