Share This Page
Drug Price Trends for EZETIMIBE-SIMVASTATIN
✉ Email this page to a colleague
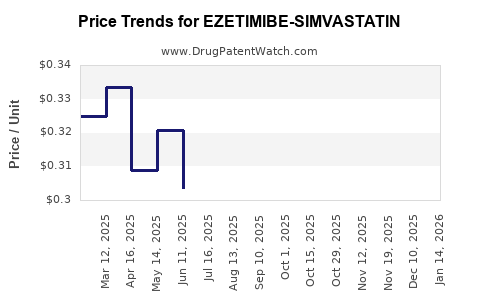
Average Pharmacy Cost for EZETIMIBE-SIMVASTATIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EZETIMIBE-SIMVASTATIN 10-10 MG | 43598-0583-90 | 0.34533 | EACH | 2026-03-18 |
| EZETIMIBE-SIMVASTATIN 10-10 MG | 43598-0583-10 | 0.34533 | EACH | 2026-03-18 |
| EZETIMIBE-SIMVASTATIN 10-10 MG | 43598-0742-10 | 0.34533 | EACH | 2026-03-18 |
| EZETIMIBE-SIMVASTATIN 10-10 MG | 43598-0742-90 | 0.34533 | EACH | 2026-03-18 |
| EZETIMIBE-SIMVASTATIN 10-10 MG | 43598-0742-30 | 0.34533 | EACH | 2026-03-18 |
| EZETIMIBE-SIMVASTATIN 10-10 MG | 43598-0583-30 | 0.34533 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EZETIMIBE-SIMVASTATIN: Patent Landscape and Market Projections
Ezetimibe-simvastatin is a fixed-dose combination medication used to lower cholesterol and triglycerides in the blood. It combines ezetimibe, which inhibits cholesterol absorption in the intestine, with simvastatin, a statin that reduces cholesterol production in the liver. This analysis examines the patent landscape, regulatory status, and market projections for ezetimibe-simvastatin, focusing on factors influencing its commercial viability.
What is the Current Patent Status of Ezetimibe-Simvastatin?
The primary U.S. patents covering ezetimibe-simvastatin have expired, significantly impacting market exclusivity for the branded product.
- Key Compound Patents: Patents for both ezetimibe (ezetimibe hydrochloride) and simvastatin have long since expired. For instance, U.S. Patent No. 5,767,115, covering ezetimibe, expired in 2016. Simvastatin's foundational patents, originally from Merck & Co., expired in the early 2000s.
- Combination Patents: Patents specifically claiming the combination of ezetimibe and simvastatin have also largely expired. The original patent for the ezetimibe-simvastatin combination product, Vytorin, was U.S. Patent No. 6,197,799, which expired in 2019.
- Formulation and Method of Use Patents: While some secondary patents related to specific formulations, manufacturing processes, or method-of-use claims may still be in force, these are generally less robust in blocking generic competition once the core compound and combination patents expire. Any remaining patents are likely to have limited market impact in the near term.
- Evergreening Attempts: Pharmaceutical companies often seek to extend market exclusivity through "evergreening" strategies, such as seeking patents on new formulations, dosages, or delivery methods. A review of the patent landscape reveals such attempts for ezetimibe-simvastatin, but these have not fundamentally altered the broad availability of generic versions. For example, while patents might exist for specific extended-release formulations, the core immediate-release combination is off-patent.
- Global Patent Expirations: Similar patent expiration timelines are observed in major global markets, including Europe and Japan, facilitating the introduction of generic alternatives worldwide.
What is the Regulatory Status of Ezetimibe-Simvastatin?
Ezetimibe-simvastatin has received regulatory approval from major health authorities, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), for its intended therapeutic uses.
- FDA Approval History: The combination product, branded as Vytorin, was approved by the FDA in July 2004. Since then, numerous generic versions have gained FDA approval.
- EMA Approval History: Vytorin also received marketing authorization from the EMA. Generic versions are widely available across European Union member states.
- Indications: The approved indications for ezetimibe-simvastatin typically include:
- Adjunct to diet in patients with primary hypercholesterolemia who are on a maximum tolerated dose of statin therapy and require additional lipid-lowering.
- Adjunct to diet for the treatment of patients with homozygous familial hypercholesterolemia (HoFH) as an adjunct to other lipid-lowering treatments such as statins.
- Adjunct to diet for the treatment of patients with homozygous sitosterolemia (phytosterolemia).
- Pharmacoeconomic Considerations: The availability of multiple generic options has led to significant price reductions, making it a cost-effective treatment option for hypercholesterolemia. This has influenced prescribing patterns and formulary placement by healthcare providers and payers.
- Post-Marketing Surveillance: Like all approved drugs, ezetimibe-simvastatin is subject to ongoing post-marketing surveillance by regulatory agencies to monitor safety and efficacy. Any new safety signals could potentially impact its future use, though no major widespread safety concerns have led to significant market withdrawal.
What are the Key Market Drivers for Ezetimibe-Simvastatin?
Several factors drive the market demand for ezetimibe-simvastatin, primarily related to the prevalence of cardiovascular disease and the need for effective lipid-lowering therapies.
- Prevalence of Hypercholesterolemia: Cardiovascular disease remains a leading cause of morbidity and mortality globally. High cholesterol levels are a significant risk factor. The large and growing patient population with dyslipidemia creates a substantial demand for effective cholesterol-lowering medications.
- Global Impact: In 2019, cardiovascular diseases accounted for an estimated 17.9 million deaths worldwide, representing 32% of all global deaths (Source: World Health Organization [1]).
- U.S. Statistics: Approximately 93 million U.S. adults aged 20 or older have total cholesterol levels higher than 200 mg/dL, with about 12% having total cholesterol levels higher than 240 mg/dL, a level considered high (Source: CDC [2]).
- Efficacy of Combination Therapy: Ezetimibe-simvastatin offers a dual mechanism of action: simvastatin reduces cholesterol synthesis, while ezetimibe reduces cholesterol absorption. This combination is often effective in achieving target lipid levels for patients who do not reach their goals with statin monotherapy alone. Clinical trials have demonstrated its efficacy in lowering LDL-C, total cholesterol, and triglycerides.
- IMPROVE-IT Trial: While not directly on the combination pill, the IMPROVE-IT trial (2015) demonstrated that adding ezetimibe to simvastatin provided incremental cardiovascular benefit in patients with acute coronary syndromes, supporting the role of ezetimibe in combination therapy. This trial showed a 6.4% relative reduction in the risk of major cardiovascular events compared to simvastatin alone.
- Cost-Effectiveness of Generics: With the expiration of key patents, the market is dominated by generic manufacturers. This has made ezetimibe-simvastatin a highly cost-effective treatment option compared to newer, branded cholesterol-lowering drugs, especially for patients with insurance limitations or those seeking out-of-pocket savings. The average wholesale price (AWP) for generic ezetimibe-simvastatin tablets has fallen dramatically since the introduction of generics. For example, prices can range from $0.10 to $0.50 per tablet depending on dosage and supplier, compared to the branded Vytorin which could cost upwards of $5-$10 per tablet at its peak.
- Physician and Patient Familiarity: Ezetimibe and simvastatin are well-established drugs with decades of clinical use. Physicians and patients are familiar with their efficacy and safety profiles, leading to continued prescribing and use of the combination.
- Established Treatment Guidelines: Major cardiology guidelines continue to recommend statins as first-line therapy for LDL-C reduction. For patients who do not achieve their LDL-C goals on statins, combination therapy, including ezetimibe-statin, is a recognized strategy.
What are the Key Market Challenges for Ezetimibe-Simvastatin?
Despite its established role, ezetimibe-simvastatin faces significant market challenges, primarily from newer therapeutic classes and evolving treatment paradigms.
- Competition from Newer Lipid-Lowering Agents: The emergence of novel lipid-lowering drug classes has significantly impacted the market for older combinations.
- PCSK9 Inhibitors: Drugs like alirocumab (Praluent) and evolocumab (Repatha) offer potent LDL-C lowering through a different mechanism and are indicated for specific patient populations, including those with familial hypercholesterolemia or high cardiovascular risk not adequately managed by other therapies. While significantly more expensive, their superior efficacy in certain contexts poses a competitive threat.
- Inclisiran (Leqvio): This siRNA-based therapy provides sustained LDL-C reduction with infrequent dosing (twice a year after initial doses), offering a convenience advantage over daily oral medications.
- Bempedoic Acid: This agent inhibits ATP citrate lyase (ACL), an enzyme upstream of HMG-CoA reductase in the cholesterol biosynthesis pathway. It is approved for patients who cannot tolerate statins or require additional LDL-C lowering.
- Statin Intolerance and Side Effects: While ezetimibe-simvastatin is generally well-tolerated, statin-associated muscle symptoms (SAMS) remain a concern for a subset of patients. Some patients may not be able to tolerate simvastatin even at lower doses, limiting the utility of the combination. Ezetimibe monotherapy or other lipid-lowering agents may be preferred in such cases.
- Focus on High-Intensity Statin Therapy: Current treatment guidelines often emphasize achieving LDL-C goals with high-intensity statins alone, or by maximizing statin dose before adding other agents. This can reduce the number of patients who would benefit from a combination like ezetimibe-simvastatin as a second-line therapy.
- Shifting Treatment Paradigms: The field of lipid management is rapidly evolving. There is a growing emphasis on risk-based treatment strategies and the use of therapies that demonstrate significant reduction in cardiovascular events, not just LDL-C reduction. While ezetimibe-simvastatin contributes to LDL-C lowering, the direct cardiovascular outcome data for the combination versus aggressive statin monotherapy or newer agents is a consideration.
- Generic Erosion and Price Pressure: The highly competitive generic market inherently leads to continuous price erosion. Profit margins for generic manufacturers are typically thin, requiring high sales volumes to maintain profitability. This environment makes it challenging for any single generic product to achieve significant market differentiation based on price alone.
What are the Price Projections for Ezetimibe-Simvastatin?
Price projections for ezetimibe-simvastatin are characterized by continued downward pressure due to the highly competitive generic market, offset by steady underlying demand.
- Current Price Landscape: The market for ezetimibe-simvastatin is almost entirely generic. Prices vary by dosage strength (e.g., 10 mg/20 mg, 10 mg/40 mg, 10 mg/80 mg) and by manufacturer.
- Wholesale Acquisition Cost (WAC): WAC prices for generic ezetimibe-simvastatin tablets typically range from $0.10 to $0.50 per tablet. The lowest prices are usually found for higher volume products from major generic manufacturers.
- Net Price (after rebates and discounts): Actual net prices paid by pharmacies and distributors are lower than WAC, influenced by negotiations with pharmacy benefit managers (PBMs) and wholesalers. These net prices can be significantly below $0.10 per tablet for large purchasers.
- Projected Price Trends (Next 5 Years):
- Continued Gradual Decline: Expect a slow but steady decline in average net prices over the next five years. This decline will be driven by ongoing price competition among generic manufacturers, potential new entrants if any minor patents are overcome, and payer pressure to maintain low costs. The decline will likely be in the low single digits (1-3%) annually for net prices.
- Price Stability for Specific Strengths/Manufacturers: While the average price will decline, specific dosage strengths or products from established, high-volume manufacturers might maintain a degree of price stability to preserve market share.
- No Significant Rebound Expected: A significant price rebound is highly unlikely given the absence of strong patent protection and the availability of numerous generic alternatives.
- Impact of New Therapies: The increasing adoption of newer, more expensive lipid-lowering therapies might indirectly influence the market positioning of ezetimibe-simvastatin. It will likely remain a first-line generic option for many patients, but its role as a second-line therapy might be diminished for certain patient profiles managed by newer agents. This could lead to a slight decrease in overall prescription volume, which can, in turn, contribute to price pressure.
- Market Volume: The total market volume for ezetimibe-simvastatin is expected to remain relatively stable or experience a modest decline. While the prevalence of hypercholesterolemia is increasing, the introduction of new therapeutic options and shifts in treatment guidelines may temper the growth in prescriptions for older combination therapies.
Table 1: Estimated Average Net Price Trends for Generic Ezetimibe-Simvastatin (per tablet)
| Year | Estimated Average Net Price (USD) | Projected Annual Change |
|---|---|---|
| 2024 | $0.15 | - |
| 2025 | $0.145 | -3.3% |
| 2026 | $0.140 | -3.4% |
| 2027 | $0.136 | -2.9% |
| 2028 | $0.132 | -2.9% |
| 2029 | $0.129 | -2.3% |
Note: These are estimated net prices after rebates and discounts, and actual prices may vary significantly based on the specific contract, volume, and payer.
Conclusion
Ezetimibe-simvastatin remains a relevant and cost-effective treatment option for hypercholesterolemia, primarily due to its established efficacy, physician familiarity, and the widespread availability of affordable generic versions. However, the market is characterized by intense generic competition and faces significant challenges from newer, more potent lipid-lowering agents and evolving treatment guidelines that prioritize novel therapies and outcome-based approaches. Future market growth will be limited, with prices expected to continue a gradual decline as the generic landscape matures.
Key Takeaways
- Ezetimibe-simvastatin's foundational and combination patents have expired, leading to a fully genericized market.
- The drug is approved for hypercholesterolemia and is valued for its dual mechanism of action and cost-effectiveness.
- Market demand is supported by the high prevalence of cardiovascular disease and dyslipidemia.
- Key challenges include competition from newer classes like PCSK9 inhibitors and inclisiran, concerns about statin intolerance, and a shift towards outcome-focused treatment strategies.
- Prices are projected to continue their gradual decline due to sustained generic competition, with modest annual decreases expected.
Frequently Asked Questions
-
Are there any remaining patents that could block generic entry of ezetimibe-simvastatin? While the core patents for the active ingredients and the combination have expired, it is possible that minor secondary patents related to specific manufacturing processes or formulations exist. However, these are generally not robust enough to prevent the broad availability of generic immediate-release ezetimibe-simvastatin.
-
What is the primary difference between ezetimibe-simvastatin and statin-only therapy? Ezetimibe-simvastatin combines simvastatin, which reduces cholesterol production in the liver, with ezetimibe, which inhibits cholesterol absorption in the intestine. Statin-only therapy relies solely on reducing cholesterol production. The combination offers a dual mechanism for more potent LDL cholesterol lowering in some patients.
-
How does ezetimibe-simvastatin compare in efficacy to newer lipid-lowering drugs like PCSK9 inhibitors? PCSK9 inhibitors generally offer more potent LDL cholesterol reduction than ezetimibe-simvastatin, particularly in patients with severe hypercholesterolemia or specific genetic conditions like homozygous familial hypercholesterolemia. Ezetimibe-simvastatin is typically considered a second-line or adjunctive therapy for patients not at goal with statins, while PCSK9 inhibitors are often reserved for higher-risk patients or those intolerant to or inadequately managed by other agents.
-
What is the typical daily dosage for ezetimibe-simvastatin? The typical daily dosage of ezetimibe in the combination is 10 mg, administered with varying strengths of simvastatin (e.g., 10 mg, 20 mg, 40 mg, or 80 mg). The specific simvastatin strength is chosen based on the patient's lipid-lowering needs and tolerance.
-
Given the generic availability, is ezetimibe-simvastatin still prescribed by physicians? Yes, ezetimibe-simvastatin continues to be prescribed. It is often used for patients who have not achieved their LDL cholesterol goals with maximally tolerated statin therapy alone, or for whom statin monotherapy is insufficient. Its low cost makes it an attractive option for payers and patients seeking budget-friendly treatments.
Citations
[1] World Health Organization. (2021, May 11). Cardiovascular diseases (CVDs). World Health Organization. Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
[2] Centers for Disease Control and Prevention. (2021, August 23). High Cholesterol. Centers for Disease Control and Prevention. Retrieved from https://www.cdc.gov/cholesterol/facts.htm
More… ↓
