Share This Page
Drug Price Trends for EMGALITY
✉ Email this page to a colleague
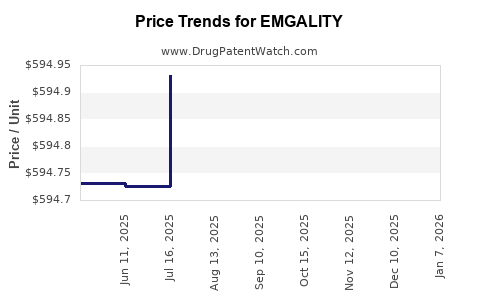
Average Pharmacy Cost for EMGALITY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EMGALITY 120 MG/ML SYRINGE | 00002-2377-11 | 734.85063 | ML | 2026-01-01 |
| EMGALITY 120 MG/ML SYRINGE | 00002-2377-01 | 734.85063 | ML | 2026-01-01 |
| EMGALITY 300 MG DOSE (100 MG/ML X 3 SYRINGES) | 00002-3115-09 | 612.14130 | ML | 2026-01-01 |
| EMGALITY 120 MG/ML PEN | 00002-1436-01 | 735.20572 | ML | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for EMGALITY (Erenumab)
What is EMGALITY and its Current Market Position?
EMGALITY (erenumab) is a calcitonin gene-related peptide (CGRP) receptor antagonist approved for migraine prevention in adults. Launched by Amgen in 2018 and co-marketed with Novartis, it is among the first CGRP-targeted therapies to reach the market. Its key competitors include Fremanezumab (Ajovy) and Eptinezumab (Vyepti), which target CGRP pathways similarly.
Market Size and Growth Trends
The global migraine market was valued at approximately $4.4 billion in 2022, with a compound annual growth rate (CAGR) of 5% projected until 2030. The segment for preventive migraine therapies accounts for roughly 60% of this, driven by increasing diagnosis rates and favorable reimbursement schemes.
Key Drivers
- Rising prevalence of migraine: Estimated 1 billion worldwide.
- Growing awareness and improved diagnosis.
- Reimbursement strategies favoring new biologics.
- Patent exclusivity until 2028-2030, delaying generic competition.
Market Share
In 2022, EMGALITY captured around 20-25% of the CGRP class, with companies like Amgen and Novartis prioritizing its expansion. The drug's sales reached approximately $400 million in 2022, with a trajectory expected to surpass $1 billion by 2028 if market conditions remain stable.
Price Structure and Reimbursement Landscape
Current Pricing
- List price (U.S.): Approximately $575 per injection.
- Dosing: Monthly, with some guidelines allowing for quarterly injections.
- Annual treatment cost: Roughly $6,900 for a standard year.
Reimbursement Policies
- Generally covered by major commercial insurers and Medicaid/Medicare plans.
- Prior authorization processes can affect patient access.
- Price negotiations and discounts vary across markets and payors.
Pricing in International Markets
In Europe, prices range from €450 to €650 per injection, influenced by national healthcare systems and pricing controls. Generic availability remains unlikely before 2028-2030 due to patent protections.
Price Projections and Market Dynamics
Short-term (2023-2025)
- Prices are expected to stabilize amid ongoing reimbursement negotiations.
- No significant price reduction predicted unless biosimilar competition emerges.
- Revenue growth hinges on increased adoption and market penetration.
Medium to Long-term (2026-2030)
- Patent expiration forecast for 2028-2030.
- When biosimilars or generics enter, prices could decline by 30-50%.
- Innovation or new delivery methods may sustain premium pricing.
Factors Influencing Pricing
- Patent expiries lead to price erosion.
- Regulatory developments affecting approval timelines.
- Competitive entries, especially biosimilars.
- Changes in healthcare policies and reimbursement models.
Competitor Pricing Strategies
| Drug | Approximate list price | Dosing Regimen | Market Position |
|---|---|---|---|
| EMGALITY | $575 per injection | Monthly or quarterly | Leading CGRP therapeutic |
| Ajovy (fremanezumab) | $575 per injection | Monthly or quarterly | Similar or lower price |
| Vyepti (eptinezumab) | $385 per injection | Quarterly infusion | Lower price, IV delivery |
Key Takeaways
- EMGALITY commands a premium price (~$575/injection) driven by patent protection and market exclusivity.
- The global migraine prophylaxis market is projected to grow at a 5% CAGR until 2030, reaching over $6 billion.
- Short-term stability in pricing is expected; significant reductions may occur post-2028 with biosimilar entry.
- Reimbursement policies heavily influence patient access and sales volumes.
- Competition from similar biologics and biosimilars remains the primary risk to pricing strategies.
FAQs
How does EMGALITY's pricing compare to other CGRP therapies?
EMGALITY's list price is similar to Ajovy but higher than Vyepti. Its monthly or quarterly dosing provides convenience, justifying premium pricing relative to some competitors.
What factors could lead to a decline in EMGALITY’s price?
Entry of biosimilars post-2028, increased competition, reimbursement pressure, and government price control measures.
Are there any upcoming regulatory hurdles affecting EMGALITY?
While no major regulatory changes are expected before patent expiry in 2028, any new safety concerns or efficacy data could influence its market position.
How does the reimbursement environment vary globally?
Pricing and reimbursement vary widely; Europe employs price controls and negotiations, while the U.S. relies heavily on insurance coverage, affecting net prices.
What is the potential impact of new delivery methods?
Oral or longer-acting formulations could disrupt current dosing regimens, affecting pricing strategies and market share.
References
[1] IQVIA. (2023). Global Migraine Market Report.
[2] Amgen. (2022). EMGALITY product information.
[3] Novartis. (2022). CGRP Therapeutic Portfolio overview.
[4] EvaluatePharma. (2022). World Pharma Market Trends.
[5] OECD. (2021). Pharmaceutical Price Indexes.
More… ↓
