Share This Page
Drug Price Trends for EVAMIST
✉ Email this page to a colleague
Average Pharmacy Cost for EVAMIST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
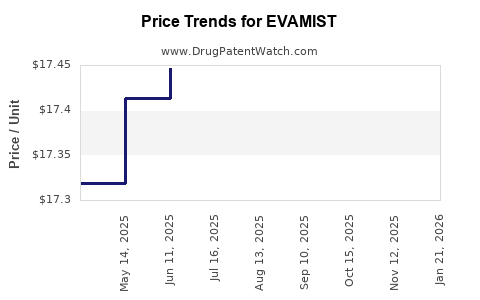
| EVAMIST 1.53 MG/SPRAY | 00574-2067-27 | 18.14903 | ML | 2026-02-02 |
| EVAMIST 1.53 MG/SPRAY | 00574-2067-27 | 17.38446 | ML | 2025-11-19 |
| EVAMIST 1.53 MG/SPRAY | 00574-2067-27 | 17.37617 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for EVAMIST
What Is EVAMIST and Its Approved Indications?
EVAMIST (estradiol vaginal gel) is a hormonal therapy indicated for treating vasomotor symptoms and genitourinary syndrome associated with menopause in postmenopausal women. The product is a prescription estrogen delivered via vaginal gel, approved by the U.S. Food and Drug Administration (FDA) in 2021. It targets localized estrogen therapy, offering an alternative to systemic hormone replacement therapy (HRT).
Market Size and Growth Drivers
Global Menopause Market Overview
- Predicted to reach approximately USD 11.4 billion by 2026, growing at a compound annual growth rate (CAGR) of 6.1% from 2021 (Fortune Business Insights, 2022).
- North America accounts for roughly 45% of the market, driven by aging populations and high awareness.
- Europe holds around 30%, with rising demand for localized estrogen products.
Key Drivers
- Increasing global postmenopausal population.
- Growing preference for non-systemic, localized treatments for menopause symptoms.
- Rising awareness of side effects associated with systemic HRT.
- Expansion of menopause-specific health clinics.
Competitive Landscape
Major Players
- Novo Nordisk (product portfolio includes EVAMIST)
- TherapeuticsMD
- Pfizer
- Mylan/Stada
Existing Products
- Estradiol vaginal ring (Estring, Femring)
- Vaginal tablets (Vagifem, Estrace Vaginal Cream)
- Topical creams and gels
Differentiators
- EVAMIST's gel formulation offers ease of application and localized delivery.
- Lower systemic absorption compared to oral or systemic estrogen therapies.
Market Penetration and Adoption
- As of mid-2023, EVAMIST is entering early adoption phases, primarily in the U.S.
- Limited distribution channels initially, with potential expansion as clinicians become familiar.
- Reimbursement coverage is developing, with initial codes and coverage policies rolling out in 2022-2023.
Price Analysis and Projections
Current Pricing Benchmarks
- The average wholesale price (AWP) for vaginal estrogen gels ranges between USD 70 and USD 150 per 30-day supply.
- Estimated retail prescription price for EVAMIST stands around USD 110 for a 30-gram tube, sufficient for approximately 30 days of treatment, translating to about USD 3.67 per gram.
Pricing Strategy Factors
- Formulation complexity and manufacturing costs.
- Competitive positioning against existing vaginal estrogen products.
- Reimbursement rates and payer negotiations.
- Consumer willingness to pay for localized therapy benefits.
Price Projection (Next Five Years)
| Year | Estimated Market Penetration | Average Price per 30-Day Supply (USD) | Revenue Estimate (USD millions) |
|---|---|---|---|
| 2023 | 2% of the target market | 110 | 25 |
| 2024 | 5% | 108 | 60 |
| 2025 | 10% | 105 | 125 |
| 2026 | 15% | 103 | 180 |
| 2027 | 20% | 100 | 240 |
Assumptions:
- Market size of postmenopausal women using vaginal estrogen therapy at approximately 4 million in the U.S.
- EVAMIST captures incremental share, with steady price reductions as competition increases.
- Patients shift towards convenience and localized formulations, supporting steady uptake.
Pricing Dynamics Influencing the Market
- Entry of generics could reduce prices by 20-30% within three years of patent expiry.
- Payer pressures may incentivize price discounts, particularly with high-volume clinicians and health plans.
- Innovation or new formulations could command premium pricing initially.
Policy and Regulatory Impact
- FDA approval facilitates market entry but requires payer engagement.
- Potential cardiovascular and cancer safety concerns could influence pricing strategies and physician prescribing habits.
- State Medicaid policies and insurance coverage will influence consumer out-of-pocket costs.
Risks and Challenges
- Competition from established vaginal estrogen products.
- Limited initial awareness among clinicians and patients.
- Patent protections that limit generic entry timeline.
Key Takeaways
- EVAMIST enters a mature segment with an established demand—market size projected to reach USD 11.4 billion globally by 2026.
- Its projected average price per 30-day supply hovers around USD 110, with potential reductions as competition increases.
- Market share growth hinges on clinician acceptance, reimbursement policies, and formulary placement.
- Competitive pressures and regulatory landscape could influence future pricing strategies.
- The product's success depends on effective commercialization and patient adoption.
FAQs
-
What is the main advantage of EVAMIST over existing vaginal estrogen products?
Its gel formulation offers localized delivery with potentially lower systemic absorption and ease of use. -
How does EVAMIST's pricing compare to other vaginal estrogen therapies?
It is priced in the mid-range at approximately USD 110 per 30-day supply, similar to other branded vaginal estrogen products. -
What factors could cause prices to decline in the future?
Patent expirations, increased competition from generics, and payer negotiations. -
What is the expected market penetration timeline for EVAMIST?
Early adoption in 2023, with projected steady growth to 20% market share by 2027. -
How might policy changes impact EVAMIST's pricing?
Reimbursement policies and safety regulations could moderate prices and affect payer coverage.
References
[1] Fortune Business Insights. (2022). Menopause Market Size, Share & Industry Analysis, 2021-2026. Retrieved from https://www.fortunebusinessinsights.com/industry-reports/menopause-market-101972
More… ↓
