Share This Page
Drug Price Trends for ERYTHROMYCIN-BENZOYL GEL
✉ Email this page to a colleague
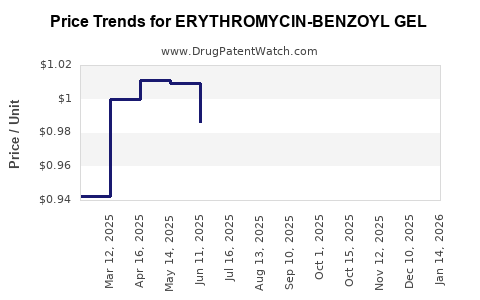
Average Pharmacy Cost for ERYTHROMYCIN-BENZOYL GEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ERYTHROMYCIN-BENZOYL GEL | 64980-0328-01 | 1.98999 | GM | 2026-04-22 |
| ERYTHROMYCIN-BENZOYL GEL | 64980-0328-02 | 0.97645 | GM | 2026-04-22 |
| ERYTHROMYCIN-BENZOYL GEL | 64980-0328-01 | 1.79381 | GM | 2026-03-18 |
| ERYTHROMYCIN-BENZOYL GEL | 64980-0328-02 | 0.97352 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ERYTHROMYCIN-BENZOYL GEL Market Analysis and Financial Projection
What is the current market for erythromycin-benzoyl gel?
Erythromycin-benzoyl gel combines erythromycin, an antibiotic, with benzoyl peroxide, a keratolytic agent, typically used for acne treatment. It is available in several formulations, mainly over-the-counter (OTC) and prescription-based, in markets such as the US, Europe, and Asia. The global acne treatment market was valued at approximately $5 billion in 2022, with topical antibiotics accounting for about 10% of the segment [1].
How does erythromycin-benzoyl gel compare to other acne treatments?
Compared to alternative treatments like clindamycin-based gels, tretinoin, or oral antibiotics, erythromycin-benzoyl gel offers a combination of antibacterial and anti-inflammatory effects. Its market share remains modest due to competition from higher-resistance rates, OTC benzoyl peroxide products without antibiotics, and newer agents such as azelaic acid and doxycycline.
| Treatment Option | Efficacy | Resistance Potential | Market Penetration (Estimated) | Cost Range (USD) |
|---|---|---|---|---|
| Erythromycin-benzoyl gel | Moderate | Increasing | 3-5% | 15-25 per tube |
| Clindamycin gels | High | High | 10-15% | 20-30 per tube |
| Benzoyl peroxide OTC | Moderate | Low | 30-40% | 5-10 per tube |
| Doxycycline (oral) | High | Moderate | 8-12% | 15-30 per pill |
What are the current market dynamics influencing erythromycin-benzoyl gel?
The sector is shaped by antibiotic resistance concerns, regulatory approvals, and patient preferences. The emergence of resistant strains has led clinicians to favor other antibiotics and non-antibiotic agents. Regulatory agencies increasingly push for restricted antibiotic use to limit resistance. The global emphasis on antibiotic stewardship impacts sales prospects.
Manufacturers are investing in reformulations or combination therapies that address resistance. Additionally, newer, non-antibiotic topical treatments have gained consumer interest due to safety profiles.
What are the forecasts for erythromycin-benzoyl gel pricing?
Pricing projections depend on market adoption, patent status, and regional regulations. Currently, the average price point per tube (15-30 grams) resides within $15 to $25.
Future price trends face constraints:
- Patent Expiry: Erythromycin formulations are often off-patent, leading to price erosion.
- Over-the-Counter Alternatives: Rise in OTC benzoyl peroxide products reduces willingness to pay premium prices for combination gels.
- Resistance and Regulation: Increased restrictions on antibiotics can raise costs due to labeling and prescription requirements.
Based on market trends, prices are likely to decrease marginally over the next 3-5 years, settling in the $12-$20 range due to increased competition and generic manufacturing.
What is the outlook for market growth?
The overall acne treatment market is projected to grow at 4-6% annually through 2028 [1]. However, erythromycin-benzoyl gel's market share may decline modestly if resistance issues persist and consumers shift toward OTC and novel non-antibiotic formulations.
Innovations like improved delivery systems or combination therapies may stabilize or modestly increase its market presence.
What regulatory and patent issues impact erythromycin-benzoyl gel?
Many erythromycin formulations lack patent protection, leading to generic proliferation. Regulatory agencies, such as the FDA, have emphasized the importance of antibiotic stewardship, potentially restricting prescriptions or incentivizing alternative therapies.
Manufacturers with proprietary formulations or delivery systems may retain pricing power for a limited period. Once patents expire, price competition intensifies, diminishing profit margins.
Key Takeaways
- The global acne treatment market was valued at around $5 billion in 2022, with topical antibiotics representing about 10%.
- Erythromycin-benzoyl gel holds a small segment due to resistance trends and competition from OTC and newer agents.
- Current pricing averages $15-$25 per tube, with future prices likely declining to $12-$20 amid increased generics.
- Resistance and regulatory pressures are a primary concern, influencing market size and growth.
- Innovation and regulatory strategies will shape the drug's future market penetration.
FAQs
1. How does resistance influence erythromycin-benzoyl gel sales?
Rising bacterial resistance reduces efficacy, limiting prescription volume and encouraging shift toward alternative treatments.
2. Are there patent protections for erythromycin-benzoyl gels?
Most formulations are off-patent, leading to increased generic competition and price erosion.
3. What regions are the largest markets for this drug?
The US, Europe, and parts of Asia-Pacific dominate, driven by high acne prevalence and regulatory environments.
4. How does the price of erythromycin-benzoyl gel compare to other acne therapies?
It is generally more expensive than OTC benzoyl peroxide but cheaper than branded topical or oral antibiotics.
5. What are prospects for new formulations of erythromycin-benzoyl gel?
Limited, due to resistance issues; focus is on alternative compounds and delivery mechanisms.
[1] MarketDataEnterprise, "Global Acne Treatment Market Insights," 2022.
More… ↓
