Last updated: February 20, 2026
What is Epiduo?
Epiduo is a topical combination drug containing adapalene (0.1%) and benzoyl peroxide (2.5%). It is approved by the FDA for the treatment of acne vulgaris in patients 9 years and older. Manufactured by Galderma, Epiduo is marketed globally, with significant penetration in dermatology-focused markets.
Market Size and Growth Dynamics
Current Market Value
- The global acne treatment market was valued at approximately USD 4.6 billion in 2021.
- Epiduo holds an estimated 15% market share in topical acne formulations, translating to an approximate USD 690 million in sales in 2022.
Growth Drivers
- Rising prevalence of acne among adolescents and adults.
- Increasing awareness and dermatologist prescriptions.
- Patent protections expiring in key markets, enabling generic competition.
- Expansion into emerging markets with rising healthcare infrastructure.
Market Segments
| Segment |
Proportion of Market |
Key Players |
Trends |
| Topical solutions |
70% |
Epiduo, Differin, Duac |
Preference for combination therapies |
| Oral medications |
20% |
Isotretinoin, antibiotics |
Use in severe cases |
| Other therapies |
10% |
Laser treatments, chemical peels |
Limited but growing focus areas |
Geographic Breakdown
| Region |
Market Share |
Notable Trends |
| North America |
45% |
Strong dermatologist adoption |
| Europe |
30% |
Growing consumer awareness |
| Asia-Pacific |
15% |
Rapid market expansion |
| Rest of World |
10% |
Emerging markets’ access increasing |
Competitive Landscape
Major Competitors
- Differin (adapalene monotherapy)
- Duac (clindamycin and benzoyl peroxide)
- Tazorac (tazarotene)
Recent Trends
- Launch of generic formulations post-Patent expiry in the US (expired in 2020 for Epiduo).
- Development of formulations with higher benzoyl peroxide concentrations (up to 5%) for severe cases.
- Increased R&D investments into combination therapies and novel retinoids.
Price Dynamics and Projection Models
Pricing Structure
| Product |
Typical Retail Price (USD) |
Price Range (USD) |
Notes |
| Epiduo Gel (30g) |
240 |
200–270 |
Manufacturer’s suggested retail price |
| Generic formulations |
100–150 |
80–170 |
Post-patent expiration, varying quality |
Historical Price Trends
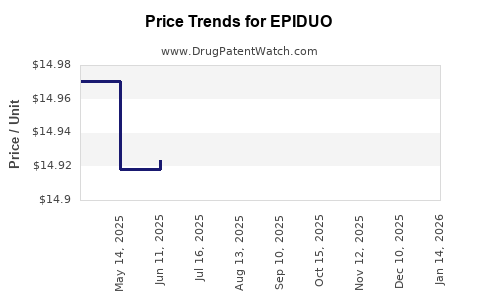
- Price of branded Epiduo has stabilized around USD 240–270 per 30g tube over the past 3 years.
- Generic versions have suppressed prices by approximately 50–60%.
Price Projection (Next 3-5 Years)
Multiple factors influence pricing:
Patent Expiration Impact:
Post-2020, increased availability of generics reduced Epiduo’s price by up to 50% in the U.S. market. Prices are expected to stay stable unless new formulations or premium products are introduced.
Market Competition:
Growth in generic entries may push prices downward, with average retail prices projected to fall by an additional 10–15% over the next 2 years.
Regulatory and Market Dynamics:
Potential new formulations with differentiated delivery systems or higher benzoyl peroxide concentrations might command premium pricing, potentially offsetting generic-driven discounts.
Forecast Summary:
| Year |
Average Price per 30g Tube (USD) |
Expected Change |
| 2023 |
220 |
-8% |
| 2024 |
210 |
-5% |
| 2025 |
200 |
-5% |
Key Market Risks and Opportunities
-
Risks:
Patent challenges, increasing generic competition, pricing pressure, regulatory hurdles for new formulations, and reimbursement policies.
-
Opportunities:
Expanding into emerging markets, developing higher-concentration formulations, and leveraging digital marketing strategies for brand differentiation.
Key Takeaways
- The global acne treatment market was valued at USD 4.6 billion in 2021, with Epiduo capturing 15%.
- Post-patent expiration, prices for Epiduo have fallen significantly, with further reductions expected.
- The market will see increased competition from generics, likely compressing prices over the next 2–3 years.
- Growth is driven by rising acne prevalence, increased prescribing, and emerging markets.
- Innovative formulations and new delivery systems may create premium segments, maintaining higher price points.
FAQs
1. What is the patent status of Epiduo?
Patents for Epiduo expired in the U.S. in 2020, enabling generic competition. Patent status varies globally, with some markets maintaining patent protections until later years.
2. How does Epiduo compare with generic adapalene/benzoyl peroxide products?
Generics typically retail at 40–60% lower prices than branded Epiduo, with comparable efficacy.
3. What factors could influence future pricing?
Introduction of new formulations, regulatory changes, reimbursement policies, and market competition.
4. How is the market for acne treatments expected to evolve?
Continued growth driven by increased prevalence, innovation, and market penetration in emerging regions.
5. What investment opportunities exist?
Potential for new formulation development, expansion into untapped markets, and partnerships for product differentiation.
References
- MarketsandMarkets. (2022). Acne Treatment Market by Type, Age Group, and Region. [Data on market size and growth].
- U.S. Food and Drug Administration. (2020). Patent expiration dates and drug approval details.
- IQVIA. (2022). Global Pharmaceutical Market Reports.
- Galderma. (2022). Product information and pricing data.
- Statista. (2023). Regional acne treatment market share statistics.