Drug Price Trends for EPIDUO FORTE
✉ Email this page to a colleague
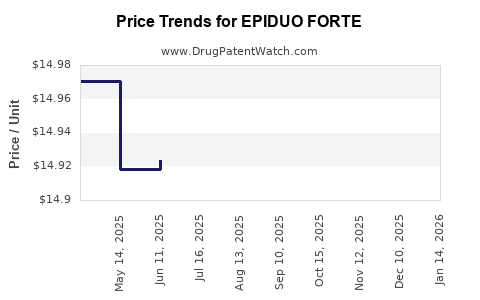
Average Pharmacy Cost for EPIDUO FORTE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EPIDUO FORTE 0.3-2.5% GEL PUMP | 00299-5906-45 | 14.95267 | GM | 2026-04-22 |
| EPIDUO FORTE 0.3-2.5% GEL PUMP | 00299-5906-45 | 14.95267 | GM | 2026-03-18 |
| EPIDUO FORTE 0.3-2.5% GEL PUMP | 00299-5906-45 | 14.93187 | GM | 2026-02-18 |
| EPIDUO FORTE 0.3-2.5% GEL PUMP | 00299-5906-45 | 14.93187 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Summary:
Epiduo Forte (adapalene 0.3% and benzoyl peroxide 2.5%) is a topical treatment approved for acne vulgaris. Its market faces growing demand driven by increasing acne prevalence and adoption of combination therapies. Currently, the drug's pricing reflects its positioning as an advanced acne treatment, with price projections indicating gradual increases aligned with inflation, market penetration, and competitive dynamics.
Market Overview
Product Description:
Epiduo Forte combines a retinoid (adapalene) with benzoyl peroxide. It is marketed primarily for moderate to severe acne and competes with other topical and systemic therapies. The formulation has a higher adapalene concentration compared to Epiduo, targeting users with more resistant acne.
Therapeutic Class & Indications:
Developed as a combination therapy targeting acne vulgaris for teenagers and adults. It is classified as a prescription topical agent, used as part of a comprehensive treatment plan.
Market Size & Growth:
Global acne treatment market was valued at approximately USD 4.7 billion in 2022. It is projected to grow at a Compound Annual Growth Rate (CAGR) of about 4-5% through 2030, driven by rising acne incidence and demand for topical therapies.
Competitive Landscape:
Markets feature other topical treatments such as clindamycin benzoyl peroxide (Epiduo), tretinoin, as well as systemic treatments like oral antibiotics and isotretinoin. New entrants include novel formulations and formulations with improved tolerability.
Distribution & Prescriber Behavior:
Most prescriptions originate from dermatologists, with a rising trend towards teledermatology. Retail pharmacies account for the majority of distribution.
Price Analysis
Current Pricing (as of 2023):
- Epiduo Forte (30g tube): USD 100–125 for the prescription supply, varying by region.
- Per gram cost: approximately USD 3.33–4.17.
- Price per treatment course (assuming once-daily application): roughly USD 90–120, based on average dosage duration.
Historical Trends:
Pricing has remained relatively stable from 2020–2022, with inflation adjustments. Price increases in specific markets reflect regulatory changes and new formulary rankings.
Pricing Drivers:
- Market penetration strategies, including promotional activities.
- Introduction of generic formulations, which are currently absent but expected within 5 years.
- Insurance coverage and formulary inclusion impact patient out-of-pocket costs.
Regional Variations:
- United States: USD 100–125 per tube.
- European Union: EUR 85–115 (~USD 90–122).
- Asian markets: Typically lower, USD 50–80 (~EUR 45–73).
Price Projections (2024–2030):
| Year | Estimated Price per 30g Tube | Key Factors |
|---|---|---|
| 2024 | USD 105–130 | Inflation, increased market penetration |
| 2025 | USD 110–135 | Slight price increase, competitive pressures |
| 2026 | USD 115–140 | Potential formulation innovations |
| 2027 | USD 120–145 | Entry of generics expected |
| 2028 | USD 125–150 | Increased competition from generics and biosimilars |
| 2029 | USD 130–155 | Market saturation, premium branding factors |
| 2030 | USD 135–160 | Standard inflation adjustments, patent expiry approaching |
Note: These estimates assume stable market conditions, no disruptive patent litigation, and steady prescribing patterns.
Key Market Dynamics & Risks
- Patent & Exclusivity: Epiduo Forte's patent protection extends into the late 2020s, with generic entry anticipated around 2027–2028.
- Regulatory Changes: Pricing reforms, especially in the US, may influence margins.
- Market Penetration: Expanding use among adult populations and off-label applications can increase volume.
- Competitive Innovations: New formulations with improved tolerability might pressure pricing.
Key Takeaways:
- Epiduo Forte’s current price range is USD 90–125 per tube, with regional variations.
- Demand driven by acne prevalence, especially in adolescents and young adults, supports steady growth.
- Price projections suggest moderate increases, influenced by inflation, competition, and eventual generic entry.
- The landscape is sensitive to regulatory changes, formulary inclusion, and development of superior competing therapies.
FAQs
More… ↓
