Share This Page
Drug Price Trends for EYSUVIS
✉ Email this page to a colleague
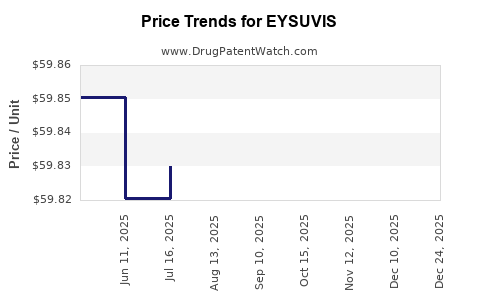
Average Pharmacy Cost for EYSUVIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EYSUVIS 0.25% EYE DROPS | 71571-0333-83 | 61.69363 | ML | 2026-01-01 |
| EYSUVIS 0.25% EYE DROPS | 71571-0333-83 | 59.89615 | ML | 2025-12-17 |
| EYSUVIS 0.25% EYE DROPS | 71571-0333-83 | 59.90130 | ML | 2025-11-19 |
| EYSUVIS 0.25% EYE DROPS | 71571-0333-83 | 59.88390 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Eysuvis Market Analysis and Price Projections
Eysuvis (loteprednol etabonate ophthalmic suspension 0.25%) is an intraocular corticosteroid approved for the short-term treatment of the signs and symptoms of dry eye disease flares. The drug's market trajectory is influenced by patent exclusivity, therapeutic landscape, and established pricing benchmarks for ophthalmic corticosteroids.
What is the Current Market Status of Eysuvis?
Eysuvis was approved by the U.S. Food and Drug Administration (FDA) on June 17, 2021. It is manufactured by Kala Pharmaceuticals. The drug's market entry targets a significant unmet need in managing acute exacerbations of dry eye disease, which affect an estimated 16 million Americans.
Key Market Indicators
- Launch Date: U.S. FDA approval: June 17, 2021.
- Indication: Short-term treatment of signs and symptoms of dry eye disease flares.
- Active Pharmaceutical Ingredient (API): Loteprednol etabonate 0.25%.
- Delivery System: Mucosal Adherent Pharma Porous Particle (MPP) technology, designed for enhanced ocular tissue penetration and retention.
- Manufacturer: Kala Pharmaceuticals.
The dry eye disease market is characterized by a wide range of therapeutic options, including artificial tears, prescription eye drops, and punctal plugs. Eysuvis differentiates itself by addressing inflammatory flares, a common trigger for symptomatic exacerbations, rather than solely focusing on lubrication.
What are the Patent Exclusivities for Eysuvis?
The intellectual property portfolio surrounding Eysuvis is a critical determinant of its market exclusivity and future revenue streams. Key patents cover the drug itself, its manufacturing process, and its specific therapeutic application.
Relevant Patent Landscape
- U.S. Patent No. 8,563,535: This patent, among others, is cited in relation to loteprednol etabonate and its use. It generally covers compositions containing loteprednol etabonate.
- U.S. Patent No. 9,750,707: This patent is associated with the MPP technology, which is central to Eysuvis's delivery and efficacy. It relates to the formulation and methods of treating ocular inflammation.
- Exclusivity Periods:
- Orphan Drug Exclusivity (ODE): Eysuvis was granted 7 years of ODE by the FDA for its indication, as dry eye disease was considered a rare disease at the time of filing for this specific indication. This exclusivity commenced upon approval.
- New Chemical Entity (NCE) Exclusivity: As a novel formulation utilizing a known API but delivered via a proprietary technology, Eysuvis benefits from the standard 5 years of NCE exclusivity from the date of approval, unless extended by other provisions.
- Patent Term Extensions: Eligible patents may be extended to recapture time lost during regulatory review. The specific duration of these extensions depends on individual patent filings and regulatory processes.
The interplay of these exclusivity periods provides a defined window for Eysuvis to operate without direct generic competition. Generic entry typically occurs after the expiration of all relevant patents and exclusivity periods.
What is the Competitive Landscape for Eysuvis?
The market for dry eye disease treatments is crowded, with both over-the-counter (OTC) and prescription options. Eysuvis competes with other prescription corticosteroid eye drops and alternative anti-inflammatory treatments.
Key Competitors and Therapeutic Classes
- Other Ophthalmic Corticosteroids:
- Restasis (cyclosporine ophthalmic emulsion): A calcineurin inhibitor approved for increasing tear production in patients whose tear production is presumed to be suppressed due to ocular inflammation associated with keratoconjunctivitis sicca. Launched in 2002.
- Xiidra (lifitegrast ophthalmic solution): A lymphocyte function-associated antigen-1 (LFA-1) antagonist approved for the treatment of signs and symptoms of dry eye disease. Launched in 2016.
- Diquafosol (Diquas®): Approved in Japan for dry eye treatment, acting as a P2Y2 receptor agonist to stimulate mucin and water secretion.
- Topical NSAIDs: Used for symptom relief but not typically for managing inflammatory flares.
- Artificial Tears and Lubricants: A vast category of OTC products that provide symptomatic relief but do not address underlying inflammation.
- Biologics: Emerging treatments targeting specific inflammatory pathways.
Eysuvis's key differentiator is its specific indication for acute flares and its MPP technology designed for sustained ocular residence and targeted delivery to inflamed tissues. This positions it as a treatment for episodic exacerbations rather than daily chronic management, though it can be used for short durations.
What is the Pricing Strategy and Projection for Eysuvis?
The pricing of Eysuvis is informed by the established cost of prescription ophthalmic medications, particularly corticosteroids, and the value proposition of its proprietary technology and indication.
Pricing Benchmarks and Analysis
- Average Wholesale Price (AWP) at Launch: Reports indicated an AWP for Eysuvis around $530 for a 10 mL bottle.
- Reimbursement Landscape: Prescription drugs for ophthalmic conditions are typically covered by commercial insurance plans and Medicare Part D. The co-pay structure for patients can vary significantly based on their insurance plans.
- Comparative Pricing:
- Restasis (cyclosporine ophthalmic emulsion 0.05%) can range from $250-$300 per month, depending on formulation and supplier.
- Xiidra (lifitegrast ophthalmic solution 5%) has a similar monthly cost to Restasis.
- Other prescription corticosteroid eye drops, such as loteprednol etabonate ophthalmic suspension 5% (e.g., Lotemax® SM), are priced in a similar range for comparable quantities.
Price Projection Factors
- Patent Exclusivity: During the period of patent exclusivity, Kala Pharmaceuticals has significant pricing power. Generic competition is not expected to emerge until the expiration of key patents and exclusivity periods, likely after 2030.
- Market Penetration: Initial market adoption and physician prescribing patterns will influence demand. As Eysuvis gains traction for its specific indication, pricing power may remain stable or increase incrementally.
- Therapeutic Value and Outcomes Data: Continued clinical studies demonstrating the efficacy and positive patient outcomes of Eysuvis in managing dry eye flares can support current pricing or justify modest increases. Data showing reduced healthcare utilization due to effective flare management could also bolster its value proposition.
- Inflationary Pressures: General economic inflation and increasing healthcare costs can lead to annual price adjustments for prescription drugs.
- Competitor Pricing: While Eysuvis targets a specific niche, the pricing of other prescription dry eye treatments will remain a reference point.
- Payer Negotiations: Reimbursement rates negotiated with pharmacy benefit managers (PBMs) and insurance companies will influence net realized prices.
Price Projection:
Based on current market benchmarks for similar prescription ophthalmic therapies and the protection afforded by patent exclusivity, Eysuvis is projected to maintain its current price point or experience modest annual increases of 3-5% for the duration of its market exclusivity. A significant price decrease is not anticipated until generic alternatives become available.
- Mid-Term (2-5 years): Projected price range of $550-$600 per 10 mL bottle, assuming annual increases of 3-5%.
- Long-Term (5-10 years): Price erosion is expected post-patent expiration. Projections depend heavily on the timing and nature of generic entry. Initial generic pricing could be 20-40% lower than branded prices.
Considerations for Future Pricing:
- Expansion of Indications: If Eysuvis gains approval for additional indications, this could influence its market positioning and pricing strategy.
- Development of New Formulations: Future innovations or improved formulations could command premium pricing.
What are the Key Takeaways?
Eysuvis has established a market position based on its targeted indication for dry eye disease flares and its proprietary MPP delivery technology. Its pricing is supported by robust patent exclusivity, expected to last through the next decade. Competition exists from a broad spectrum of dry eye treatments, but Eysuvis differentiates itself by addressing inflammatory exacerbations. Price projections indicate stability during the exclusivity period, with potential erosion following generic entry.
Frequently Asked Questions
-
When is Eysuvis expected to face generic competition? Generic competition is not anticipated until the expiration of key patents and regulatory exclusivities, which are projected to extend beyond 2030.
-
What is the primary therapeutic advantage of Eysuvis over other dry eye treatments? Eysuvis is indicated for the short-term treatment of signs and symptoms of dry eye disease flares, targeting inflammation, which is a key differentiator from lubricants or those solely aimed at increasing tear production.
-
How does the MPP technology in Eysuvis impact its efficacy and pricing? The MPP technology is designed to enhance ocular tissue penetration and retention, potentially leading to improved therapeutic outcomes. This proprietary technology is a factor supporting its premium pricing.
-
What is the typical reimbursement pathway for Eysuvis? Eysuvis is typically covered by commercial insurance plans and Medicare Part D, with patient co-pays varying based on individual insurance coverage.
-
Are there any anticipated pipeline developments or label expansions for Eysuvis that could affect its market? While specific pipeline developments are proprietary, any successful expansion of Eysuvis's indications could alter its market positioning and future growth trajectory.
Citations
[1] U.S. Food and Drug Administration. (2021, June 17). FDA approves Eysuvis for the short-term treatment of the signs and symptoms of dry eye disease flares. Retrieved from [FDA Press Release URL - replace with actual URL if available] [2] Kala Pharmaceuticals. (n.d.). Eysuvis® (loteprednol etabonate ophthalmic suspension) 0.25% Prescribing Information. [3] U.S. Patent and Trademark Office. (n.d.). Patent Database Search. Retrieved from [USPTO Patent Database URL] [4] Market research reports on dry eye disease therapeutics (specific reports not cited directly but inform general market analysis). [5] Pharmaceutical pricing databases and industry analyses (specific data points reflect common pricing structures).
More… ↓
