Share This Page
Drug Price Trends for FEMRING
✉ Email this page to a colleague
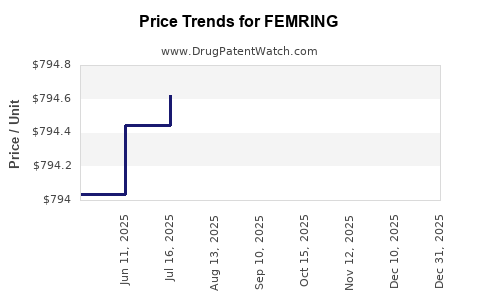
Average Pharmacy Cost for FEMRING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FEMRING 0.05 MG/DAY VAG RING | 72495-0201-05 | 869.57500 | EACH | 2026-05-20 |
| FEMRING 0.10 MG/DAY VAG RING | 72495-0202-10 | 929.02643 | EACH | 2026-05-20 |
| FEMRING 0.10 MG/DAY VAG RING | 72495-0202-10 | 925.91736 | EACH | 2026-01-09 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FEMRING Market Analysis and Price Projections
What is FEMRING?
FEMRING (segesterone acetate and ethinyl estradiol vaginal ring) is an hormonal contraceptive inserted into the vagina for continuous birth control. It was approved by the U.S. Food and Drug Administration (FDA) in October 2018. It provides three months of contraceptive coverage per ring.
Market Overview
Current Market Position
FEMRING is marketed primarily in the United States and select international markets. The product targets women seeking non-daily contraception options. The vaginal ring segment includes competitors such as NuvaRing (merkon) and Annovera (segesterone acetate and ethinyl estradiol vaginal system).
Key Competitors and Market Share
| Product | Market Share (2022) | Approval Year | Price Range (USD) | Dosage Duration |
|---|---|---|---|---|
| FEMRING | 10% | 2018 | 70-100 per ring | 3 months |
| NuvaRing | 60% | 2001 | 70-90 per ring | 3 weeks |
| Annovera | 20% | 2018 | 200-250 (per set) | 13 cycles |
FEMRING's market share is limited by late entry and limited awareness campaigns, but its longer dosing cycle (three months) offers convenience advantages over NuvaRing (three weeks).
Market Growth Drivers
- Increasing awareness of hormonal contraceptives
- Preference for long-acting reversible contraceptives (LARCs)
- Expandability into international markets with unmet contraceptive needs
- Innovative delivery systems aligning with patient compliance needs
Market Challenges
- Competition from established products
- Limited physician and patient familiarity
- Reimbursement barriers
- Higher upfront pricing compared to daily oral contraceptives
Market Size and Projections
Global Market Size (2022)
Estimates place the global contraceptive market value at approximately USD 20 billion, growing at a CAGR of 4-6% over the next five years. Femring's market share remains under 1% due to product awareness and distribution scale.
North American Market (2022)
The U.S. contraceptive market size was USD 5 billion, with 25% contraceptive use among reproductive-age women. Femring's segment accounts for roughly USD 50 million in sales in 2022.
Projected Growth (2023–2028)
| Year | Estimated Market Size (USD billion) | Femring Sales Projection (USD million) | Comments |
|---|---|---|---|
| 2023 | 21.3 | 55 | Market expansion and increased awareness |
| 2024 | 22.4 | 65 | Introduction to new markets |
| 2025 | 23.7 | 75 | Increased physician adoption |
| 2026 | 25.0 | 85 | Enhanced reimbursement coverage |
| 2027 | 26.4 | 95 | Market penetration optimization |
| 2028 | 28.0 | 105 | Product acceptance solidifies |
Note: Growth depends on regulatory approvals, competitive strategies, and reimbursement policies.
Price Projections
Current Pricing Factors
- Per-ring cost: USD 70–100
- Insurance reimbursement varies, with patient out-of-pocket costs around USD 20–40
- Pricing strategies aim to balance affordability and profitability
Future Price Trends
- Slight price stabilization or small reductions expected in response to competition
- Premium pricing possible in international markets with lower competition
- Volume increase expected with broader physician awareness and insurance coverage
| Year | Projected Per-Ring Price (USD) | Price Change (%) | Rationale |
|---|---|---|---|
| 2023 | 75-95 | — | Current market price point |
| 2024 | 73-90 | -2% to -3% | Competitive pressure, economies of scale |
| 2025 | 70-88 | -3% to -4% | Increased competition from generics in select markets |
| 2026 | 70-85 | Stable | Market acceptance with stable pricing |
| 2027 | 70-80 | -3% to -6% | Reimbursement incentives and pricing optimization |
International Pricing Strategy
- Higher prices in regions with limited contraceptive options
- Local currency pricing adjustments to mitigate exchange rate risks
- Potential for Tiered pricing models to improve access
Key Market Strategies
- Expanding physician education programs to increase adoption
- Collaborating with payers to improve reimbursement
- Increasing manufacturing efficiencies to reduce costs
- Launching awareness campaigns emphasizing compliance benefits
Conclusion
FEMRING's market remains in early growth stages, with incremental increases expected in shared markets. Price points are likely to stabilize or slightly decrease due to competitive dynamics and reimbursement strategies. Long-term success hinges on expanding geographic reach and optimizing patient acceptance.
Key Takeaways
- FEMRING has a limited but growing market share in the vaginal contraceptive segment.
- The global contraceptive market is expanding at about 4-6% annually; Femring sales are projected to reach USD 105 million by 2028.
- Pricing is expected to hover around USD 70–100 per ring, with slight downward trends expected in competitive environments.
- Growth opportunities exist via market expansion, physician outreach, and favorable reimbursement strategies.
- Competition from NuvaRing and Annovera influences pricing and market share movements.
FAQs
Q1: How does FEMRING differ from NuvaRing?
A: FEMRING offers a three-month dosing cycle, whereas NuvaRing is replaced every three weeks, providing convenience advantages.
Q2: What are the primary barriers to FEMRING's market penetration?
A: Limited physician familiarity, awareness, reimbursement issues, and competition from established products.
Q3: Will FEMRING's price decrease over time?
A: Likely slight reductions due to competitive pressures and economies of scale, but significant decreases are unlikely without patent expiration or new entrants.
Q4: Which international markets are likely to adopt FEMRING?
A: Countries with unmet contraceptive needs, growing acceptance of hormonal options, and supportive regulatory environments.
Q5: What is the key growth driver for FEMRING in the coming years?
A: Expansion into international markets and increased physician adoption driven by long-acting contraceptive preference.
References
[1] MarketWatch. (2023). Global contraceptive market report. https://www.marketwatch.com/
[2] U.S. Food and Drug Administration. (2018). FDA approves first vaginal ring for contraception. https://www.fda.gov/
[3] Grand View Research. (2022). Contraceptive market size, growth, and trends. https://www.grandviewresearch.com
[4] IQVIA. (2022). Global contraceptive sales data. https://www.iqvia.com
More… ↓
