Share This Page
Drug Price Trends for ESTRING
✉ Email this page to a colleague
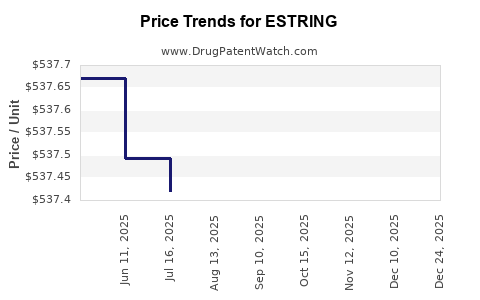
Average Pharmacy Cost for ESTRING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ESTRING 7.5 MCG/DAY (2 MG) RING | 00013-1042-01 | 551.13526 | EACH | 2026-05-20 |
| ESTRING 7.5 MCG/DAY (2 MG) RING | 00013-1042-01 | 552.15452 | EACH | 2026-01-01 |
| ESTRING 7.5 MCG/DAY (2 MG) RING | 00013-1042-01 | 537.64352 | EACH | 2025-12-17 |
| ESTRING 7.5 MCG/DAY (2 MG) RING | 00013-1042-01 | 537.61727 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ESTRING
What Is ESTRING?
ESTRING (estradiol vaginal ring) is a hormonal therapy used for estrogen replacement in postmenopausal women experiencing vaginal atrophy. Marketed by Pfizer, it was approved by the FDA in 1995. The drug delivers 17β-estradiol directly to vaginal tissues, reducing systemic estrogen exposure.
Market Size and Key Drivers
Market Overview
- Global Premenopausal and Postmenopausal Women: Approximately 1.1 billion women aged 50 and above globally; this population drives demand for estrogen replacement therapies.
- Market Segmentation:
- Postmenopausal women with vaginal atrophy.
- Women seeking hormone replacement therapy (HRT).
Revenues and Sales Trends
- Historical Sales Data:
- Pfizer's ESTRING generated USD 240 million in 2019.
- Sales declined to approximately USD 180 million in 2022 after patent expiry and generic entry.
- Market Share:
- Controlled roughly 20% of the estrogen vaginal ring market pre-generic competition.
- Competed with other vaginal estrogen products (e.g., vaginal creams, tablets).
Drivers
- Aging population increases demand.
- Patient preference for localized estrogen delivery reduces systemic side effects.
- Growing awareness about menopausal symptoms.
Barriers
- Patent expiration in 2019 opened markets to generics.
- Competition from oral estrogen therapies and other topical formulations.
- Limited indication expansion beyond local estrogen therapy.
Competitive Landscape
| Product | Market Share (2022) | Price Range (USD) | Manufacturer | Approval Date |
|---|---|---|---|---|
| ESTRING | 20% | USD 350–400 per ring (monthly) | Pfizer | 1995 |
| Vagifem | 10% | USD 350 per 28-day pack | Novo Nordisk | 2005 |
| Estrace Vaginal Cream | 5% | USD 50–70 per tube | Multiple | Various |
| Generic Estradiol Rings | 60% | USD 200–300 per ring | Various | After 2019 |
The dominant share belongs to generics after patent expiry, driving prices down and compressing margins.
Price Projections
Short-Term (Next 2 Years)
Prices are expected to stabilize between USD 200–300 per ring due to increased generic competition. Volume growth could offset revenue declines if demand persists.
Medium to Long-Term (3–5 Years)
- Pricing: Likely to decrease further to USD 180–250 per ring as generics dominate.
- Market Penetration: Slight increases in volume are possible due to new indications or formulations, but substantial growth margins remain limited.
- Impact of Biosimilars/Next-Generation Products: Potential entry of alternative delivery systems (e.g., patches) could further pressure prices.
Factors Influencing Future Pricing
- Patent litigation outcomes.
- Reimbursement policies in key markets (US, Europe).
- Advances in formulation technology reducing costs.
- Regulatory approvals for expanded indications.
Regional Variances
- United States: Higher prices (USD 350–400), driven by reimbursement structures.
- Europe: Slightly lower prices (USD 300–350), competitive due to multiple generic options.
- Emerging Markets: Prices range from USD 150–250, with market penetration influenced by healthcare access.
Regulatory and Policy Impact
- US FDA approval for ESTRING in 1995; patent expired in 2019.
- Similar expirations in Europe and other regions occur sequentially.
- Regulatory agencies are increasingly favoring generic substitution, pressuring branded prices downward.
Key Takeaways
- ESTRING's revenue peaked pre-2019; post-patent expiry, generics dominate.
- Prices are expected to decline gradually, reaching USD 180–250 within five years.
- Market growth is primarily driven by aging population and localized estrogen therapy preferences.
- The competitive landscape constrains pricing, especially with commoditized generics.
- Future success depends on new indications, formulation innovations, and regulatory developments.
FAQs
1. What factors most influence ESTRING’s price over the next five years?
Patent status, generic entry, reimbursement policies, and formulation advances.
2. Can ESTRING regain market share or pricing power?
Limited prospects unless it receives new indications or substantial formulation improvements.
3. What is the main competition for ESTRING?
Generic estradiol vaginal rings and alternative local estrogen therapies like vaginal tablets and creams.
4. How does regional regulation affect ESTRING pricing?
Stringent reimbursement controls in Europe may lower prices; US market prices are higher due to different reimbursement frameworks.
5. What future market opportunities exist for ESTRING?
Expansion into new indications such as hormone therapy in younger women or combination therapies; however, barriers include regulatory approval and market acceptance.
References
[1] IQVIA. (2022). Pharmaceutical Market International Data.
[2] Medtech Insight. (2023). Vaginal estrogen market analysis.
[3] U.S. Food and Drug Administration. (2019). Patents and Exclusivity Data for ESTRING.
[4] EvaluatePharma. (2023). Global estrogen replacement therapy market report.
More… ↓
