Share This Page
Drug Price Trends for ERYTHROMYCIN
✉ Email this page to a colleague
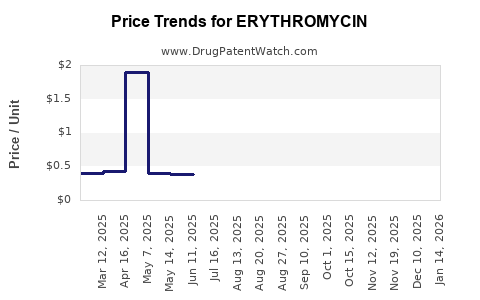
Average Pharmacy Cost for ERYTHROMYCIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ERYTHROMYCIN 2% SOLUTION | 45802-0038-46 | 0.34583 | ML | 2026-05-20 |
| ERYTHROMYCIN 0.5% EYE OINTMENT | 72485-0670-35 | 2.86414 | GM | 2026-05-20 |
| ERYTHROMYCIN 2% GEL | 45802-0966-96 | 0.72052 | GM | 2026-05-20 |
| ERYTHROMYCIN 0.5% EYE OINTMENT | 00574-4024-35 | 2.86414 | GM | 2026-05-20 |
| ERYTHROMYCIN 2% GEL | 45802-0966-94 | 0.60345 | GM | 2026-05-20 |
| ERYTHROMYCIN 0.5% EYE OINTMENT | 24208-0910-55 | 2.86414 | GM | 2026-05-20 |
| ERYTHROMYCIN 2% SOLUTION | 42571-0384-25 | 0.34583 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ERYTHROMYCIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ERYTHROMYCIN BASE 250MG TAB | Golden State Medical Supply, Inc. | 51407-0855-30 | 30 | 113.54 | 3.78467 | EACH | 2024-05-15 - 2028-06-14 | FSS |
| ERYTHROMYCIN BASE 500MG TAB | Golden State Medical Supply, Inc. | 51407-0856-30 | 30 | 150.68 | 5.02267 | EACH | 2024-05-15 - 2028-06-14 | FSS |
| ERYTHROMYCIN 2% PLEDGET SOLN,TOP | Golden State Medical Supply, Inc. | 61748-0202-60 | 60 | 18.75 | 0.31250 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ERYTHROMYCIN ES 200MG/5ML SUSP,PWD (10 DAY) | Golden State Medical Supply, Inc. | 62559-0440-01 | 100ML | 228.58 | 2.28580 | ML | 2023-06-15 - 2028-06-14 | FSS |
| ERYTHROMYCIN 0.5% OINT,OPH,PKG,1GM | Golden State Medical Supply, Inc. | 24208-0910-19 | 50X1GM | 272.00 | 2023-11-10 - 2028-06-14 | FSS | ||
| ERYTHROMYCIN ES 200MG/5ML SUSP,PWD (10 DAY) | Golden State Medical Supply, Inc. | 62559-0440-02 | 200ML | 193.27 | 0.96635 | ML | 2023-06-15 - 2028-06-14 | FSS |
| ERYTHROMYCIN BASE 250MG TAB,EC | Golden State Medical Supply, Inc. | 51407-0853-30 | 30 | 58.62 | 1.95400 | EACH | 2024-05-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Erythromycin: Patent Landscape and Market Outlook
Erythromycin, a macrolide antibiotic, faces a complex patent and market landscape characterized by expiring primary patents, the emergence of generic competition, and the development of newer macrolide derivatives. The original compound is largely off-patent globally, allowing for broad generic accessibility. However, ongoing patent activity centers on novel formulations, delivery systems, and new uses of erythromycin and its related compounds. Market projections indicate a stable but mature demand for erythromycin, with price sensitivity driven by generic availability and competition from alternative antibiotics.
What is the Current Patent Status of Erythromycin?
The foundational patents covering the discovery and initial synthesis of erythromycin have long expired. Erythromycin was first isolated in 1952 by Eli Lilly and Company [1]. As a result, the active pharmaceutical ingredient (API) itself is available as a generic. Patent protection now primarily focuses on:
- New Chemical Entities (NCEs) derived from erythromycin: These are modified versions of the erythromycin molecule, often designed to improve efficacy, reduce side effects, or overcome resistance. Examples include clarithromycin and azithromycin, which have their own patent histories.
- Novel Formulations: Patents may cover specific dosage forms, such as extended-release formulations, topical preparations, or ophthalmic solutions, designed to enhance patient compliance or therapeutic outcomes.
- Manufacturing Processes: Improvements or novel methods for synthesizing erythromycin or its derivatives can be patented, offering a competitive advantage to manufacturers.
- New Medical Uses: Discovering and patenting new therapeutic applications for erythromycin, even if the drug itself is off-patent, can provide market exclusivity for that specific indication.
Key Patent Expirations for Erythromycin API:
- Original discovery and synthesis patents expired in the mid-to-late 20th century. Specific dates vary by country due to differing patent laws and extension periods at the time of original filing. For the United States, original patents would have expired decades ago.
Current Patent Trends:
- Focus on Generics and Biosimilars (for Biologics): While erythromycin is a small molecule, the principle of generic competition is paramount. The market is heavily influenced by the cost-effectiveness of generic erythromycin products.
- Intellectual Property for Derivatives: Significant patent activity exists for second and third-generation macrolides like azithromycin and clarithromycin, which are chemically related to erythromycin but possess distinct pharmacological profiles and patent protection.
- Formulation Patents: These are a critical area for ongoing innovation. For instance, a patent might cover a specific enteric coating that ensures erythromycin's release in the small intestine, improving its stability and absorption [2].
- Combination Therapies: Patents may also be filed for fixed-dose combinations of erythromycin with other active pharmaceutical ingredients for synergistic effects, though this is less common for established antibiotics.
How Do Derivatives and Extended-Release Formulations Impact the Market?
The development of erythromycin derivatives and advanced formulations has significantly shaped the antibiotic market by addressing limitations of the parent compound and extending its commercial lifecycle.
Erythromycin Derivatives:
-
Azithromycin and Clarithromycin: These are the most prominent semi-synthetic macrolide derivatives of erythromycin.
- Azithromycin: Offers improved pharmacokinetic properties, including a longer half-life allowing for once-daily dosing and shorter treatment courses. It is effective against a broader spectrum of bacteria, including some Gram-negative pathogens [3]. Azithromycin has its own patent expiry timeline, with generics now widely available, impacting the market for original branded azithromycin.
- Clarithromycin: Also exhibits enhanced acid stability and a broader spectrum of activity compared to erythromycin, particularly against Helicobacter pylori and certain atypical pathogens [3]. Like azithromycin, it faces robust generic competition.
-
Market Impact: The introduction of these derivatives created new market segments and often displaced erythromycin for certain indications where their improved profiles offered a clear clinical advantage. However, as their primary patents have expired, the market for these derivatives is also dominated by generics, leading to price erosion. The clinical utility and cost-effectiveness of erythromycin API remain relevant for specific indications and markets.
Extended-Release (ER) Formulations:
- Purpose: ER formulations of erythromycin are designed to maintain therapeutic drug concentrations for a longer duration, reducing dosing frequency and potentially improving patient adherence. They also aim to minimize gastrointestinal side effects often associated with immediate-release formulations.
- Examples: Various ER technologies have been developed, including osmotic pump systems, matrix tablets, and coated pellets.
- Patent Significance: Patents on specific ER technologies can provide market exclusivity for a branded product, even if the underlying API is off-patent. These patents are often time-limited but can significantly influence pricing and market share during their term.
- Market Impact: ER formulations can command a premium price over immediate-release generics. However, the expiration of patents on ER technologies leads to the introduction of generic ER versions, which can significantly reduce pricing and introduce competition. For example, U.S. Patent 5,490,999, which describes an extended-release pharmaceutical formulation of erythromycin, has long expired. New patents focus on novel approaches to ER delivery.
What Are the Key Therapeutic Uses and Their Market Significance?
Erythromycin remains a vital antibiotic due to its efficacy against a range of common bacterial infections. Its primary therapeutic uses and associated market significance are:
-
Respiratory Tract Infections:
- Indications: Community-acquired pneumonia, bronchitis, sinusitis, pharyngitis, and tonsillitis caused by susceptible organisms like Streptococcus pyogenes and Streptococcus pneumoniae.
- Market Significance: This is a core market for erythromycin. Despite the availability of newer agents, erythromycin's cost-effectiveness and established safety profile make it a first-line option, especially in resource-limited settings or for patients with penicillin allergies.
-
Skin and Soft Tissue Infections:
- Indications: Impetigo, cellulitis, and erysipelas caused by Staphylococcus aureus and Streptococcus pyogenes.
- Market Significance: A significant application, particularly for topical erythromycin formulations. The generic availability of topical products makes this a competitive segment.
-
Gastrointestinal Infections:
- Indications: Used as a prokinetic agent to stimulate gastrointestinal motility in conditions like gastroparesis, particularly in diabetic patients. It also treats certain bacterial gastroenteritis.
- Market Significance: The prokinetic use is a distinct and valuable niche. While other prokinetic agents exist, erythromycin's mechanism of action provides a therapeutic option.
-
Ophthalmic Infections:
- Indications: Conjunctivitis and blepharitis caused by susceptible bacteria.
- Market Significance: Topical ophthalmic preparations are widely used, especially for neonates (prophylaxis against ophthalmia neonatorum) and in over-the-counter (OTC) formulations for common eye infections.
-
Other Infections:
- Indications: Diphtheria (as an adjunct to antitoxin), pertussis (as prophylaxis and treatment), and as an alternative for penicillin-allergic patients in treating syphilis and gonorrhea.
- Market Significance: These represent smaller but important applications, highlighting erythromycin's broad utility and role in public health.
Market Considerations for Therapeutic Uses:
- Antibiotic Resistance: The increasing prevalence of bacterial resistance to erythromycin and other macrolides is a significant challenge. This can limit its empirical use and necessitate susceptibility testing, impacting treatment patterns.
- Competition from Other Antibiotics: Broad-spectrum antibiotics, cephalosporins, and fluoroquinolones offer alternative treatments for many of the same infections, creating competitive pressure.
- Cost-Effectiveness: For many indications, erythromycin's low cost as a generic antibiotic makes it highly competitive, particularly in emerging markets and for large-scale public health programs.
What Are the Projected Market Sizes and Price Trends for Erythromycin?
The market for erythromycin API is mature and largely driven by generic production. Projections indicate continued stability with price sensitivity.
Market Size:
- Global Market Value: Estimating the precise global market value solely for erythromycin API is challenging due to the widespread use of generics and its inclusion in combination products. However, the overall macrolide market is substantial. Some reports place the global macrolide antibiotics market in the tens of billions of dollars, with erythromycin and its derivatives forming a significant portion. For example, the global macrolide antibiotics market was valued at approximately $13.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 3.5% through 2030 [4]. Erythromycin API itself contributes a significant, though declining, share of this as newer macrolides and other antibiotic classes gain traction.
- Volume: The volume of erythromycin produced and consumed globally remains high due to its widespread use in various formulations and indications, particularly in developing countries.
Price Trends:
-
API Pricing: The price of erythromycin API is highly competitive and subject to global supply and demand dynamics.
- Generic API: Prices for bulk erythromycin API have historically been low and are expected to remain so, likely within the range of $10 to $30 per kilogram, depending on purity, grade, and supplier volume [5]. This is influenced by manufacturing costs and competition among API producers, primarily in Asia.
- Fluctuations: Prices can fluctuate based on raw material costs, production capacities, and geopolitical factors affecting supply chains.
-
Finished Dosage Forms (FDFs):
- Immediate-Release Generics: Prices for generic erythromycin tablets or capsules are very low, often ranging from $0.05 to $0.20 per unit dose (e.g., per 250mg capsule), depending on the market and packaging.
- Extended-Release Generics: Generic ER formulations are priced higher than immediate-release versions but significantly lower than branded ER products during their patent exclusivity. Prices might range from $0.50 to $2.00 per unit dose, reflecting the more complex manufacturing involved.
- Topical and Ophthalmic: These formulations also follow a generic pricing model, with competitive pricing among manufacturers.
- Branded Products (for Derivatives/Specific Formulations): While erythromycin API is generic, branded derivatives (like azithromycin or clarithromycin) or patented ER formulations of erythromycin can command higher prices during their exclusivity periods. However, with widespread generic entry for these as well, price erosion is a consistent trend.
Factors Influencing Future Pricing:
- Generic Competition: The ongoing presence of multiple generic manufacturers worldwide will continue to suppress prices for erythromycin API and FDFs.
- Demand in Emerging Markets: Continued demand in regions where cost is a primary factor will sustain sales volume, but at low price points.
- Development of Resistance: Increasing resistance may lead to reduced use in certain indications, potentially impacting overall demand and pricing power.
- Competition from Novel Antibiotics: The introduction of new antibiotic classes or drugs with superior efficacy or resistance profiles could gradually displace erythromycin for specific uses.
- Regulatory Policies: Government pricing regulations and formulary decisions in healthcare systems will influence market access and pricing.
What Are the Major Manufacturers and Their Role in the Market?
The erythromycin market involves a broad spectrum of manufacturers, from API producers to finished dosage form (FDF) manufacturers, with a significant presence of generic drug companies.
Key Players by Category:
-
Erythromycin API Manufacturers:
- Major Asian Producers: A significant portion of global erythromycin API production is concentrated in China and India. Companies such as Shanxi Xinhua Pharmaceutical Co., Ltd. (China), Jiangxi Pharma (China), and several Indian pharmaceutical ingredient manufacturers are key suppliers. These companies focus on large-scale, cost-efficient manufacturing.
- Global Distributors: International chemical and pharmaceutical distributors also play a role in sourcing and supplying API to various markets.
-
Finished Dosage Form (FDF) Manufacturers:
- Generic Pharmaceutical Companies: These are the dominant players in the erythromycin FDF market. Examples include:
- Teva Pharmaceutical Industries Ltd. (Israel)
- Viatris Inc. (USA/Global)
- Sanofi S.A. (France) (though more focused on branded products, it has generic portfolios)
- Mylan N.V. (now part of Viatris)
- Numerous regional and country-specific generic manufacturers (e.g., Dr. Reddy's Laboratories, Sun Pharmaceutical Industries Ltd. in India; Aurobindo Pharma).
- Specialty Formulators: Some companies may specialize in specific erythromycin formulations (e.g., ophthalmic, topical, or ER).
- Generic Pharmaceutical Companies: These are the dominant players in the erythromycin FDF market. Examples include:
-
Manufacturers of Derivatives: Companies that developed and initially marketed branded derivatives like azithromycin and clarithromycin (e.g., Pfizer Inc. for Zithromax/azithromycin, Abbott Laboratories/AbbVie Inc. for Biaxin/clarithromycin) are still involved, but their market share is heavily influenced by generic competition for these products as well.
Role in the Market:
- API Manufacturers: Their role is to produce high-quality, cost-effective erythromycin API that meets pharmacopeial standards (USP, EP, JP). Their pricing and production capacity directly influence the cost of generic FDFs.
- Generic FDF Manufacturers: These companies formulate the API into various dosage forms (tablets, capsules, suspensions, topical creams, ophthalmic solutions) and market them under their own brands or as private label products. They drive accessibility and affordability for patients worldwide.
- Innovation (Limited for Erythromycin API): While direct API innovation for erythromycin is minimal, FDF manufacturers and derivative producers continue to seek value through novel formulations, combination products, or new therapeutic indications, leveraging existing intellectual property or developing new patents.
- Global Supply Chain: The market relies on a complex global supply chain where API is manufactured in one region and formulated into finished products in another, then distributed globally.
Key Takeaways
Erythromycin's market is defined by its status as a mature, off-patent antibiotic API. While foundational patents have expired, intellectual property remains active in areas of novel formulations, delivery systems, and new uses of erythromycin and its derivatives. Generic competition is the primary driver of pricing and market accessibility, resulting in low per-unit costs for API and finished dosage forms. Derivatives like azithromycin and clarithromycin have carved out distinct market segments but also face extensive generic competition. Projected market stability for erythromycin is underpinned by its cost-effectiveness and continued use in established indications, particularly in emerging markets, despite challenges posed by antibiotic resistance and competition from newer agents.
Frequently Asked Questions
-
Are there any active patents that would prevent generic erythromycin production? No, the basic synthesis and composition of matter patents for erythromycin have long expired, allowing for the global production and sale of generic erythromycin. However, patents may exist for specific extended-release formulations, novel combination therapies, or unique manufacturing processes.
-
What are the main factors contributing to the low price of generic erythromycin? The low price is attributed to the absence of patent protection on the active pharmaceutical ingredient, leading to intense competition among numerous global generic manufacturers, primarily in Asia, who focus on high-volume, cost-efficient production.
-
How does antibiotic resistance affect the market for erythromycin? Increasing resistance to erythromycin in certain bacterial strains can reduce its efficacy and lead to a decline in its empirical use for specific infections. This may shift prescribing patterns towards alternative antibiotics, impacting demand for erythromycin in those applications.
-
What is the difference in market potential between erythromycin and its derivatives like azithromycin? Erythromycin API's market is characterized by high volume and low price points, driven by broad-spectrum generic use. Derivatives like azithromycin, while also generic, initially offered improved pharmacokinetic profiles and a wider spectrum, creating distinct market segments. However, as these derivatives have also aged and gone off-patent, their markets too are now heavily price-driven and competitive.
-
Will new therapeutic uses for erythromycin significantly alter its market size or pricing? Discovering and patenting new therapeutic uses for off-patent erythromycin could create temporary market exclusivity for that specific indication, potentially commanding higher prices for the duration of the patent. However, given the drug's established nature and the challenges in antibiotic development, such new uses are unlikely to dramatically alter the overall global market size or pricing trends of generic erythromycin API, which remain dominated by existing applications.
Citations
[1] McVie, J. G. (2001). Macrolides. In General and Vocational Medicine (pp. 309-315). Churchill Livingstone.
[2] U.S. Patent 5,490,999 A. (1996). Extended release pharmaceutical formulation of erythromycin. Issued to Kwon, Y.K., & Yang, D.C.
[3] Zhanel, G. G., Ge, M., Millan, C., Garg, A., & Lagacé-Simard, J. (2011). Macrolide resistance in pediatric patients: prevalence and clinical significance. Clinical Infectious Diseases, 52(Supplement_1), S111-S119.
[4] Grand View Research. (2023). Macrolide Antibiotics Market Size, Share & Trends Analysis Report By Product (Azithromycin, Clarithromycin, Erythromycin, Others), By Application, By Distribution Channel, By Region, And Segment Forecasts, 2023-2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/macrolide-antibiotics-market (Note: Specific values are subject to report access and may vary slightly between market research firms.)
[5] Pharmaceutical industry market intelligence reports (proprietary data and custom research) indicate a general price range for bulk erythromycin API. Specific pricing is highly variable and subject to direct negotiation between suppliers and buyers.
More… ↓
