Share This Page
Drug Price Trends for EPINEPHRINE
✉ Email this page to a colleague
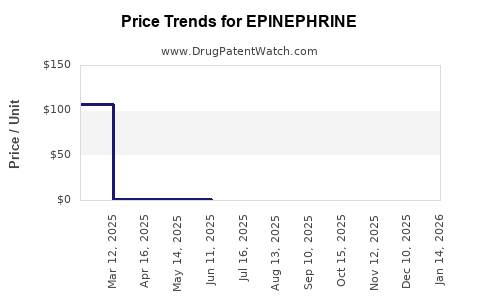
Average Pharmacy Cost for EPINEPHRINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EPINEPHRINE 0.3 MG AUTO-INJECT | 00093-5986-19 | 135.38910 | EACH | 2026-05-20 |
| EPINEPHRINE 0.15 MG AUTO-INJCT | 00115-1695-49 | 120.95481 | EACH | 2026-05-20 |
| EPINEPHRINE 0.15 MG AUTO-INJCT | 49502-0101-02 | 142.93970 | EACH | 2026-05-20 |
| EPINEPHRINE 0.15 MG AUTO-INJCT | 00093-5985-19 | 129.86330 | EACH | 2026-05-20 |
| EPINEPHRINE 0.15 MG AUTO-INJCT | 49502-0101-01 | 142.93970 | EACH | 2026-05-20 |
| EPINEPHRINE 0.15 MG AUTO-INJCT | 00093-5985-27 | 129.86330 | EACH | 2026-05-20 |
| EPINEPHRINE 0.15 MG AUTO-INJCT | 00115-1695-30 | 120.95481 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for EPINEPHRINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| AUVI-Q 0.1MG/0.1ML INJECTOR | Kaleo, Inc. | 60842-0021-01 | 2X0.1ML | 252.45 | 2023-05-15 - 2028-05-14 | FSS | ||
| AUVI-Q 0.15MG/0.15ML INJECTOR | Kaleo, Inc. | 60842-0022-01 | 2X0.15ML | 568.79 | 2024-01-01 - 2028-05-14 | FSS | ||
| AUVI-Q 0.1MG/0.1ML INJECTOR | Kaleo, Inc. | 60842-0021-01 | 2X0.1ML | 252.45 | 2023-06-16 - 2028-05-14 | Big4 | ||
| AUVI-Q 0.3MG/0.3ML INJECTOR | Kaleo, Inc. | 60842-0023-01 | 2X0.3ML | 227.26 | 2023-05-15 - 2028-05-14 | FSS | ||
| AUVI-Q 0.1MG/0.1ML INJECTOR | Kaleo, Inc. | 60842-0021-01 | 2X0.1ML | 546.91 | 2023-06-16 - 2028-05-14 | FSS | ||
| AUVI-Q 0.3MG/0.3ML INJECTOR | Kaleo, Inc. | 60842-0023-01 | 2X0.3ML | 227.26 | 2023-06-16 - 2028-05-14 | Big4 | ||
| AUVI-Q 0.15MG/0.15ML INJECTOR | Kaleo, Inc. | 60842-0022-01 | 2X0.15ML | 179.80 | 2023-05-15 - 2028-05-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Epinephrine Market Analysis and Price Projections
Epinephrine, a catecholamine hormone and neurotransmitter, is critical for treating severe allergic reactions (anaphylaxis), cardiac arrest, and asthma exacerbations. The global epinephrine market is driven by the increasing prevalence of allergic diseases, growing awareness of anaphylaxis management, and advancements in drug delivery devices. Pricing is influenced by manufacturing costs, patent exclusivity, regulatory hurdles, and competitive landscape.
What Factors Drive Epinephrine Demand?
The demand for epinephrine is primarily fueled by several key factors:
- Rising Incidence of Allergic Diseases: Global rates of food allergies, insect sting allergies, and other hypersensitivity reactions are increasing. This rise directly translates to a greater need for immediate treatment with epinephrine auto-injectors. For instance, the prevalence of peanut allergy in the United States has increased significantly, with estimates suggesting it affects up to 3% of children and 1% of adults [1]. Similar trends are observed globally for other allergens.
- Increased Public Awareness and Education: Public health campaigns and patient advocacy groups have successfully raised awareness about the dangers of anaphylaxis and the importance of carrying and knowing how to use epinephrine auto-injectors. This heightened awareness encourages individuals with known allergies to obtain prescriptions and maintain a supply of the medication.
- Guidelines for Anaphylaxis Management: Medical guidelines from organizations like the National Institute of Allergy and Infectious Diseases (NIAID) and the World Allergy Organization (WAO) recommend epinephrine as the first-line treatment for anaphylaxis [2]. These consistent recommendations drive physician prescribing habits and patient access.
- Technological Advancements in Delivery Devices: The development of user-friendly and reliable epinephrine auto-injectors has been a significant driver. Devices like the EpiPen, Auvi-Q, and others are designed for ease of use by patients and caregivers, even in emergency situations. Innovations continue to focus on improving needle safety, dosage accuracy, and device portability.
- Growth in Emergency Medical Services: The expansion of emergency medical services (EMS) and their protocols for anaphylaxis treatment also contributes to epinephrine demand. Paramedics and EMTs are equipped with and trained to administer epinephrine during critical events.
What is the Current Market Size and Segmentation for Epinephrine?
The global epinephrine market is substantial and segmented by application, route of administration, and distribution channel.
Market Size: The global epinephrine market was valued at approximately USD 1.2 billion in 2023 [3]. Projections indicate continued growth, with an estimated compound annual growth rate (CAGR) of 5.5% to 6.0% over the next five to seven years, potentially reaching USD 1.7 billion to 1.9 billion by 2028 [3, 4].
Key Market Segments:
- By Application:
- Anaphylaxis: This is the largest and fastest-growing segment, accounting for over 70% of the market share [3].
- Cardiac Arrest: Used in emergency resuscitation protocols.
- Asthma Exacerbations: Particularly for severe, life-threatening asthma attacks.
- Others (e.g., hypotensive shock, adjunct to local anesthetics).
- By Route of Administration:
- Intramuscular Injection: The predominant route, utilized by auto-injectors and pre-filled syringes.
- Intravenous Injection: Primarily used in hospital settings for cardiac arrest and severe hypotension.
- Inhalation: For certain asthma treatments.
- By Distribution Channel:
- Hospital Pharmacies: A major channel due to inpatient use and emergency department dispensing.
- Retail Pharmacies: For prescription-based sales of auto-injectors.
- Online Pharmacies: Increasing in prevalence for prescription refills.
- Government Tenders: For public health initiatives and stocking emergency supplies.
What is the Competitive Landscape and Key Players?
The epinephrine market is characterized by a few dominant players, particularly in the auto-injector segment, alongside generic manufacturers and injectable formulations.
Key Companies:
- Viatris (formerly Mylan): A leading player with its EpiPen® and EpiPen Jr.® auto-injectors. Viatris holds a significant market share globally.
- Teva Pharmaceutical Industries: Offers generic versions of epinephrine auto-injectors and injectable formulations.
- Kaleo Inc.: Manufacturer of the Auvi-Q® (marketed as Allerject® in Canada), an injector with voice instructions.
- GSK plc: Offers a generic epinephrine auto-injector in some markets.
- Amneal Pharmaceuticals: Provides generic epinephrine products, including injectables.
- Emergent BioSolutions: While historically a major supplier of the U.S. military's anthrax vaccine, they have also been involved in epinephrine production.
Competitive Dynamics:
- Patent Expirations and Generics: The expiration of patents for branded epinephrine auto-injectors has opened the door for generic competition. This has led to price reductions and increased accessibility in some markets. For example, the U.S. saw the introduction of a generic EpiPen (from Teva) in 2021 [5].
- Product Differentiation: Companies are differentiating through device features, such as ease of use (Auvi-Q's voice guidance), needle safety mechanisms, and dual-needle systems.
- Supply Chain Resilience: Recent supply disruptions have highlighted the importance of robust manufacturing and supply chain management. Companies are investing in diversifying production and ensuring consistent availability.
- Regulatory Approvals: Obtaining and maintaining regulatory approvals (e.g., FDA in the U.S., EMA in Europe) for new formulations, delivery devices, or manufacturing sites is critical for market access.
What are the Pricing Trends and Projections for Epinephrine?
Epinephrine pricing is complex, influenced by manufacturing costs, intellectual property, market competition, and payer policies.
Historical Pricing: Branded epinephrine auto-injectors have historically commanded premium prices. For example, the list price of a two-pack of EpiPens could exceed USD 600 in the United States prior to the introduction of significant generic competition [5]. This high pricing became a subject of public and political scrutiny.
Current Pricing Dynamics:
- Generic Impact: The introduction of FDA-approved generic epinephrine auto-injectors has led to significant price erosion. Generic versions are available at substantially lower price points, often ranging from USD 100 to USD 300 for a two-pack, depending on the manufacturer and pharmacy [6].
- Manufacturer Strategies:
- Viatris has introduced a lower-list-price authorized generic of EpiPen, priced at USD 250 for a two-pack, to compete with independent generics [7].
- Kaleo has offered the Auvi-Q with a patient assistance program and a maximum out-of-pocket cost of USD 0 for eligible commercially insured patients, aiming to address affordability concerns [8].
- Inflationary Pressures: While generic competition drives prices down, underlying manufacturing costs, raw material prices, and labor can exert upward pressure. This is a general trend impacting pharmaceutical manufacturing globally.
- Payer Influence: Pharmacy benefit managers (PBMs) and insurance companies negotiate rebates and formulary placements, which can influence the net price paid by the end consumer and the market share of different products.
Price Projections:
- Continued Generic Dominance: The trend towards lower prices is expected to continue as more generic competitors enter or expand their market presence. The average selling price (ASP) for epinephrine auto-injectors is projected to remain lower than historical branded prices.
- Stable to Modest Increase for Branded/Differentiated Products: For differentiated products with unique features (e.g., Auvi-Q’s voice guidance), pricing may remain higher, supported by their unique value proposition and patient assistance programs. However, significant price hikes on the scale seen historically are less likely due to competitive pressures and regulatory scrutiny.
- Overall Market Value Growth Driven by Volume: The projected growth in the overall market value will likely be driven more by increased unit sales (volume) due to rising allergy prevalence and awareness, rather than substantial price increases per unit.
- Injectable Formulations: Prices for generic epinephrine injectable solutions (vials, pre-filled syringes) are generally much lower, typically in the range of USD 5 to USD 50 per unit, and are expected to remain relatively stable with minor fluctuations based on manufacturing costs and competition among generic suppliers.
- Potential for Increased Government Pricing Negotiations: As healthcare systems face increasing costs, there is ongoing pressure for governments and large purchasing groups to negotiate lower prices for essential medicines like epinephrine.
The median out-of-pocket cost for commercially insured patients for a two-pack of epinephrine auto-injectors in the U.S. has decreased from an average of USD 126 in 2022 to USD 90 in 2023, reflecting the impact of generic availability and PBM negotiations [9]. However, uninsured or underinsured individuals may still face significantly higher costs for branded products if available.
What are the Regulatory and Patent Landscape Considerations?
The regulatory and patent landscape significantly shapes market access, competition, and pricing.
Regulatory Bodies:
- U.S. Food and Drug Administration (FDA): Oversees the approval, manufacturing, and labeling of epinephrine products in the United States. The FDA's approval of generic versions is a key driver of market competition.
- European Medicines Agency (EMA): The equivalent regulatory body in the European Union, responsible for authorizing medicinal products.
- Other National Regulatory Agencies: Health Canada, Pharmaceuticals and Medical Devices Agency (Japan), etc., each with their own approval processes.
Key Regulatory Aspects:
- ANDA Pathway: Abbreviated New Drug Applications (ANDAs) are used for generic drug approvals, requiring demonstration of bioequivalence to the reference listed drug.
- Post-Marketing Surveillance: Regulators monitor for safety issues, manufacturing quality, and ensure continued compliance.
- Device Regulations: Epinephrine auto-injectors are considered medical devices, subject to specific regulations regarding design, manufacturing, and user safety.
Patent Landscape:
- Composition of Matter Patents: These patents protect the chemical structure of epinephrine itself and were long expired.
- Formulation Patents: Patents covering specific compositions of epinephrine, excipients, or stabilizers within a drug product.
- Delivery Device Patents: These are often the most significant patents for auto-injector products, covering the mechanics, design, and user interface of the device. Patent expiration for these devices has been a major trigger for generic entry.
- Manufacturing Process Patents: Patents that protect specific methods of synthesizing or purifying epinephrine or manufacturing its delivery device.
Patent Cliffs and Generic Entry:
The expiration of key patents for branded epinephrine auto-injectors has led to the "patent cliff" phenomenon, where market exclusivity ends, and generic competition rapidly emerges. For example, the patent protection for the original EpiPen design has largely expired, allowing for the introduction of generic versions.
Potential for Future Innovation:
While the core molecule is well-established, innovation continues in:
- Novel Delivery Devices: Devices with improved safety, ease of use, connectivity (e.g., for tracking), or integrated features.
- Alternative Formulations: Research into more stable formulations or alternative routes of administration, though these face significant development hurdles and lengthy regulatory review.
Key Takeaways
- The global epinephrine market is projected to grow between 5.5% and 6.0% annually, reaching up to USD 1.9 billion by 2028, driven by rising allergy prevalence and improved awareness.
- Anaphylaxis treatment is the largest application segment, accounting for over 70% of the market.
- Viatris (EpiPen) and Kaleo (Auvi-Q) are key players in the auto-injector segment, facing increasing competition from generic manufacturers.
- The introduction of generic epinephrine auto-injectors has significantly reduced prices, with two-packs now available for USD 100-300, compared to over USD 600 historically for branded products.
- Future market value growth will be primarily volume-driven, with prices for auto-injectors likely to remain lower than pre-generic levels.
- Patent expirations, particularly for delivery devices, have been critical in enabling generic competition and influencing pricing dynamics.
Frequently Asked Questions
1. What is the primary driver of increased epinephrine demand in recent years?
The primary driver is the rising global incidence of allergic diseases, particularly food allergies and insect sting allergies, coupled with enhanced public and medical awareness of anaphylaxis and its immediate treatment needs.
2. How has the introduction of generic epinephrine auto-injectors impacted pricing?
The introduction of FDA-approved generic epinephrine auto-injectors has led to substantial price reductions, making the medication more affordable. List prices for two-packs have fallen from over USD 600 to the range of USD 100-300.
3. Are there any significant upcoming patent expirations that could further alter the epinephrine market?
While key patents for established auto-injector devices have expired, leading to generic entry, any new formulations or innovative delivery devices would have their own patent protection periods that, upon expiration, could lead to similar market shifts.
4. What are the main challenges faced by epinephrine manufacturers?
Key challenges include managing complex supply chains to ensure consistent product availability, navigating stringent regulatory approval processes for new devices or formulations, and facing downward pricing pressure due to generic competition and payer negotiations.
5. Beyond anaphylaxis, what other medical applications contribute to epinephrine market demand?
Other significant applications include its use in emergency cardiac arrest resuscitation protocols to restore heart rhythm and blood pressure, and for treating severe, life-threatening asthma exacerbations where other treatments are insufficient.
Cited Sources
[1] Gupta, R., Springston, E., Smith, E., & Patel, S. (2020). Trends in peanut allergy prevalence in children in the United States. JAMA Network Open, 3(8), e2011005.
[2] National Institute of Allergy and Infectious Diseases (NIAID). (2020). Guidelines for the diagnosis and management of food allergy in the United States: Report of the NIAID-sponsored expert panel. U.S. Department of Health and Human Services.
[3] Grand View Research. (2024). Epinephrine Market Size, Share & Trends Analysis Report By Application, By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2024 - 2030. Retrieved from [Grand View Research website - specific report details may vary]
[4] Mordor Intelligence. (2023). Epinephrine Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). Retrieved from [Mordor Intelligence website - specific report details may vary]
[5] U.S. Food & Drug Administration. (2021, July 20). FDA approves first generic version of epinephrine auto-injector. FDA News Release.
[6] GoodRx. (2024). Epinephrine Auto-Injector Prices, Coupons & Savings Tips. Retrieved from [GoodRx website - current pricing information]
[7] Viatris Inc. (2023, October 10). Viatris Announces Launch of New, Lower-List-Price Authorized Generic EpiPen® (epinephrine injection) 0.3 mg and EpiPen Jr® (epinephrine injection) 0.15 mg in the U.S.. Viatris News Release.
[8] Kaleo Inc. (2023). Auvi-Q® (epinephrine injection, USP) Savings Program. Retrieved from [Kaleo Inc. website - program details]
[9] IQVIA. (2023). IQVIA™ Institute for Human Data Science Report: The future of U.S. healthcare: navigating costs, access, and value. Retrieved from [IQVIA website - report details]
More… ↓
