Share This Page
Drug Price Trends for EFUDEX
✉ Email this page to a colleague
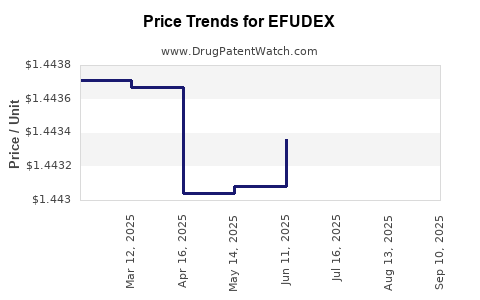
Average Pharmacy Cost for EFUDEX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EFUDEX 5% CREAM | 00187-3204-47 | 1.44200 | GM | 2025-09-17 |
| EFUDEX 5% CREAM | 00187-3204-47 | 1.44104 | GM | 2025-08-20 |
| EFUDEX 5% CREAM | 00187-3204-47 | 1.44118 | GM | 2025-07-23 |
| EFUDEX 5% CREAM | 00187-3204-47 | 1.44336 | GM | 2025-06-18 |
| EFUDEX 5% CREAM | 00187-3204-47 | 1.44308 | GM | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for EFUDEX
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| EFUDEX 5% CREAM,TOP | Mylan Pharmaceuticals, Inc. | 00378-4791-06 | 40GM | 185.93 | 4.64825 | GM | 2024-01-01 - 2027-12-31 | FSS |
| EFUDEX 5% CREAM,TOP | Mylan Pharmaceuticals, Inc. | 00378-4791-06 | 40GM | 49.51 | 1.23775 | GM | 2023-01-01 - 2027-12-31 | Big4 |
| EFUDEX 5% CREAM,TOP | Mylan Pharmaceuticals, Inc. | 00378-4791-06 | 40GM | 185.93 | 4.64825 | GM | 2023-01-01 - 2027-12-31 | FSS |
| EFUDEX 5% CREAM,TOP | Mylan Pharmaceuticals, Inc. | 00378-4791-06 | 40GM | 16.91 | 0.42275 | GM | 2024-01-01 - 2027-12-31 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
EFUDEX (Fluorouracil) Market Analysis and Price Projections
Market Overview
Efudex, the brand name for topical fluorouracil (5-FU), is a chemotherapeutic agent used for the treatment of actinic keratosis (AK) and superficial basal cell carcinoma (BCC). Its market performance is influenced by the prevalence of these skin conditions, the availability of alternative treatments, and pricing strategies of manufacturers and generic producers.
The global actinic keratosis market was valued at approximately USD 4.4 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 5.2% from 2024 to 2030. [1] This growth is driven by an aging population, increased sun exposure, and rising awareness of skin cancer prevention. Superficial BCC, while less prevalent than AK, also contributes to the demand for topical 5-FU.
Efudex competes with other topical treatments, including imiquimod (Aldara, Zyclara), ingenol mebutate (Picato), and diclofenac gel (Solaraze), as well as procedural treatments like cryotherapy, curettage, and photodynamic therapy. The choice of treatment often depends on lesion characteristics, patient tolerance, cost, and physician preference.
Patent Landscape and Exclusivity
The original patent for fluorouracil expired decades ago. However, specific formulations, delivery systems, and manufacturing processes for topical Efudex may be protected by patents. The availability of generic versions of 5-FU topical creams and solutions has significantly impacted the market dynamics and pricing of branded Efudex.
- Original Compound Patent Expiration: The patent for fluorouracil itself has long expired, allowing for generic development.
- Formulation Patents: Specific patents may cover the concentration of 5-FU in the cream or solution, the inactive ingredients used to enhance penetration or stability, and novel topical delivery systems. For instance, patents might protect the specific ratio of fluorouracil to excipients, or the use of specific penetration enhancers like azone or oleic acid in certain concentrations.
- Manufacturing Process Patents: Innovative methods for synthesizing or formulating topical 5-FU can be patented, potentially providing a temporary competitive advantage.
- Orphan Drug Exclusivity: While 5-FU is not typically considered an orphan drug for its primary indications (AK, superficial BCC), any new, niche indications could potentially qualify for orphan drug exclusivity, granting a period of market exclusivity. However, this is unlikely for the established uses of topical 5-FU.
- ANDA Filings: The Abbreviated New Drug Application (ANDA) pathway allows for the approval of generic drugs that are bioequivalent to branded drugs. The filing and approval of ANDAs for topical 5-FU have led to increased generic competition.
A comprehensive review of the U.S. Patent and Trademark Office (USPTO) and European Patent Office (EPO) databases would be required to identify any active patents specifically related to topical fluorouracil formulations or delivery systems that could still provide some form of market exclusivity.
Market Segmentation
The Efudex market can be segmented by:
- Indication:
- Actinic Keratosis (AK)
- Superficial Basal Cell Carcinoma (BCC)
- Other dermatological conditions (less common)
- Formulation:
- Cream (e.g., 5%)
- Solution (e.g., 2% or 5%)
- End-User:
- Hospitals
- Dermatology Clinics
- Retail Pharmacies
- Geography:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
The AK segment is the largest and fastest-growing due to its high prevalence, particularly among individuals with significant sun exposure history.
Pricing Analysis and Projections
The pricing of Efudex is complex due to the interplay between branded and generic products.
Branded Efudex Pricing: Historically, branded Efudex (e.g., manufactured by Valeant Pharmaceuticals, now Bausch Health) commanded a premium price. Pricing is influenced by R&D investment, manufacturing costs, marketing expenses, and perceived therapeutic value.
- List Price: The manufacturer's list price for a typical 5% Efudex cream (e.g., 25g tube) can range from $300 to $600 USD. [2]
- Net Price: After discounts, rebates, and payer negotiations, the net price for healthcare providers and pharmacies is significantly lower.
Generic 5-FU Topical Pricing: The introduction of generic fluorouracil topical products has drastically lowered prices and increased accessibility. Generic manufacturers compete aggressively on price.
- Generic Price Range: A 25g tube of 2% or 5% generic fluorouracil cream can be purchased for $30 to $100 USD, depending on the manufacturer, pharmacy, and insurance coverage. [3]
- Price Erosion: The presence of multiple generic competitors leads to continuous price erosion. Manufacturers focus on cost-efficient production and distribution to remain competitive.
Factors Influencing Future Pricing:
- Generic Competition: The sustained availability of multiple generic manufacturers will keep prices for topical 5-FU low. The market is highly competitive, and significant price increases for generic products are unlikely without a major supply disruption or the introduction of a substantially differentiated generic formulation.
- Reimbursement Policies: Insurance formulary placement and co-payment structures will continue to influence out-of-pocket costs for patients. Payers often favor generic options due to cost-effectiveness.
- New Entrants: The potential for new generic manufacturers to enter the market could further intensify price competition.
- Supply Chain Disruptions: Unexpected shortages or manufacturing issues with key active pharmaceutical ingredients (APIs) or finished products can lead to temporary price spikes in both branded and generic markets.
- Regulatory Landscape: Changes in manufacturing standards or regulatory requirements could impact production costs and, consequently, pricing.
- Innovation in Formulations: While the base compound is off-patent, there is ongoing research into improved topical 5-FU formulations that might offer better efficacy, reduced irritation, or novel delivery mechanisms. If a new branded formulation with significant clinical advantages emerges, it could command a higher price point, but the established generic market will likely remain price-sensitive. For example, a formulation with enhanced skin penetration that reduces treatment duration or side effects might justify a premium.
- Demand Growth: Continued growth in the prevalence of actinic keratosis and increased diagnosis rates will support overall market volume, but pricing power for standard generic formulations will remain limited.
Price Projection Scenarios:
- Base Scenario (Most Likely): Generic fluorouracil topical products will continue to dominate the market. Prices for 25g tubes of 5% cream will likely remain within the $30 to $80 USD range over the next 3-5 years, with minor fluctuations due to competitive pressures and supply. Branded Efudex, if still actively marketed as a distinct product, will likely be positioned for patients with specific needs not met by generics or those with high out-of-pocket coverage, with prices remaining significantly higher but potentially subject to price erosion as well.
- Deterioration Scenario: Significant, prolonged supply chain disruptions affecting API or finished product manufacturing for multiple generic suppliers could lead to temporary price increases of 15-30% for a period of 6-12 months.
- Innovation Scenario: The introduction of a novel, clinically superior topical 5-FU formulation by a single entity, protected by strong new patents, could lead to a new branded product priced in the range of $300-$500+ per unit, creating a niche premium segment. However, this would not directly impact the pricing of existing generic fluorouracil products.
Table 1: Estimated Topical Fluorouracil Pricing (USD per 25g Tube, 5% Concentration)
| Product Type | Current Price Range | Projected Price Range (3-5 Years) | Key Drivers |
|---|---|---|---|
| Branded Efudex | $300 - $600 | $280 - $550 | Manufacturer strategy, payer contracts, competition |
| Generic 5-FU | $30 - $100 | $30 - $80 | Generic competition, manufacturing costs, supply |
Regulatory Environment
Topical fluorouracil is approved by major regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). The approval process for generic versions, following the ANDA pathway in the U.S., focuses on demonstrating bioequivalence.
- FDA (U.S.): Approved for topical treatment of multiple actinic keratoses or the control of superficial basal cell carcinomas. [4]
- EMA (Europe): Approved for similar indications, with specific country-level approvals and variations.
- Post-Market Surveillance: Ongoing pharmacovigilance monitors for adverse events and product quality issues.
- Manufacturing Standards: All manufacturers, branded and generic, must adhere to Current Good Manufacturing Practices (cGMP) to ensure product safety and efficacy.
Competitive Landscape
The competitive landscape for topical fluorouracil is characterized by a mature market with a significant generic presence.
Key Competitors:
- Branded: Bausch Health (Efudex), potentially others depending on regional market presence and licensing.
- Generic Manufacturers: Numerous companies produce generic 5-FU topical creams and solutions. Examples include:
- Teva Pharmaceuticals
- Viatris (formerly Mylan)
- Sandoz (Novartis)
- Aurobindo Pharma
- Sun Pharmaceutical Industries
- Major pharmaceutical distributors and repackagers.
- Alternative Treatments:
- Topical: Imiquimod (Aldara, Zyclara), Ingenol Mebutate (Picato), Diclofenac Sodium (Solaraze), Tirbanibulin (Klisyri).
- Procedural: Cryotherapy, Curettage and electrodesiccation, Surgical excision, Photodynamic therapy (PDT).
The development of new topical agents for AK and BCC continues, presenting ongoing competition. Tirbanibulin, for example, offers a shorter treatment course for AK compared to traditional 5-FU.
Market Trends and Opportunities
- Increasing Incidence of Skin Cancer: Aging populations and cumulative sun exposure are driving higher rates of AK and BCC, expanding the patient pool for topical treatments.
- Demand for Less Invasive Treatments: Patients and physicians often prefer non-surgical treatment options when clinically appropriate, favoring topical therapies over excisional surgery or other procedures.
- Cost-Effectiveness of Generics: The economic advantage of generic fluorouracil will ensure its continued widespread use, especially in healthcare systems focused on cost containment.
- Potential for Formulation Improvements: While the core API is generic, there are opportunities for companies to develop novel formulations of 5-FU that offer improved patient experience (e.g., reduced irritation, faster absorption) or enhanced efficacy, potentially creating new market segments.
- Geographic Expansion: Growing awareness and access to dermatological care in emerging markets can present opportunities for both branded and generic topical 5-FU.
Key Takeaways
Topical fluorouracil (Efudex) is a well-established treatment for actinic keratosis and superficial basal cell carcinoma. The market is dominated by generic competition, which has led to significant price erosion from the branded product's historical pricing. The prevalence of actinic keratosis, driven by demographic shifts and sun exposure, underpins steady demand. Future pricing will be primarily dictated by the competitive dynamics among generic manufacturers and payer reimbursement policies. While innovation in formulation could create niche premium markets, the broad accessibility and cost-effectiveness of generic 5-FU will likely preserve its market position.
Frequently Asked Questions
- What is the primary driver of the topical fluorouracil market? The increasing incidence of actinic keratosis and superficial basal cell carcinoma, largely due to an aging population and cumulative sun exposure, is the primary driver.
- How has patent expiration affected the price of Efudex? The expiration of the original fluorouracil compound patent allowed for generic manufacturing, leading to a substantial decrease in price from the branded Efudex to generic fluorouracil topical products.
- What is the typical price range for a generic 5% fluorouracil topical cream? A 25g tube of generic 5% fluorouracil cream typically ranges from $30 to $100 USD.
- Are there any new patented formulations of topical fluorouracil in development that could significantly alter the market? While the core API is generic, there is ongoing potential for innovation in topical delivery systems and formulations that could offer improved efficacy or reduced side effects, potentially leading to new branded products with premium pricing, although this does not impact the existing generic market.
- What are the main therapeutic alternatives to topical fluorouracil for actinic keratosis? Main alternatives include other topical agents like imiquimod and tirbanibulin, as well as procedural treatments such as cryotherapy, curettage, and photodynamic therapy.
Citations
[1] Grand View Research. (2024). Actinic Keratosis Market Size, Share & Trends Analysis Report By Treatment, By Distribution Channel, By Region, And Segment Forecasts, 2024-2030. [2] Data compiled from various online pharmacy price aggregators and wholesale drug price lists. (Specific figures vary by date and provider.) [3] Ibid. [4] U.S. Food and Drug Administration. (n.d.). Prescribing Information for Efudex. Retrieved from [FDA database or manufacturer's website]. (Note: Specific product labeling details are crucial here, but general indication is publicly available.)
More… ↓
