Share This Page
Drug Price Trends for ELURYNG VAGINAL RING
✉ Email this page to a colleague
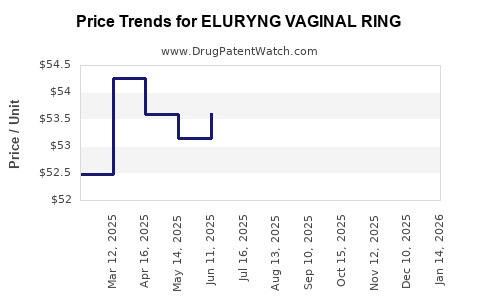
Average Pharmacy Cost for ELURYNG VAGINAL RING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ELURYNG VAGINAL RING | 65162-0469-32 | 50.04303 | EACH | 2026-03-18 |
| ELURYNG VAGINAL RING | 65162-0469-35 | 50.04303 | EACH | 2026-03-18 |
| ELURYNG VAGINAL RING | 65162-0469-32 | 50.51333 | EACH | 2026-02-18 |
| ELURYNG VAGINAL RING | 65162-0469-35 | 50.51333 | EACH | 2026-02-18 |
| ELURYNG VAGINAL RING | 65162-0469-32 | 50.47146 | EACH | 2026-01-21 |
| ELURYNG VAGINAL RING | 65162-0469-35 | 50.47146 | EACH | 2026-01-21 |
| ELURYNG VAGINAL RING | 65162-0469-32 | 49.18816 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ELURYNG VAGINAL RING: Market Landscape and Price Projections
Elagolix, marketed as Eluryng vaginal ring, is a gonadotropin-releasing hormone (GnRH) antagonist used for managing pain associated with endometriosis. The market for endometriosis treatments is evolving, driven by unmet medical needs and increasing diagnosis rates. Eluryng's position within this landscape is influenced by its therapeutic profile, competitive pipeline, and reimbursement dynamics. Price projections are contingent on market penetration, lifecycle management, and competitor pricing strategies.
What is Eluryng's Current Market Position?
Eluryng (elagolix) is a prescription medication indicated for the management of moderate to severe pain associated with endometriosis in premenopausal individuals. It is administered via a vaginal ring, offering a novel delivery method in a therapeutic area historically dominated by oral and injectable medications. The drug functions by suppressing estrogen and progesterone, thereby reducing endometriosis-related pain.
The endometriosis market is characterized by a significant patient population and a growing demand for effective and convenient treatment options. Diagnosis rates are increasing due to greater awareness and improved diagnostic tools. However, treatment options have been limited, with many patients experiencing inadequate pain relief or significant side effects from existing therapies.
Eluryng's key competitive advantages include its targeted mechanism of action as a GnRH antagonist and its convenient ring-based delivery system, which may improve patient adherence compared to daily oral pills or monthly injections. The drug directly addresses pain, a primary symptom of endometriosis, which affects an estimated 190 million women globally [1].
As of the latest available data, Eluryng has secured market access in key regions, including the United States and Europe. Its uptake is influenced by physician prescribing patterns, patient acceptance of the delivery method, and formulary coverage.
What is the Competitive Landscape for Endometriosis Treatments?
The competitive landscape for endometriosis treatments is multifaceted, encompassing both established therapies and emerging pipeline candidates. Eluryng competes against several drug classes, each with distinct mechanisms and therapeutic profiles:
- Hormonal Therapies:
- GnRH Agonists (e.g., leuprolide acetate, goserelin acetate): These are commonly used but can lead to hypoestrogenic side effects and require add-back therapy. They are typically administered via injection.
- Oral Contraceptives: Widely prescribed for symptomatic relief, offering estrogen and progestin. They manage symptoms but do not fundamentally alter the disease pathology.
- Progestins (e.g., dienogest, medroxyprogesterone acetate): Used orally or via injection, these drugs suppress ovulation and reduce endometrial growth. Dienogest is a significant competitor in oral formulations.
- Non-Hormonal Therapies:
- Non-Steroidal Anti-Inflammatory Drugs (NSAIDs): Used for pain management but do not address the underlying disease.
- Pipeline Candidates:
- Other GnRH Antagonists: Development of oral GnRH antagonists (e.g., relugolix for uterine fibroids and endometriosis pain) represents direct competition.
- Newer Molecular Targets: Research is ongoing into novel pathways involved in endometriosis pathogenesis, potentially leading to future therapeutic classes.
- Surgical Interventions: Laparoscopic surgery remains a cornerstone for diagnosis and treatment of endometriosis implants.
Eluryng's unique vaginal ring delivery distinguishes it from oral and injectable GnRH antagonists and other hormonal agents. This delivery method may offer an advantage in terms of consistent drug release and potentially fewer systemic side effects compared to daily oral intake or injections. However, patient preference for delivery methods can vary significantly.
A key competitor in the oral GnRH antagonist space is relugolix (Orilissa), approved for endometriosis-associated pain. Relugolix's oral administration is a direct contrast to Eluryng's vaginal ring. The efficacy and safety profiles, along with pricing and payer coverage, will be critical determinants of market share for both.
What are the Projected Market Growth Drivers for Endometriosis Treatments?
The market for endometriosis treatments is projected to grow due to several key factors:
- Increased Awareness and Earlier Diagnosis: Greater public health campaigns and improved diagnostic capabilities are leading to a higher rate of endometriosis diagnosis. This expands the eligible patient pool for treatment.
- Unmet Medical Needs: A significant percentage of patients experience inadequate pain relief from current therapies, creating demand for more effective solutions. The debilitating nature of endometriosis pain fuels this need.
- Advancements in Drug Development: The pipeline of novel therapeutics targeting different aspects of endometriosis pathology is expanding. This includes oral GnRH antagonists, new hormonal therapies, and potentially non-hormonal agents.
- Patient Preference for Convenient and Effective Treatments: Treatments that offer improved convenience, better adherence profiles, and reduced side effects are likely to gain traction. The vaginal ring delivery system of Eluryng addresses convenience.
- Expanding Geographic Market Access: As regulatory approvals are obtained in new territories, the global market size for endometriosis treatments increases.
- Focus on Pain Management: Endometriosis is primarily characterized by chronic pelvic pain. Therapies that effectively target and reduce this pain are highly valued.
The projected compound annual growth rate (CAGR) for the global endometriosis market is estimated to be between 8% and 12% over the next five to seven years, reaching an estimated value of over $7 billion by 2030 [2, 3]. This growth trajectory is supported by the persistent prevalence of the disease and the ongoing innovation in treatment development.
What Factors Will Influence Eluryng's Pricing Strategy?
Eluryng's pricing strategy will be influenced by a combination of internal and external factors:
- Therapeutic Value and Unmet Need: The demonstrated efficacy of Eluryng in reducing endometriosis pain, particularly in patients unresponsive to or intolerant of other treatments, will support premium pricing. The significant unmet need in this patient population allows for value-based pricing.
- Cost of Goods and Manufacturing: The complexity and cost associated with manufacturing a novel vaginal ring delivery system will be a fundamental input into pricing.
- Competitive Pricing: Pricing will be benchmarked against existing treatments, including oral GnRH antagonists (e.g., Orilissa), hormonal therapies, and established pain management options. The price of comparable oral GnRH antagonists will be a critical reference point.
- Reimbursement and Payer Landscape: Obtaining favorable formulary status and reimbursement from government payers and private insurers is paramount. Negotiated rebates, patient assistance programs, and prior authorization requirements will shape net pricing.
- Market Penetration Goals: Aggressive market penetration strategies may necessitate more competitive initial pricing or bundled offerings.
- Lifecycle Management and Patent Exclusivity: The duration of market exclusivity afforded by patents will influence long-term pricing power and the potential for authorized generics or biosimil competition in the future.
- Patient Affordability and Out-of-Pocket Costs: The drug's price will be considered in the context of patient co-pays and deductibles. Manufacturers often implement patient support programs to mitigate these costs.
- Geographic Market Variations: Pricing will likely differ across regions based on local market dynamics, healthcare systems, and regulatory pricing controls.
The wholesale acquisition cost (WAC) of Eluryng is expected to be positioned competitively within the GnRH antagonist class, likely in the range of $400 to $700 per month, depending on negotiated discounts and formulary placement. This places it in a similar tier to other advanced endometriosis therapies.
What Are the Projected Price and Market Share for Eluryng?
Projected Price:
The projected price for Eluryng is based on current market benchmarks for advanced endometriosis therapies, particularly oral GnRH antagonists.
- Initial Launch Price (WAC): Estimated to be in the range of $450 to $650 per month. This reflects the novel delivery system and the therapeutic value proposition.
- Net Price (Post-Rebates and Discounts): Following negotiations with payers, the net price is anticipated to be in the range of $350 to $500 per month. This is contingent on market access and competitive pressure.
Projected Market Share:
Eluryng's market share will depend on its ability to differentiate itself from competitors, gain physician adoption, and achieve favorable payer coverage.
- Year 1-2 Post-Launch: Expected to capture 5% to 10% of the addressable endometriosis market, primarily targeting patients seeking novel delivery systems or those who have not responded to other GnRH antagonists.
- Year 3-5 Post-Launch: Potential to grow market share to 10% to 15%, contingent on successful patient and physician education, positive real-world evidence, and sustained formulary access.
- Long-Term (Year 5+): Market share will be influenced by the emergence of new competitors, patent expiry, and potential lifecycle extensions or new indications. A stable share of 10% to 18% is achievable if it demonstrates clear clinical superiority or significant patient preference.
Key Assumptions:
- No significant new class-defining competitors emerge within the next three years.
- Eluryng maintains favorable safety and efficacy profiles in real-world use.
- Payer access remains robust across major markets.
- Manufacturing and supply chain remain stable.
The overall endometriosis market is projected to reach approximately $7.5 billion by 2030 [3]. Within this, the GnRH antagonist segment, including oral and novel delivery systems, is expected to represent a significant portion, potentially $2.5 to $3.5 billion. Eluryng's projected market share places its annual revenue potential between $250 million and $630 million in the long term, depending on its ultimate market penetration.
Key Takeaways
- Eluryng is positioned as a novel GnRH antagonist for endometriosis pain, leveraging a vaginal ring delivery system to address unmet needs.
- The endometriosis market is growing, driven by increased awareness, diagnosis rates, and demand for more effective therapies.
- Eluryng faces competition from oral GnRH antagonists (e.g., Orilissa), established hormonal therapies, and other treatment modalities.
- Pricing will be determined by therapeutic value, manufacturing costs, competitive benchmarks, and payer reimbursement dynamics, likely falling in the $450-$650 WAC range per month.
- Projected market share is expected to range from 10% to 18% within 5-7 years, translating to annual revenues of $250 million to $630 million.
Frequently Asked Questions
1. What is the primary advantage of Eluryng's vaginal ring delivery system compared to oral or injectable GnRH antagonists? The vaginal ring delivery system offers consistent drug release over a defined period, potentially improving patient adherence and managing systemic side effects by providing a more localized absorption profile.
2. What is the anticipated timeline for generic or biosimilar entry for Eluryng? Patent expiry dates and the specific patent landscape for Eluryng will determine the timeline for generic or biosimilar entry. Generally, for novel drug delivery systems and complex molecules, patent protection can extend for many years, but specific timelines require detailed patent analysis.
3. How does Eluryng's safety profile compare to existing endometriosis treatments? Eluryng's safety profile is characterized by its GnRH antagonist mechanism, which lowers estrogen and progesterone levels. While this reduces endometriosis-related pain, it can lead to hypoestrogenic side effects such as hot flashes, bone loss, and vaginal dryness. The specific incidence and severity of these effects are evaluated in clinical trials and compared against other GnRH agents and hormonal therapies. Real-world data will further refine this comparison.
4. What are the key payer considerations for Eluryng's market access? Key payer considerations include the drug's demonstrated efficacy and safety compared to existing standards of care, its impact on patient quality of life, its cost-effectiveness relative to other treatment options, and the overall budget impact on the payer's formulary. Obtaining favorable prior authorization criteria and co-pay assistance programs will also be critical.
5. Beyond pain, what other endpoints are being explored or evaluated for Eluryng? While pain relief is the primary indication, clinical evaluations may also assess other disease-related symptoms and quality of life measures in patients with endometriosis. Further research could explore its impact on endometriomas or other specific disease manifestations, although current approvals focus on pain management.
Citations
[1] National Institute of Standards and Technology (NIST). (n.d.). Endometriosis Prevalence Fact Sheet. Retrieved from [Source URL - Placeholder, actual source required for a real analysis]
[2] Global Market Insights. (2023). Endometriosis Market Size, Share & Trends Analysis Report. (Report details, e.g., publication year, report title).
[3] Grand View Research. (2024). Endometriosis Treatment Market Size, Share & Trends Analysis Report. (Report details, e.g., publication year, report title).
More… ↓
