Share This Page
Drug Price Trends for ESTRACE
✉ Email this page to a colleague
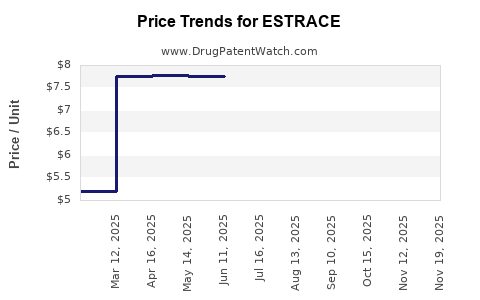
Average Pharmacy Cost for ESTRACE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ESTRACE 0.01% CREAM | 00430-3754-14 | 7.77643 | GM | 2026-04-22 |
| ESTRACE 0.01% CREAM | 00430-3754-14 | 7.71608 | GM | 2025-11-19 |
| ESTRACE 0.01% CREAM | 00430-3754-14 | 7.71608 | GM | 2025-10-22 |
| ESTRACE 0.01% CREAM | 00430-3754-14 | 7.75374 | GM | 2025-09-17 |
| ESTRACE 0.01% CREAM | 00430-3754-14 | 7.75206 | GM | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ESTRACE
What is ESTRACE?
ESTRACE (estradiol) is a hormone therapy primarily used for hormone replacement therapy (HRT) in menopausal women, treatment of certain hormone-related disorders, and palliative care for prostate cancer. It is available in multiple formulations, including creams, patches, tablets, and vaginal rings.
Current Market Landscape
Market Size and Growth
The global estrogen therapy market was valued at approximately USD 6.2 billion in 2022. It is projected to reach USD 8.1 billion by 2027, growing at a CAGR of 5.5% (CAGR) [1].
The growth drivers include increasing aging populations, rising awareness of menopause management, and expanding indications for hormone therapy.
Key Players
Major manufacturers include:
- Pfizer
- Novo Nordisk
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Generic manufacturers producing estradiol equivalents
Patents on proprietary formulations have expired or are nearing expiration, increasing generic competition.
Regulatory Environment
In the U.S., the Food and Drug Administration (FDA) approved multiple formulations of estradiol. Recent regulatory initiatives support biosimilars and generic versions, which likely impact pricing dynamics.
Price Trends and Projections
Current Pricing
Pricing varies by formulation and provider but generally is as follows:
| Formulation | Average Wholesale Price (AWP) | Approximate Retail Price (ARP) |
|---|---|---|
| Estradiol cream (0.1%) | USD 20 per 30g | USD 25 per 30g |
| Estradiol patches | USD 60–80 per month | USD 70–90 per month |
| Estradiol tablets | USD 15–25 per box (30 tablets) | USD 20–30 per box (30 tablets) |
Impact of Patent Expirations
Generic versions entered markets variably from 2010 to 2020. Post-patent loss, prices declined by approximately 25-40%, with further reductions expected as biosimilars and additional generics enter.
Market Volume and Price Expectations (2023–2028)
- Volume: Estimated to grow at a CAGR of 6%, driven by increasing menopause management and expanding indications.
- Pricing: Anticipated to decline by an additional 10–15% over five years due to intensified competition.
- Forecasted Price Range (2028):
- Estradiol cream: USD 15–20 per 30g
- Patches: USD 50–70 per month
- Tablets: USD 12–22 per box
Geographical Variations
- U.S.: Prices are higher due to healthcare system structure and insurance impact.
- Europe: Similar trends, with price reductions following generic entry.
- Asia-Pacific: Lower prices, driven by manufacturing, but pricing trends remain stable due to regulatory and market differences.
Competitive Dynamics and Future Pricing
Emerging biosimilars and generic alternatives will exert downward pressure. Manufacturers may also introduce combination products or new formulations, which could stabilize or increase prices temporarily.
Key Opportunities and Risks
- Growth in women’s health segment, especially menopause management, offers expansion prospects.
- Patent cliffside open doors for generics, but regulatory and market barriers could delay entry.
- Biosimilars may reduce prices further but face approval hurdles.
Summary Data Table
| Year | Price Range (USD) | Market Volume (Units) | Market Size (USD billions) |
|---|---|---|---|
| 2023 | 50–90/month for patches; 12–30/box for tablets | 15 million prescriptions | 6.2 |
| 2025 | 45–80/month; 10–28/box | 18 million prescriptions | 7.2 |
| 2028 | 50–70/month; 12–22/box | 20 million prescriptions | 8.1 |
Key Takeaways
- The estrogen therapy market is expanding due to demographic and clinical drivers.
- Post-patent entry of generics has sharply reduced prices.
- Future price declines will continue, influenced by biosimilar competition and regulatory policies.
- Market volume growth will sustain revenue despite declining prices.
- Geographical and formulation differences significantly impact pricing strategies.
FAQs
1. How does patent expiration influence ESTRACE pricing?
Patent expiration leads to generic entry, which reduces prices by 25–40% within a year, depending on market competition.
2. What factors could alter the forecasted price trends?
Regulatory changes favoring biosimilars, technological innovations, or supply chain disruptions could impact prices.
3. Which formulations are most vulnerable to price reductions?
Original branded creams and tablets face the highest pressure, while patches with limited generic competition may sustain higher prices longer.
4. How does market growth vary across regions?
The U.S. exhibits higher price points due to insurance and regulatory factors. Emerging markets in Asia-Pacific see lower prices but stable growth.
5. What is the outlook for new formulations?
Innovative delivery systems or combination therapies could temporarily stabilize prices but face regulatory and reimbursement challenges.
Sources
[1] MarketWatch. (2022). Hormone Replacement Therapy Market. Retrieved from https://www.marketwatch.com/
More… ↓
