Share This Page
Drug Price Trends for ENDOMETRIN
✉ Email this page to a colleague
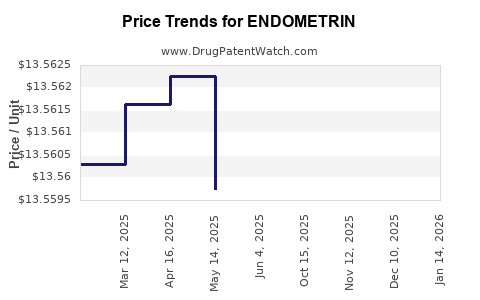
Average Pharmacy Cost for ENDOMETRIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-03 | 14.87384 | EACH | 2026-01-09 |
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-02 | 14.87384 | EACH | 2026-01-09 |
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-01 | 14.87384 | EACH | 2026-01-09 |
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-03 | 14.07175 | EACH | 2025-12-17 |
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-01 | 14.07175 | EACH | 2025-12-17 |
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-02 | 14.07175 | EACH | 2025-12-17 |
| ENDOMETRIN 100 MG VAG INSERT | 55566-6500-02 | 14.09632 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ENDOMETRIN Market Analysis and Price Projections
ENDOMETRIN (progesterone), a prescription medication used in assisted reproductive technology (ART) cycles, faces a dynamic market shaped by evolving regulatory landscapes, competitive pressures, and growing demand for fertility treatments. This analysis provides market insights and price projections for ENDOMETRIN.
What is the Current Market Size and Growth Trajectory for ENDOMETRIN?
The global market for drugs used in ART, including progesterone supplements like ENDOMETRIN, is projected to experience sustained growth. This expansion is driven by increasing rates of infertility worldwide, advancements in ART procedures, and growing patient access to fertility treatments.
The specific market size for ENDOMETRIN is influenced by its adoption rate in various ART protocols and its competitive positioning against other progesterone formulations. While precise figures for ENDOMETRIN's standalone market share are proprietary, the broader progesterone market within ART is estimated to be in the hundreds of millions of U.S. dollars annually. Growth rates for this segment are generally projected to be in the high single digits to low double digits, mirroring the overall ART market expansion.
Key factors contributing to market growth include:
- Rising Infertility Rates: Global infertility rates have been increasing, with estimates suggesting that 1 in 6 couples experience infertility [1]. This demographic trend directly translates to a higher demand for ART services and the associated medications.
- Technological Advancements in ART: Innovations in IVF, such as intracytoplasmic sperm injection (ICSI), preimplantation genetic testing (PGT), and improved embryo cryopreservation, have increased success rates and broadened the patient pool seeking treatment.
- Expanding Access to Fertility Treatments: In some regions, insurance coverage for ART is improving, and government initiatives are supporting fertility care, thereby increasing affordability and accessibility.
- Growing Awareness and Reduced Stigma: Increased public awareness and a reduction in the stigma associated with seeking fertility treatment have encouraged more individuals and couples to pursue ART.
Who are ENDOMETRIN's Primary Competitors?
ENDOMETRIN competes with other progesterone formulations used for luteal phase support in ART cycles. These include:
- Vaginal Gels: Other brand-name and generic vaginal progesterone gels. These share the same route of administration and mechanism of action as ENDOMETRIN, making them direct substitutes in many treatment protocols.
- Intramuscular Injections: Progesterone in oil solutions administered via intramuscular injection. While effective, these can be associated with pain, injection site reactions, and require administration by a healthcare professional or patient self-injection training.
- Vaginal Inserts/Suppositories: Other forms of vaginal progesterone, such as suppositories, offer an alternative delivery method.
- Oral Progesterone: While less commonly used for luteal phase support in ART due to potential side effects and variable bioavailability, oral formulations are also available.
The competitive landscape is further influenced by:
- Generic Competition: The availability of generic versions of progesterone products can drive down prices and impact the market share of branded products like ENDOMETRIN.
- Formulation Preferences: Clinicians and patients may have preferences for specific formulations based on ease of use, side effect profiles, and perceived efficacy.
- Cost-Effectiveness: Payers and healthcare systems increasingly scrutinize the cost-effectiveness of ART medications, favoring options that offer comparable efficacy at a lower price point.
What is the Patent Status and Exclusivity for ENDOMETRIN?
ENDOMETRIN, originally developed by Preglem (now part of Gedeon Richter), has benefited from patent protection. The precise status of its core patents and their expiration dates are critical for understanding future market dynamics and the potential for generic entry.
Information regarding specific patent numbers, their filing dates, and expiration dates for ENDOMETRIN is typically found in public patent databases such as the United States Patent and Trademark Office (USPTO) or the European Patent Office (EPO). These patents would generally cover the active pharmaceutical ingredient (API), specific formulations, methods of use, or manufacturing processes.
- Orphan Drug Exclusivity: For certain indications, drugs can receive Orphan Drug Designation, granting a period of market exclusivity independent of patent life. The eligibility for such designations depends on the rarity of the disease or condition. The primary indication for ENDOMETRIN (luteal phase support in ART) is not typically considered a rare disease.
- Data Exclusivity: Regulatory agencies like the U.S. Food and Drug Administration (FDA) grant periods of data exclusivity upon drug approval, preventing generic manufacturers from relying on the innovator's clinical trial data for their applications. The duration of this exclusivity varies by jurisdiction and is distinct from patent protection.
- Patent Expiration: The expiration of key patents is a significant event. Once primary patents expire and any remaining regulatory exclusivities lapse, generic manufacturers can seek approval to market their versions of the drug. This typically leads to a substantial decrease in price and a shift in market share.
Analyzing the patent landscape for ENDOMETRIN involves tracking the expiry of core composition-of-matter patents, formulation patents, and method-of-use patents. The strategic decisions of generic manufacturers to challenge existing patents or await their expiration also play a crucial role.
What are the Current Pricing and Reimbursement Policies for ENDOMETRIN?
ENDOMETRIN is typically prescribed by fertility clinics and dispensed through specialty pharmacies. Pricing is influenced by several factors:
- Manufacturer's Pricing Strategy: The list price set by the manufacturer, Gedeon Richter, is the starting point.
- Pharmacy Markups: Retail or specialty pharmacies add their dispensing fees and markups.
- Insurance Coverage and Co-pays: The extent to which private health insurance plans or government programs cover ENDOMETRIN significantly impacts out-of-pocket costs for patients. Coverage varies widely by plan and employer. Many fertility treatments, including medications, are not universally covered by insurance.
- Rebate Programs and Discounts: Manufacturers may offer patient assistance programs, co-pay cards, or discounts to mitigate costs for eligible patients.
- Competitive Pricing: The pricing of competing progesterone products, both branded and generic, influences ENDOMETRIN's market position.
Reimbursement Policies:
Reimbursement for ENDOMETRIN is highly variable.
- Limited Insurance Coverage: A significant portion of ART costs, including medications, may not be covered by standard health insurance plans in many countries, including the U.S. This often means patients bear a substantial financial burden.
- State Mandates: A few U.S. states have mandates requiring insurance coverage for certain ART services, which can indirectly impact medication coverage.
- Specialty Pharmacy Networks: Some insurance plans direct patients to use specific specialty pharmacies for ART medications, which can influence pricing and dispensing practices.
- Prior Authorization: Insurers often require prior authorization for high-cost medications like those used in ART, necessitating clinical justification from the prescribing physician.
Average Wholesale Price (AWP) and Estimated Patient Out-of-Pocket Costs:
The AWP for a typical course of ENDOMETRIN can range from $300 to $800 or more, depending on the dosage and duration of treatment prescribed for an ART cycle.
- Without Insurance: Patients without adequate insurance coverage could face the full AWP, potentially exceeding $1,000 for a single ART cycle.
- With Insurance: For patients with insurance that covers ART medications, co-pays can range from $20 to $100 per prescription or a percentage of the cost, depending on the plan’s structure. However, coverage is often limited in quantity or duration.
- Patient Assistance Programs: These programs can reduce out-of-pocket costs to $0 to $50 per prescription for eligible patients, but eligibility criteria can be strict.
These figures are estimates and can fluctuate based on pharmacy, geographic location, and specific insurance plan terms.
What are the Projected Price Trends for ENDOMETRIN?
Projected price trends for ENDOMETRIN are influenced by a confluence of factors, primarily the impending or actual entry of generic alternatives and the ongoing evolution of ART treatment protocols.
Factors Influencing Future Pricing:
-
Generic Competition: This is the most significant driver of price reduction. Once key patents expire and regulatory exclusivities lapse, generic manufacturers can introduce bioequivalent versions of ENDOMETRIN.
- Impact: The introduction of generics typically leads to a 30% to 70% decrease in the price of the originator product within 1-2 years. Competition among multiple generic manufacturers can accelerate this price erosion.
- Timeline: The exact timing of generic entry depends on specific patent expirations and any legal challenges. Without definitive knowledge of specific patent expiry dates, projecting an exact year is speculative, but for many innovator drugs, this period typically begins 10-15 years post-launch or upon patent expiry.
-
Continued Demand for ART: Sustained or increasing demand for fertility treatments will provide a baseline for market activity, even with price reductions due to generics. This ensures a continued market for progesterone support.
-
Innovations in ART and Progesterone Delivery:
- New Formulations: Development of novel progesterone formulations (e.g., extended-release, improved absorption profiles) could introduce new branded products that compete with or supersede current options, potentially commanding premium pricing initially.
- Treatment Protocol Evolution: Shifts in ART protocols that might reduce the need for luteal phase support or favor alternative agents could impact demand for ENDOMETRIN.
-
Reimbursement and Payer Negotiations: As healthcare systems focus on cost containment, payers will likely exert greater pressure on manufacturers and pharmacies to negotiate lower prices. This can include formulary restrictions and preferred provider arrangements favoring lower-cost alternatives, including generics.
-
Manufacturer Strategies: The originator manufacturer may employ strategies such as authorized generics or lifecycle management (e.g., developing new indications or formulations) to mitigate the impact of generic competition, though these strategies are often limited.
Price Projections:
- Short-Term (1-3 years): If ENDOMETRIN has not yet faced significant generic competition, prices are likely to remain relatively stable, subject to minor annual increases driven by inflation and minor market dynamics. However, if a generic entrant emerges within this period, expect a sharp price decline of 30-50% for the branded product as market share erodes.
- Medium-Term (3-7 years): With established generic competition, ENDOMETRIN's price (both branded and generic) will stabilize at a significantly lower level. Branded ENDOMETRIN prices are likely to fall by 50-70% from their peak, while generic prices will dictate the market rate, potentially around $150-$300 per treatment course.
- Long-Term (7+ years): Prices will likely continue to be dictated by generic competition and the availability of newer, potentially more advanced progesterone therapies. Expect prices to remain at or below the lower end of the medium-term projection, $150-$250 per treatment course, as the market matures.
Table 1: Projected Price Trends for ENDOMETRIN (Estimates)
| Timeframe | Scenario | Estimated Price Range (per ART Cycle) | Key Drivers |
|---|---|---|---|
| Current | Branded product, limited generic presence | $300 - $800 | Manufacturer pricing, pharmacy markups, limited insurance coverage |
| Short-Term | Emergence of generic competition | $150 - $400 | Generic entry, price erosion of originator, payer negotiations |
| No significant generic competition | $320 - $850 | Stable demand, minor inflation | |
| Medium-Term | Established generic competition | $150 - $300 | Mature generic market, multiple generic suppliers, continued demand |
| Long-Term | Mature market, potential for next-generation products | $150 - $250 | Price competition among generics, emergence of alternative therapies |
Note: Prices are estimates and do not include insurance co-pays or patient assistance program impacts.
What are the Regulatory Considerations Affecting ENDOMETRIN's Market?
Regulatory bodies play a crucial role in ENDOMETRIN's market access, approval, and pricing. Key considerations include:
- FDA/EMA Approval and Labeling: The approval status for ENDOMETRIN in key markets (e.g., U.S., EU) dictates its availability. Labeling requirements, including indications, contraindications, and warnings, influence prescribing patterns and physician confidence.
- Generic Drug Approval Pathways: Regulatory agencies have established Abbreviated New Drug Application (ANDA) pathways in the U.S. and similar processes in other regions for generic drug approval. These pathways require bioequivalence studies, demonstrating that the generic product performs comparably to the branded product.
- Post-Market Surveillance: Regulatory agencies monitor drug safety and efficacy after approval. Any new safety signals or post-market studies could lead to label changes, restricted use, or even withdrawal from the market, impacting demand and pricing.
- Good Manufacturing Practices (GMP): Manufacturers of both branded and generic ENDOMETRIN must adhere to strict GMP regulations to ensure product quality, safety, and consistency. Regulatory inspections and audits are part of this oversight.
- Orphan Drug Exclusivity and Other Exclusivities: As mentioned earlier, specific designations can grant market exclusivity beyond patent terms, influencing the timing of generic entry.
- Pricing Regulations (Limited in U.S./EU): While direct price controls are less common in the U.S. and EU for branded pharmaceuticals, regulatory bodies can influence market access and reimbursement through formulary decisions, health technology assessments, and negotiations with national health services in some European countries.
Key Takeaways
- ENDOMETRIN operates within a growing ART market driven by rising infertility rates and technological advancements.
- Key competitors include other vaginal progesterone gels, intramuscular injections, and suppositories, with generic competition being the primary threat to branded product pricing.
- Patent expiration is the most critical factor for future price erosion. Precise patent expiry dates are paramount for accurate forecasting.
- Current pricing is substantial, with significant out-of-pocket expenses for patients due to variable insurance coverage.
- Projected price trends indicate a sharp decline upon generic entry, with prices stabilizing at significantly lower levels in the medium to long term.
- Regulatory approvals, generic pathways, and post-market surveillance are critical for market access and product lifecycle management.
Frequently Asked Questions
- When is ENDOMETRIN expected to face its first major generic competition in the United States? The exact timing depends on the expiration of all relevant U.S. patents and any potential patent litigation outcomes. Without specific patent expiry data, this timeframe is speculative but typically follows a 10-15 year lifecycle for innovator drugs.
- How do insurance providers typically categorize ENDOMETRIN for coverage decisions? Insurers often classify ENDOMETRIN as a specialty pharmaceutical or fertility medication. Coverage varies widely, with many plans requiring prior authorization and imposing co-pays or deductibles, while some may not cover it at all.
- What is the primary mechanism by which generic progesterone products will impact ENDOMETRIN's price? Generic products will offer a bioequivalent alternative at a lower manufacturing cost, forcing the branded ENDOMETRIN to reduce its price to remain competitive or lose market share to the lower-cost generics.
- Are there any alternative progesterone formulations being developed that could disrupt ENDOMETRIN's market position? Research and development in ART medications are ongoing. Potential disruptions could arise from new formulations with improved efficacy, reduced side effects, or more convenient administration routes, though ENDOMETRIN's established use provides a strong incumbent advantage.
- What is the typical duration of a course of ENDOMETRIN treatment within a standard IVF cycle, and how does this affect overall treatment cost? A typical course of ENDOMETRIN in an IVF cycle usually spans from the start of stimulation to the pregnancy test, often lasting 10-14 days or longer if pregnancy is achieved, significantly contributing to the total cost of an ART cycle.
Citations
[1] Practice Committee of the American Society for Reproductive Medicine. (2021). Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertility and Sterility, 116(4), 855-863.
More… ↓
