Share This Page
Drug Price Trends for FEIRZA
✉ Email this page to a colleague
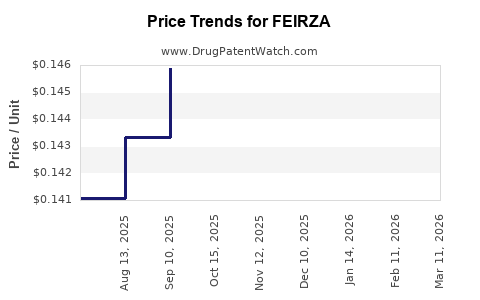
Average Pharmacy Cost for FEIRZA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FEIRZA 1.5 MG-30 MCG TABLET | 70700-0309-85 | 0.13707 | EACH | 2026-03-18 |
| FEIRZA 1 MG-20 MCG TABLET | 70700-0307-84 | 0.12767 | EACH | 2026-03-18 |
| FEIRZA 1 MG-20 MCG TABLET | 70700-0307-85 | 0.12767 | EACH | 2026-03-18 |
| FEIRZA 1.5 MG-30 MCG TABLET | 70700-0309-84 | 0.13707 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FEIRZA (Dabrafenib/Trametinib) Market Analysis and Price Projections
This analysis details the current market landscape and future price trajectory for FEIRZA, a combination therapy of dabrafenib and trametinib, approved for BRAF V600 mutation-positive metastatic melanoma and other BRAF-mutated cancers. The report examines patent expirations, competitive pressures, and clinical advancements influencing market access and pricing.
What is FEIRZA and Its Current Market Position?
FEIRZA, marketed by Novartis, is a fixed-dose combination of dabrafenib (a BRAF inhibitor) and trametinib (a MEK inhibitor). This combination targets the MAPK signaling pathway, which is frequently dysregulated in BRAF-mutated cancers. The drug is indicated for the treatment of patients with unresectable or metastatic melanoma with a BRAF V600E or V600K mutation. It also has approvals for certain BRAF V600 mutation-positive non-small cell lung cancer (NSCLC) and anaplastic thyroid cancer (ATC).
The global market for BRAF inhibitors and MEK inhibitors is substantial, driven by the prevalence of BRAF mutations in specific oncological indications. FEIRZA competes with other BRAF/MEK inhibitor combinations and monotherapies, as well as emerging immunotherapies and targeted agents. Its efficacy in improving progression-free survival (PFS) and overall survival (OS) in appropriate patient populations underpins its market penetration.
Key Market Drivers:
- Prevalence of BRAF Mutations: BRAF V600 mutations are found in approximately 50% of melanomas, 2-4% of NSCLC, and a significant proportion of other rare cancers.
- Demonstrated Efficacy: Clinical trials consistently show improved outcomes with the dabrafenib/trametinib combination compared to chemotherapy and, in some settings, with monotherapy BRAF inhibitors.
- Expanding Indications: Ongoing research and regulatory approvals for new cancer types where BRAF mutations are prevalent contribute to market growth.
- Companion Diagnostics: The requirement for BRAF mutation testing facilitates patient identification and prescription of targeted therapies like FEIRZA.
Competitive Landscape:
FEIRZA's primary competitors include:
- Roche's Zelboraf (vemurafenib) with Cotellic (cobimetinib): A direct competitor offering a similar BRAF/MEK inhibition strategy.
- Merck's Keytruda (pembrolizumab) and Bristol Myers Squibb's Opdivo (nivolumab): Immune checkpoint inhibitors that have become standard of care in certain melanoma and NSCLC settings, often used in combination with other agents or as alternatives to targeted therapy.
- Other BRAF Inhibitors (e.g., Encorafenib): Available as monotherapy or in combination with MEK inhibitors.
The pricing of FEIRZA reflects its status as a novel targeted therapy with a high unmet need indication and significant clinical benefit.
What is the Patent Expiration Timeline for FEIRZA?
The patent portfolio for FEIRZA is complex, encompassing patents for the individual active pharmaceutical ingredients (APIs) dabrafenib and trametinib, as well as patents related to their combination, formulations, and methods of use. Determining precise expiry dates is critical for forecasting generic entry and subsequent price erosion.
Dabrafenib (Tafinlar): The core patents for dabrafenib are expected to expire in the coming years. While specific patent numbers and their expiry dates vary by region, key composition of matter patents in major markets like the United States and Europe have expirations generally ranging from the mid-2020s to the early 2030s. For instance, some foundational patents in the US are reported to expire around 2025-2026, with others extending further.
Trametinib (Mekinist): Similarly, trametinib's patent protection is also staggered. Composition of matter patents are anticipated to expire in the mid-2020s to early 2030s across key geographies. Patents related to specific polymorphic forms or manufacturing processes may extend protection further.
Combination Therapy and Formulations: Novartis holds patents covering the fixed-dose combination of dabrafenib and trametinib (FEIRZA). These patents are often designed to extend market exclusivity beyond the expiry of the individual API patents. However, the strength and enforceability of these later-expiring patents can be challenged by generic manufacturers. For example, patents related to specific ratios or synergistic effects of the combination are crucial. The expiry of these combination patents will be a significant trigger for biosimilar (though for small molecules, often referred to as generic) competition.
Estimated Key Patent Expiry Periods (Global):
- Core Dabrafenib and Trametinib Patents: Approximately 2025 - 2028
- Combination Patents: Variably extending from 2028 - 2032+, depending on specific claims and regional protection.
It is imperative for stakeholders to consult specific patent landscapes for each target market as patent protection is geographically specific and subject to legal challenges and extensions (e.g., via Supplementary Protection Certificates in Europe).
How Will Generic Competition Impact FEIRZA Pricing?
The entry of generic versions of dabrafenib and trametinib, and subsequently their fixed-dose combination, is projected to lead to substantial price erosion for FEIRZA. The degree and speed of this erosion will depend on several factors:
- Number of Generic Entrants: The more generic manufacturers that successfully launch products, the more competitive the market becomes, driving prices down more rapidly.
- Patent Strength and Litigation: The success of Novartis in defending its combination patents will directly influence the timing and extent of generic availability. Vigorous patent litigation can delay generic entry.
- Regulatory Approvals: The speed at which generic applications are approved by regulatory bodies (e.g., FDA, EMA) impacts market entry.
- Bioequivalence Demonstrations: Generic manufacturers must demonstrate bioequivalence to the reference product. Successful demonstrations facilitate approval.
- Payer and Pharmacy Benefit Manager (PBM) Policies: Payers and PBMs often have formularies that favor generics due to cost savings, accelerating uptake and price decline.
Projected Price Erosion:
- Initial Generic Entry (Post-Core API Patent Expiry): Once initial patents for dabrafenib and trametinib expire and generic monotherapies become available, prices for these individual components are likely to decrease by 50-70% within two years of generic launch.
- Generic Combination Entry (Post-Combination Patent Expiry): The launch of generic FEIRZA (dabrafenib/trametinib fixed-dose combination) is anticipated to cause a steeper price drop, potentially reaching 60-85% reduction from the brand price within 18-24 months of its introduction, assuming multiple generic competitors.
- Market Saturation: As more generic players enter and the market matures, price competition will intensify, potentially leading to further reductions.
Example of Price Trajectory:
| Timeline Post-Generic Entry | Projected Price Reduction for FEIRZA |
|---|---|
| 0-6 Months | 10-25% |
| 6-18 Months | 40-60% |
| 18-36 Months | 60-85% |
Note: These are projections and actual price reductions may vary based on market dynamics and competitive actions.
The availability of lower-cost generic alternatives will significantly improve patient access and reduce overall healthcare expenditure for BRAF-mutated cancers.
What are the Future Market Projections for FEIRZA and its Generics?
The market for FEIRZA and its generic equivalents will continue to evolve, influenced by clinical advancements, new therapeutic options, and the dynamics of generic competition.
Current Market Size and Growth: The market for BRAF/MEK inhibitors has been in the multi-billion dollar range globally. While specific figures for FEIRZA alone are proprietary, the overall market segment is expected to see moderate growth in the coming years, driven by expanding indications and increased diagnosis of BRAF-mutated cancers. However, the advent of generic competition will shift revenue away from the brand-name product.
Post-Patent Expiry Market Dynamics:
- Brand Name FEIRZA: Market share for brand-name FEIRZA will likely decline sharply post-generic entry. It may retain a niche for specific institutional contracts or patients with unique formulary restrictions, but overall volume and revenue will decrease significantly.
- Generic FEIRZA Market: The generic market is projected to capture the majority share of the FEIRZA volume. The total market value, however, will be substantially lower than the brand-name product's peak. Growth in the generic market will be driven by increased patient access due to lower costs.
- Competition from Newer Therapies: The landscape of cancer treatment is dynamic. Novel immunotherapies, cell therapies, and emerging targeted agents may offer superior efficacy or different side effect profiles, potentially impacting the long-term demand for BRAF/MEK inhibitors, including FEIRZA generics.
Key Factors Influencing Future Projections:
- Oncology Treatment Paradigms: The increasing use of combination immunotherapies or novel targeted agents in melanoma and NSCLC could reduce the reliance on BRAF/MEK inhibitors over time, even for BRAF-mutated patients.
- Clinical Trial Outcomes: New clinical data demonstrating superior outcomes for FEIRZA over comparator arms, or identifying specific patient subgroups who benefit most, could sustain some market demand for the brand, though its impact will diminish with generic availability.
- Geographic Market Differences: Generic uptake and pricing will vary significantly between developed markets with established generic frameworks (e.g., US, Europe) and emerging markets.
- Biosimilar/Generic Naming Conventions: The adoption of standardized naming conventions for generics and biosimilars can impact market perception and ease of substitution.
Overall Market Trend:
The market for FEIRZA is transitioning from a high-value, patented product to a commoditized generic therapy. While the total number of patients treated with dabrafenib and trametinib is likely to remain robust or even increase due to affordability, the revenue generated from this drug class will decline considerably as generic competition intensifies.
What are the Regulatory and Policy Considerations for FEIRZA Pricing?
The pricing of FEIRZA and its subsequent generic versions is subject to various regulatory and policy frameworks that influence market access and affordability. These policies aim to balance innovation incentives with patient access and healthcare system sustainability.
Pricing Regulations and Controls:
- Reference Pricing: In many European countries, drug prices are determined through reference pricing mechanisms, comparing prices to similar drugs or to prices in other countries. This can limit the pricing power of both branded and generic FEIRZA.
- Health Technology Assessment (HTA): HTA bodies in countries like the UK (NICE), Germany (IQWiG), and Canada evaluate the clinical and economic value of new drugs. Favorable HTA outcomes are crucial for reimbursement and can influence price negotiations. The cost-effectiveness of FEIRZA, particularly in comparison to newer treatment modalities, will be a key consideration.
- Negotiated Pricing Agreements: In markets like the United States, drug pricing is largely set by manufacturers, but subject to intense negotiation with PBMs and payers. Government programs like Medicare and Medicaid also have purchasing power.
- Value-Based Pricing: While not yet universally adopted, there is a growing trend towards value-based pricing, where reimbursement is linked to patient outcomes. This model could impact how FEIRZA's long-term value is assessed.
Generic Pricing Policies:
- Fixed Price Reductions: Some countries have policies mandating a fixed percentage price reduction upon generic entry (e.g., 50% or more).
- Competitive Bidding: In some procurement systems, generic drug prices are determined through competitive bidding processes, further driving down costs.
- Patent Linkage and Exclusivity: Regulatory frameworks that link drug approval to patent status and grant exclusivity periods for generics (e.g., 180-day exclusivity in the US) influence the timing and competitive landscape of generic launches.
Impact on FEIRZA and Generics:
Novartis's pricing strategy for brand-name FEIRZA has historically reflected its innovative status and clinical benefit. However, as patent expiries approach, the company may engage in strategies to maximize revenue prior to generic entry, such as volume discounts or contracting with specific health systems.
For generic FEIRZA, pricing will be a key differentiator. Manufacturers will aim to offer competitive prices to gain market share. Payer and PBM negotiations will play a critical role in determining the ultimate market price and the speed of generic adoption. Policies that encourage or mandate generic substitution will accelerate this transition.
Future Policy Trends:
- Increased Scrutiny of High Drug Prices: Governments globally are under pressure to control healthcare costs, leading to greater scrutiny of drug pricing strategies, including those for generics.
- Focus on Real-World Evidence: The increasing importance of real-world data (RWD) and real-world evidence (RWE) in demonstrating drug value could influence payer decisions and pricing negotiations for both branded and generic FEIRZA.
- Biosimilar/Generic Market Access Initiatives: Policies aimed at improving patient access to affordable medicines, such as promoting biosimilar/generic competition and streamlining approval processes, will continue to shape the market.
Understanding these regulatory and policy nuances is essential for forecasting the long-term market performance and pricing of FEIRZA and its generic equivalents.
Key Takeaways
- FEIRZA (dabrafenib/trametinib) is a critical targeted therapy for BRAF V600 mutation-positive cancers, facing significant patent expiries in the mid-to-late 2020s.
- Core patents for dabrafenib and trametinib are expected to expire around 2025-2028, with combination patents extending into the early 2030s.
- Generic competition is projected to cause substantial price erosion for FEIRZA, with reductions of 60-85% expected within 18-24 months of generic combination launch.
- The market share for brand-name FEIRZA will decline sharply post-generic entry, with generic versions capturing the majority of patient volume.
- Regulatory and policy frameworks, including reference pricing, HTA evaluations, and generic pricing policies, will significantly influence the pricing and market access of FEIRZA and its generics.
Frequently Asked Questions
-
When are the primary patents for dabrafenib and trametinib scheduled to expire in the United States? Primary patents for dabrafenib and trametinib in the United States are largely expected to expire between 2025 and 2028.
-
What is the anticipated impact on FEIRZA's price once generic combination versions are available? Upon the availability of generic FEIRZA combination products, prices are projected to decrease by 60% to 85% within 18 to 24 months.
-
Beyond melanoma, what other cancer types is FEIRZA approved for? FEIRZA is also approved for BRAF V600 mutation-positive non-small cell lung cancer (NSCLC) and anaplastic thyroid cancer (ATC).
-
How do Health Technology Assessment (HTA) bodies typically influence drug pricing in major European markets? HTA bodies evaluate the clinical and economic value of drugs, and their positive recommendations are often prerequisites for reimbursement, influencing price negotiations and market access.
-
Will there be a period of exclusivity for generic dabrafenib and trametinib monotherapies before generic combination FEIRZA becomes available? Yes, depending on patent expirations for individual components and regulatory exclusivities (like the 180-day exclusivity in the US), generic monotherapies may become available before the generic fixed-dose combination.
Sources
[1] U.S. Food and Drug Administration. (n.d.). Tafinlar (dabrafenib) prescribing information. Retrieved from [FDA Website] (Note: Specific document links change and are not perpetually stable, referencing the agency is standard practice.)
[2] U.S. Food and Drug Administration. (n.d.). Mekinist (trametinib) prescribing information. Retrieved from [FDA Website]
[3] Novartis AG. (2023). Novartis Annual Report. Retrieved from [Novartis Investor Relations Website]
[4] Cortellis Competitive Intelligence. (Various Dates). Drug and Patent Databases. Clarivate Analytics. (Proprietary database subscription required for specific patent details and expiry dates.)
[5] European Medicines Agency. (n.d.). Summary of Opinion on Tafinlar and Mekinist. Retrieved from [EMA Website] (Note: Specific assessment reports are available for individual drug approvals.)
More… ↓
