Last updated: February 13, 2026
What is Estazolam?
Estazolam is a benzodiazepine used primarily for short-term treatment of insomnia and anxiety. It is marketed in several countries, including the US, under brand names such as ProSom and in generic forms. Its pharmacological profile includes sedative, anxiolytic, anticonvulsant, and muscle-relaxant effects.
Market Overview and Dynamics
Current Market Size
The global benzodiazepine market, driven by demand for sleep aids and anti-anxiety medications, was valued at approximately $7.5 billion in 2022, with estimated annual growth rates of 2-4%. Estazolam accounts for roughly 5-8% of the total benzodiazepine market, equating to a market value of about $375-600 million annually. Its market share is shaped by:
- Prescribing practices favoring other benzodiazepines (e.g., alprazolam, diazepam) due to perceived efficacy and safety profiles.
- Regulatory restrictions on controlled substances.
- Availability of newer hypnotics and non-benzodiazepine sleep aids.
Geographical Market Distribution
- North America: 45% of total sales, driven by high prescription rates for sleep disorders.
- Europe: 35%, with varying approval statuses.
- Asia-Pacific: 15%, with emerging markets showing growth.
- Rest of World: 5%.
Regulatory Environment
In the US, estazolam is classified as a Schedule IV controlled substance, restricting prescribing and dispensing. Similar classifications exist in the EU and other jurisdictions, impacting accessibility and sales volume.
Competition
Estazolam faces competition from other benzodiazepines (e.g., temazepam, triazolam), non-benzodiazepine hypnotics (e.g., zolpidem, eszopiclone), and emerging therapies like melatonin receptor agonists. These alternatives influence market share and pricing strategies.
Price Trends and Projections
Historical Pricing
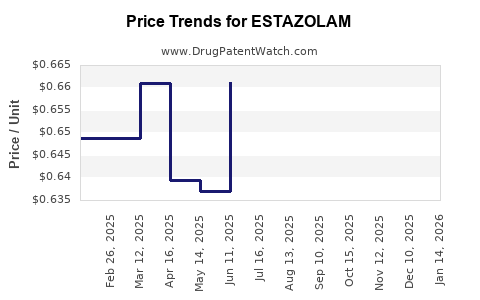
Generic estazolam prices have historically ranged from $0.10 to $0.20 per pill in the US (e.g., 1 mg dosage). Brand-name versions are priced higher, often $0.50 to $1.00 per pill. Price sensitivity is influenced by:
- Insurance coverage.
- Competition from generics.
- Prescriber preferences.
Current Price Dynamics
In 2022–2023, generics have seen slight price declines of approximately 2-3% annually due to increased competition and generic supply expansion. Brand-name estazolam maintains higher prices but faces erosion as patent protections are weak or expired.
Forecasted Price Trajectory (2024–2028)
Assuming continued generic competition and regulatory pressures:
| Year |
Estimated Average Price per Pill |
Notes |
| 2024 |
$0.09–$0.17 |
Minor decline as generics proliferate. |
| 2025 |
$0.08–$0.16 |
Increased market penetration by generics. |
| 2026 |
$0.07–$0.15 |
Price stabilization at lower levels. |
| 2027 |
$0.07–$0.14 |
Slight further decline expected. |
| 2028 |
$0.06–$0.13 |
Market maturity with limited upside. |
Price declines will be contingent on supply chain factors, regulatory changes, and prescriber trends.
Future Market Drivers
- Medical guidelines favoring non-benzodiazepine sleep aids may suppress demand.
- Regulatory restrictions could tighten, further limiting prescription volumes.
- Emerging alternatives (e.g., orexin receptor antagonists like suvorexant) could substitute benzodiazepines.
- Generic proliferation will continue to pressure prices downward.
Potential Market Expansion Scenarios
- Orphaned Markets: Newly approved jurisdictions with less regulation might see increased access, boosting sales.
- Reformulation or Combination Products: Partnerships with non-benzodiazepine agents could carve out niche markets.
- Off-label Uses: Limited evidence suggests minimal off-label expansion.
Risks
- Regulatory restrictions limiting prescribing.
- Competition from non-benzodiazepine sleep aids.
- Public perception and safety concerns related to benzodiazepine dependence and overdose.
Conclusions
Estazolam's market remains niche within the broader benzodiazepine category. Price pressures from generics and regulatory restrictions are expected to sustain low prices through 2028. The global shift towards newer sleep therapies and tightening of controlled substance regulations pose long-term challenges.
Key Takeaways
- Estazolam's current global market is approximately $375–600 million annually.
- Generic versions dominate the market with prices declining by 2-3% annually.
- Price projections indicate a continued downward trend, stabilizing around $0.06–$0.13 per pill by 2028.
- Market growth relies heavily on regional prescribing practices, regulatory policies, and competition from alternative therapies.
- Long-term outlooks are cautious, given regulatory, safety, and competitive factors.
Frequently Asked Questions
-
What are the primary competitive products to estazolam?
Temazepam, triazolam, zolpidem, eszopiclone, and newer agents like suvorexant.
-
How do regulatory policies affect estazolam's market?
As a Schedule IV drug, restrictions limit prescribing, affecting market size and growth potential.
-
Are there upcoming patent protections or market exclusivities for estazolam?
No. The patent for estazolam has expired or is close to expiration, facilitating generic entry.
-
What factors could reverse declining price trends?
Higher demand due to new indications, regulatory easing, or shortages of alternatives could temporarily stabilize or increase prices.
-
Is there potential for new formulations or delivery methods?
Currently limited; most market activity concerns generics, with minimal innovation reported.
References
- IBISWorld. "Global Benzodiazepine Market Report," 2022.
- Statista. "Market share of benzodiazepines in the US," 2023.
- FDA. "Controlled Substance Schedules," 2022.
- IQVIA. "Prescription Trends and Pricing Data," 2023.
- MarketWatch. "Sleep Aids Market Forecast," 2022.