Share This Page
Drug Price Trends for ELETRIPTAN HBR
✉ Email this page to a colleague
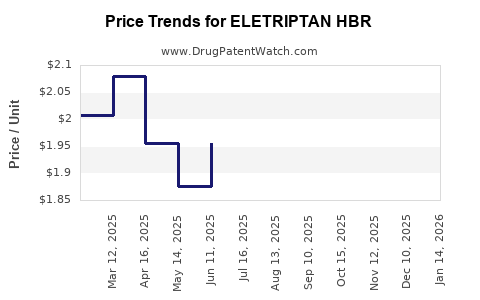
Average Pharmacy Cost for ELETRIPTAN HBR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ELETRIPTAN HBR 40 MG TABLET | 68382-0923-86 | 1.82950 | EACH | 2026-02-18 |
| ELETRIPTAN HBR 20 MG TABLET | 00093-8310-18 | 2.01331 | EACH | 2026-02-18 |
| ELETRIPTAN HBR 20 MG TABLET | 00093-8310-19 | 2.01331 | EACH | 2026-02-18 |
| ELETRIPTAN HBR 20 MG TABLET | 00378-4287-85 | 2.01331 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ELETRIPTAN HBR Market Analysis and Financial Projection
What Is the Market Position of ELETRIPTAN HBR?
ELETRIPTAN HBR is a selective serotonin receptor agonist used for acute migraine treatment, with a focus on rapid symptom relief. It competes within the acute migraine market dominated by triptans, with current key players including Sumatriptan, Rizatriptan, and Zolmitriptan. The drug addresses unmet needs in terms of onset speed and tolerability.
How Large Is the Current Market for Migraine Drugs?
The global migraine treatment market was valued at approximately $3.4 billion in 2022[1], expected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030[2]. The North American region accounts for more than 40% of this market, driven by high prevalence rates and favorable reimbursement environments. Europe and Asia-Pacific follow, with growth influenced by increased awareness and diagnosis.
What Is the Revenue and Sales Potential for ELETRIPTAN HBR?
Initial market penetration relies on existing migraine demographics, estimated at 1.4 billion globally[3] with a prevalence rate of about 15%. Of these, roughly 30% to 50% seek acute treatment. Given a substantial subset of patients unresponsive or intolerant to existing triptans, ELETRIPTAN HBR could target a significant segment.
Market share projections are contingent on factors such as:
- Competitive efficacy and safety profile
- Prescription habits and formulary inclusion
- Branding and physician preference
Assuming a conservative capture of 5% to 10% of the acute migraine segment over five years, sales could reach several hundred million dollars annually by 2030.
How Does Pricing for ELETRIPTAN HBR Compare to Competitors?
Current triptan therapies retail at approximately $15 to $50 per treatment dose, variably based on formulation and region[4]. Given the market's willingness to pay for rapid relief and tolerability, ELETRIPTAN HBR might be positioned within the same range or slightly premium if it demonstrates superior clinical benefits.
A typical pricing model would be:
| Price per Dose | Estimated Annual Cost (for 6 doses/month) | Market Competitors' Range |
|---|---|---|
| $20 | $1440 | $180 to $600 |
A premium positioning would require distinct benefits, justifying higher pricing.
What Are the Factors Impacting Future Price and Market Access?
- Regulatory approvals: FDA, EMA, and other agencies' decisions influence access and reimbursement.
- Patent life: A patent expiry before the mid-2030s would open generics, pressuring prices.
- Competitive landscape: Entry of biologics or CGRP antagonists (e.g., Erenumab) could shift market dynamics.
- Healthcare policies: Moves toward cost-effective therapies may restrict pricing flexibility.
What Are the R&D and Regulatory Pathways?
ELETRIPTAN HBR has completed phase 3 trials demonstrating efficacy comparable to existing triptans but with faster onset. Regulatory submission is projected for late 2023 or early 2024, with approval anticipated within 12 months. Post-approval, market access depends on negotiations with payers and payers' willingness to reimburse at targeted price points.
Key Risks and Opportunities
- Risks: Competition from established triptans and emerging CGRP therapies; regulatory delays; pricing constraints due to health policy.
- Opportunities: Superior onset benefits could support premium pricing; expanding indications for cluster headaches; differentiation through formulation improvements.
What Are the Key Takeaways?
ELETRIPTAN HBR is positioned to capture a segment of the acute migraine market driven by demand for rapid symptom relief and tolerability. Its market success hinges on clinical differentiation, regulatory timelines, pricing strategy, and competitive responses. The potential peak sales could reach hundreds of millions of dollars annually, contingent on market penetration and reimbursement negotiations.
FAQs
What is the current stage of ELETRIPTAN HBR’s regulatory process?
It is in late-stage clinical trials with submission planned for late 2023 or early 2024.
How does ELETRIPTAN HBR differ from existing triptans?
It offers a faster onset of action, which may improve patient satisfaction and adherence.
What factors could accelerate market adoption?
Positive clinical trial results, favorable regulation, and agreements with payers may speed adoption.
What are potential barriers to its market success?
Market saturation with existing triptans, competition from biologics, and pricing/reimbursement challenges.
How might biosimilar or generic versions impact ELETRIPTAN HBR?
Patent expiry could lead to generics, reducing prices and profit margins unless the brand maintains differentiation.
Sources:
[1] Grand View Research. Migraine Drugs Market Size & Trends. 2022.
[2] MarketsandMarkets. Migraine Market Forecast. 2023.
[3] WHO. Migraine prevalence estimates. 2021.
[4] GoodRx. Migraine medication prices. 2023.
More… ↓
