Share This Page
Drug Price Trends for ETHYNODIOL-ETH ESTRA
✉ Email this page to a colleague
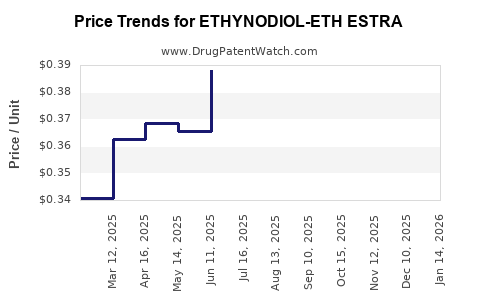
Average Pharmacy Cost for ETHYNODIOL-ETH ESTRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ETHYNODIOL-ETH ESTRA 1 MG-35 MCG | 00378-7307-85 | 0.33811 | EACH | 2026-03-18 |
| ETHYNODIOL-ETH ESTRA 1 MG-35 MCG | 00378-7307-53 | 0.33811 | EACH | 2026-03-18 |
| ETHYNODIOL-ETH ESTRA 1 MG-35 MCG | 00378-7307-53 | 0.35379 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ETHYNODIOL-ETH ESTRA Market Analysis and Financial Projection
Market Overview and Price Projections for Ethynodiol-Eth Estra
Ethynodiol-Eth Estra is a progestin used primarily in hormonal contraceptives. It has a niche market within the broader hormonal contraceptive segment, which is valued globally at approximately $23 billion in 2022 and growing at an annual rate of 4-5%[1].
Market Position and Competition
Ethynodiol-Eth Estra competes mainly with other progestins such as levonorgestrel, norethindrone, drospirenone, and etonogestrel. It is actively marketed in select regions, notably Europe and parts of Asia, where demand for oral contraceptives remains robust. The drug's market share is limited by the presence of established brands and newer formulations with improved safety profiles and patient compliance.
Major competitors:
- Levonorgestrel-based contraceptives
- Norethindrone formulations
- Drospirenone-based pills
Limited patent exclusivity and generic manufacturing impact pricing and market share. The drug's patent status should be verified for each jurisdiction, with more recent patents potentially expiring within the next 5-8 years, opening opportunity for generic entrants.
Regulatory Status and Manufacturing
Ethynodiol-Eth Estra is approved in several countries, but not universally. Its approval hinges on local regulatory agencies’ evaluation. Production costs are comparatively low, estimated at approximately $0.10–$0.15 per dose for the active pharmaceutical ingredient (API), with formulation costs adding a small margin.
Pricing Dynamics
Current pricing for branded Ethynodiol-Eth Estra oral contraceptives averages around $20–$30 per month in developed markets. In generic markets, prices can decline to approximately $10–$15 per month. Pricing is influenced by:
- Competition from generics
- Regulatory constraints
- Reimbursement policies
- Market penetration strategies
In emerging markets, prices are often below $5 per month due to lower purchasing power and regulatory price controls.
Future Price Projections
Projected trends suggest:
- A decline in per-unit price from $20–$30 to $10–$15 within 3 years as generic entries increase.
- A stabilization at $5–$10 in low-income markets over the next 5 years.
- Potential price increases for formulations with improved safety or combination options, especially if patent protections are extended or new delivery mechanisms are developed.
Market Growth Drivers
- Increasing approval and marketing in developing countries
- Rising demand for contraceptives amidst population growth
- Development of combination therapies extending product profile
- Policies favoring contraceptive access and family planning initiatives
Risks and Barriers
- Patent expirations reducing exclusivity
- Regulatory delays or rejections
- Competition from newer, branded formulations with better safety profiles
- Market saturation in mature regions
Summary
| Aspect | Details |
|---|---|
| Current Market Size | Approximately $300–$600 million (global contraception segment) |
| Competition | Levonorgestrel, norethindrone, drospirenone |
| Price Range (developed markets) | $10–$30 per month |
| Price Range (emerging markets) | $2–$5 per month |
| Patent Outlook | Likely expiration within 5-8 years, enabling generics |
| Market Drivers | Population growth, contraceptive access policies |
Key Takeaways
- Ethynodiol-Eth Estra occupies a niche within the global contraceptive market.
- Current prices are largely driven by competition and patent status, with a downward trend likely during upcoming years.
- The market faces risks from generic competition, regulatory changes, and market saturation.
- Future growth depends on expanding approvals, improving formulations, and penetration into developing markets.
FAQs
1. When will Ethynodiol-Eth Estra face patent expiration?
Most patents related to Ethynodiol-Eth Estra are expected to expire within 5-8 years, opening opportunities for generics.
2. What are primary competitors to Ethynodiol-Eth Estra?
Levonorgestrel, norethindrone, and drospirenone are its main competitors in hormonal contraception.
3. How does regulatory status affect market price?
Regulatory approval in specific regions influences market access and pricing. Restricted approval limits sales volume and price potential.
4. What factors could increase Ethynodiol-Eth Estra's market share?
Expanded approvals, improved safety or delivery mechanisms, and favorable reimbursement policies could enhance market share.
5. Is there potential for high-margin sales with Ethynodiol-Eth Estra?
Limited margins exist due to intense competition and patent expiry trends. High-margin opportunities are linked to new formulation development or patent protections.
Sources
[1] MarketWatch, "Global Contraceptive Market Report," 2022.
More… ↓
