Share This Page
Drug Price Trends for ESOMEPRAZOLE DR
✉ Email this page to a colleague
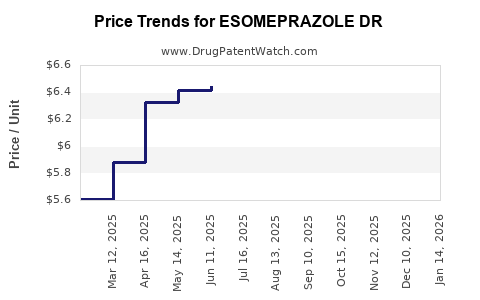
Average Pharmacy Cost for ESOMEPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ESOMEPRAZOLE DR 10 MG PACKET | 59651-0803-30 | 6.19832 | EACH | 2026-05-13 |
| ESOMEPRAZOLE DR 5 MG PACKET | 59651-0802-30 | 6.21689 | EACH | 2026-05-13 |
| ESOMEPRAZOLE DR 20 MG PACKET | 59651-0804-30 | 5.47006 | EACH | 2026-05-13 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Esomeprazole DR Market Analysis and Price Projections
What is Esomeprazole DR?
Esomeprazole DR (Delayed-Release) is a proton pump inhibitor (PPI) used primarily for treating gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. The drug is marketed under various brand names, notably Nexium, and is available both as a prescription and over-the-counter (OTC) medication.
Market Overview
Global Revenue and Volume Trends
In 2022, the global market for PPI drugs, including esomeprazole, was valued at approximately USD 9.6 billion. Esomeprazole represents roughly 45% of the market share among PPIs, driven by its high potency and efficacy. The market has grown at a compound annual growth rate (CAGR) of approximately 4.5% from 2017 to 2022.
Key Market Segments
- Prescription Segment: Accounts for 70% of revenue.
- OTC Segment: Growing at a CAGR of 6%, driven by availability and consumer awareness.
- Geographic Distribution:
- North America: 45%
- Europe: 25%
- Asia-Pacific: 20%
- Rest of the world: 10%
Competitive Landscape
Major players include AstraZeneca (Nexium), Takeda Pharmaceuticals, and Teva Pharmaceuticals. Generic versions of esomeprazole are widely available, reducing prices and margins, especially outside the US.
Pricing Dynamics and Regulatory Impact
Branding vs. Generics
- Brand-Name (Nexium): Average wholesale price (AWP) is USD 250 for a 30-day supply.
- Generic versions: Range from USD 50 to USD 75 for the same period.
Patent Status and Market Entry
- Nexium's primary patent expired in the US in 2015 but held secondary patents and formulations until 2019, after which generics flooded the market.
- In the EU, patent expiry occurred in 2014. Market share shifted rapidly toward generics post-expiry.
Price Trends
- Post-generic entry, average prices declined by up to 80% over five years.
- OTC prices are approximately 30-50% lower than prescription prices, affecting overall revenue streams.
Policy and Payer Dynamics
- Price controls in countries like the UK, Canada, and Australia influence pricing.
- US prices are largely influenced by pharmacy benefit managers (PBMs) and insurance reimbursements, leading to variable net prices.
Projection of Future Market and Prices (2023–2028)
Market Growth
- Expected CAGR of 3.8% for esomeprazole (including generics) through 2028, driven by aging populations and increasing GERD prevalence.
- Uptake of OTC versions is projected to expand at a CAGR of 5%, especially in emerging markets.
Price Projections
| Year | Prescription Price Range | OTC Price Range | Expected Market Share (Generic/Brand) |
|---|---|---|---|
| 2023 | USD 80–USD 120 | USD 30–USD 50 | 60% generics, 40% brand |
| 2025 | USD 70–USD 100 | USD 25–USD 45 | 70% generics, 30% brand |
| 2028 | USD 60–USD 90 | USD 20–USD 40 | 75% generics, 25% brand |
Prices will stabilize as patent exclusivity diminishes further, with discounting campaigns increasing competitive pressure.
Key Regional Variations
- U.S.: Prices declining moderately due to competition and formulary dynamics.
- Europe: Price caps and reimbursement policies suppress prices; expected to remain steady.
- Asia-Pacific: Growing demand will sustain higher prices in certain markets; generics will penetrate rapidly.
Key Factors Impacting the Market
- Patent litigation and expiry timelines.
- Regulations governing OTC switches.
- Consumer preferences toward OTC versus prescription.
- Entry of new PPI formulations or alternative therapies.
- Pricing regulations in different jurisdictions.
Risks to Market Projections
- Emergence of new drug classes (e.g., potassium-competitive acid blockers).
- Regulatory restrictions impacting OTC availability.
- Price pressure from increased generic competition.
- Clinical guidelines influencing prescribing habits.
Key Takeaways
- The global esomeprazole market is transitioning from high-priced brands to widespread generics, reducing overall prices.
- Market growth continues modestly, supported by aging populations and GERD prevalence.
- Price declines will persist due to patent expiries and increased generic competition, especially outside North America.
- OTC sales will contribute significantly to overall volume growth, but at lower prices.
- Regulatory policies will influence regional pricing dynamics, especially in Europe and Asia.
FAQs
1. When will the patent expiry for esomeprazole occur globally?
Patent expiry for Nexium has already occurred in Europe (2014) and the US (2015). Other markets may have different timelines affected by secondary patents and regulatory actions.
2. How do generic versions of esomeprazole impact prices?
Generics typically reduce prices by up to 80%, leading to lower revenue from branded formulations but increasing overall market volume.
3. What are the key factors influencing esomeprazole pricing in emerging markets?
Regulatory policies, market penetration of generics, and consumer demand for OTC options are primary factors.
4. How does OTC availability affect the market?
OTC sales increase overall volume and accessibility but at lower price points, compressing margins for manufacturers.
5. What are the competitive advantages for new entrants?
Developing formulations with improved bioavailability, fixed-dose combinations, or reduced side effects can provide differentiation amid price competition.
References
[1] Market Research Future, 2022. Proton Pump Inhibitors Market Trends and Forecast.
[2] IMS Health, 2022. Global Prescription Drug Market Analysis.
[3] U.S. Patent and Trademark Office, 2015. Patent Expiry Dates for Nexium.
[4] European Medicines Agency, 2014. Patent and Market Authorization Data.
[5] IQVIA, 2022. Price Trends for Proton Pump Inhibitors.
More… ↓
