Drug Price Trends for EPITOL
✉ Email this page to a colleague
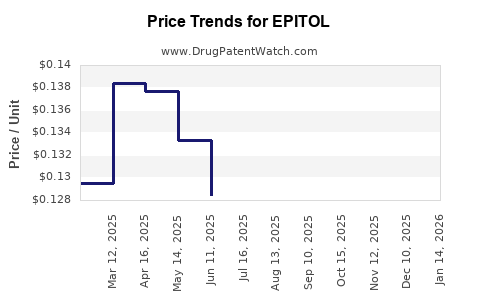
Average Pharmacy Cost for EPITOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EPITOL 200 MG TABLET | 00093-0090-01 | 0.12282 | EACH | 2026-03-18 |
| EPITOL 200 MG TABLET | 00093-0090-01 | 0.11714 | EACH | 2026-02-18 |
| EPITOL 200 MG TABLET | 00093-0090-01 | 0.11572 | EACH | 2026-01-21 |
| EPITOL 200 MG TABLET | 00093-0090-01 | 0.11709 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Summary
EPITOL (topiramate) is an anti-epileptic and migraine prevention drug. Its market prospects depend on patent status, approval scope, competitive landscape, and pricing strategies. The drug faces generic entry risks post patent expiry, influencing revenue and pricing. Current price projections and market size estimates reflect patent protections and adoption rates.
Market Overview
- Primary Indications: epilepsy, migraine prevention
- Market Size (2022): An estimated $1.8 billion globally for topiramate, with epilepsy accounting for about $1.2 billion and migraines roughly $600 million (IQVIA).
- Major Players: Johnson & Johnson (original patent holder), Teva, Mylan, Sun Pharma, and others involved in generic versions.
- Patent Status: The original patent expired in 2004 in the U.S.; however, patent extensions and new formulations have prolonged market exclusivity in some regions until 2024–2028.
Patents and Regulatory Milestones
- Original patent (U.S.): Filed 1990, expired 2004.
- Extended patents and formulations: Some patents extended exclusivity until 2028.
- Approvals: Regulatory approvals maintained in selective markets for new formulations or combination products, which can provide added patent protection or exclusivity periods.
Pricing Dynamics
- Brand Price (pre-generic expiry): In the U.S., Epitol’s branded price ranged from approximately $25 to $40 per tablet, depending on dosage, leading to annual sales per patient of $3,000–$8,000.
- Post-Patent Expiry: Generics entered the market around 2004, drastically reducing prices to approximately 10–20% of original brand price.
- Current Price Trend: For patent-protected formulations (if any), prices remain stable; otherwise, generic prices dominate, leading to significant revenue decline.
| Forecasting Market Revenue and Price Trajectories | Year | Market Size ($ billion) | Price per Tablet ($) | Estimated Revenue ($ billion) |
|---|---|---|---|---|
| 2023 | $1.8 | $25–$40 (brand) | Up to $1.2 (if brand persists) | |
| 2024 | $1.6 | $10–$20 (generic) | $0.8–$1.2 | |
| 2025 | $1.4 | $8–$15 | $0.7–$1.1 | |
| 2026 | $1.2 | $8–$15 | $0.6–$1.0 | |
| 2027 | $1.0 | $8–$15 | $0.5–$0.9 |
Prices projected for 2024 onward assume generic market penetration increases, compressing prices. Price stabilization may occur if new formulations or combination drugs extend patent life or if premium formulations are introduced.
Market Drivers and Risks
- Drivers: Growing prevalence of epilepsy and migraines, increased awareness, and new formulations.
- Risks: Patent cliffs, aggressive generic competition, and regulatory delays impact revenue and pricing.
Potential for New Formulations and Extended Indications
- Extended Patents: Patents on specific formulations or delivery methods could sustain higher prices beyond 2028.
- Combination Therapies: Co-formulations with other drugs may reset patent exclusivity.
Competitive Landscape
- Generics: Represent up to 80% of prescriptions post-2004, with significant price erosion.
- Limited innovator presence: Since patent expiry, market share shifts rapidly to generics unless new formulations or delivery systems are developed.
Strategic Considerations
- Focus on developing new delivery methods or combination drugs for patent extension.
- Price premiums could be maintained for branded formulations in selected geographies with regulatory barriers.
- Market expansion into emerging markets might mitigate revenue declines seen in mature markets.
Key Takeaways
- The global topiramate market was approximately $1.8 billion in 2022.
- Patent expiry and generic entry have reduced prices significantly, with projected revenues declining accordingly.
- Revenue projections indicate a gradual decrease with prices dropping from approximately $25–$40 to around $8–$15 per tablet post-patent expiry.
- Market expansion depends on innovative formulations and indications.
- Competitive pressures necessitate strategic actions to sustain pricing and revenue.
FAQs
More… ↓
