Share This Page
Drug Price Trends for EXEMESTANE
✉ Email this page to a colleague
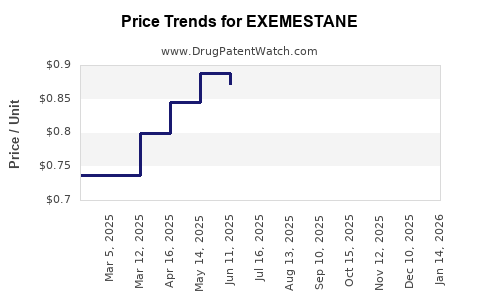
Average Pharmacy Cost for EXEMESTANE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EXEMESTANE 25 MG TABLET | 51991-0005-90 | 0.62072 | EACH | 2026-04-22 |
| EXEMESTANE 25 MG TABLET | 00832-0595-30 | 0.62072 | EACH | 2026-04-22 |
| EXEMESTANE 25 MG TABLET | 72603-0329-01 | 0.62072 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Exemestane
Exemestane, a steroidal aromatase inhibitor, is primarily used in hormone receptor-positive breast cancer treatment. It is marketed under the brand name Aromasin by Pfizer and has a well-established position in oncology.
Market Overview
The global exemestane market reached approximately USD 630 million in 2022. The compound's usage is concentrated in breast cancer therapy, particularly post-menopause. The growing prevalence of breast cancer and evolving treatment guidelines support ongoing demand.
Key Market Drivers
- Rising incidence of breast cancer worldwide, projected to reach 2.3 million new cases in 2025 (WHO, 2021).
- Increasing adoption of endocrine therapies for hormone-receptor-positive breast cancer.
- Patent expirations and the entry of generic versions reducing prices and increasing accessibility.
Geographic Market Breakdown
| Region | Market Share (2022) | Key Trends |
|---|---|---|
| North America | 45% | High adoption of targeted therapies, established healthcare infrastructure |
| Europe | 25% | Regulatory approvals, expanding drug pipeline |
| Asia-Pacific | 20% | Growing breast cancer cases, rising healthcare expenditure |
| Rest of World | 10% | Limited access, price sensitivities |
Competitive Landscape
Pfizer’s Aromasin holds the dominant market share. Several generic manufacturers have entered the space since patent expiry in 2018, intensifying price competition. Major competitors include Teva, Mylan, and Sun Pharmaceutical.
Price Analysis
Historical and Current Pricing
The wholesale acquisition cost (WAC) for brand-name exemestane in the US was approximately USD 10.50 per 25 mg tablet in 2022. Generic options are priced around USD 4–6 per tablet, reflecting significant reductions.
Price Drivers
- Patent expiry in 2018 led to increasing generic penetration.
- Manufacturing costs for generics are substantially lower, enabling discounted pricing.
- Regional price regulations influence retail and insurance reimbursement levels.
Future Price Projections
| Year | US Average Price per 25 mg Tablet | Global Price Estimate | Remarks |
|---|---|---|---|
| 2023 | USD 4.50 | USD 4–6 | Continued generic competition; price stability expected |
| 2025 | USD 4.00 | USD 3.50–5 | Increased market penetration; regulatory pressures |
| 2030 | USD 3.50 | USD 3–4 | Widespread generic availability, potential price stabilization |
Prices are expected to decline modestly over the next decade owing to market saturation and manufacturing efficiencies.
Patent and Regulatory Landscape
Pfizer’s patent for Aromasin expired in 2018 for most markets. Some regions retain data exclusivity periods until 2022–2023, delaying generic entry. Regulatory approvals for generics are largely aligned with US FDA and EMA standards.
Market Challenges
- Pricing pressures from generics limit revenue potential.
- Patent litigation in certain regions may delay generic availability.
- Competition from new hormonal agents and combination therapies.
Opportunities
- Expansion into developing markets with rising breast cancer prevalence.
- Development of biosimilar or combination formulations.
- Increasing use of aromatase inhibitors in premenopausal women under specific therapy protocols.
Price and Market Growth Projections Summary
The global exemestane market is expected to grow at a compound annual growth rate (CAGR) of approximately 4% from 2023 to 2030. Price declines are projected to continue slowly, with US prices stabilizing around USD 3–4 per tablet by 2030.
Key Takeaways
- The market for exemestane is driven by rising breast cancer incidence and acceptance of endocrine therapies.
- Patent expirations have led to increased generic options, exerting downward pressure on prices.
- Market growth is most robust in Asia-Pacific and developing regions.
- Price projections suggest modest declines, with stabilization expected as generics dominate.
- Competitive pressures and regional regulatory environments influence pricing strategies.
FAQs
1. How does the patent status affect exemestane pricing?
Patent expiry in 2018 allowed generic manufacturers to enter the market, leading to significant price reductions and increased competition.
2. What factors influence the growth of exemestane in emerging markets?
Rising breast cancer rates, expanding healthcare access, and increasing awareness drive growth. Regulatory approvals and price affordability also play roles.
3. Are biosimilars expected for exemestane?
No. Exemestane is a small-molecule drug, not a biologic, thus biosimilars are not applicable. Generics serve as the primary competitive alternative.
4. What is the average wholesale price for exemestane?
In the US, approximately USD 4.50 per 25 mg tablet for generics as of 2022.
5. How does exemestane compare with other aromatase inhibitors?
It is similar in efficacy but has a unique steroidal mechanism. Cost and patent status differentiate it from non-steroidal alternatives like letrozole and anastrozole.
References
[1] World Health Organization. (2021). Global Cancer Statistics. WHO.
[2] IQVIA. (2022). Global Oncology Drug Market Review.
[3] Pfizer. (2022). Aromasin (Exemestane) Prescribing Information.
[4] European Medicines Agency. (2022). Regulatory Status of Exemestane.
[5] US Food and Drug Administration. (2022). Market Exclusivity and Patent Data for Exemestane.
More… ↓
