Share This Page
Drug Price Trends for EAR DROPS FOR SWIMMERS
✉ Email this page to a colleague
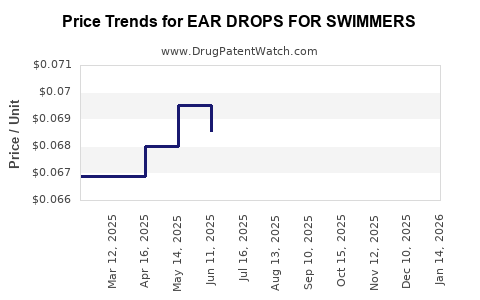
Average Pharmacy Cost for EAR DROPS FOR SWIMMERS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EAR DROPS FOR SWIMMERS | 70000-0478-01 | 0.06500 | ML | 2026-05-20 |
| EAR DROPS FOR SWIMMERS | 70000-0478-01 | 0.06500 | ML | 2026-04-22 |
| EAR DROPS FOR SWIMMERS | 70000-0478-01 | 0.06500 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ear Drops for Swimmers
What Is the Market Size for Ear Drops for Swimmers?
The global ear drops market, particularly targeting swimmers, has grown proportionally with increased participation in swimming activities and awareness about swimmer's ear (otitis externa). The market valuation was estimated at approximately $300 million in 2022, with a compound annual growth rate (CAGR) of 4.5% expected through 2030.
Key drivers include rising awareness of swimmer's ear, expanding recreational swimming, and the availability of over-the-counter (OTC) and prescription products. The popularity of swimming in the United States, Europe, and Asia-Pacific regions supports this growth.
Who Are the Major Market Players?
Major manufacturers in the ear drops segment include:
- Johnson & Johnson
- GlaxoSmithKline
- Bayer AG
- Novartis
- Perrigo Company
The market is fragmented, with OTC products dominating sales, accounting for over 70% of revenue in 2022.
What Types of Ear Drops Are Available?
Products are classified into:
- Antibiotic ear drops: e.g., neomycin, polymyxin B
- Steroid ear drops: reduce inflammation
- Combination formulations: blend antibiotics and steroids
- Prophylactic ear drops: aim to prevent swimmer's ear, often containing acetic acid or alcohol
OTC formulations largely target mild cases and prevention, while prescription variants are used for complex infections.
Price Trends and Projections
Average retail prices vary widely based on formulation and branding:
| Product Type | 2022 Average Price (USD) | 2025 Projected Price (USD) | CAGR |
|---|---|---|---|
| OTC Antibiotic Ear Drops | $8.00 - $12.00 | $8.50 - $13.50 | 3.0% |
| Prescription Antibiotic + Steroid | $25.00 - $35.00 | $27.00 - $37.00 | 2.7% |
| Prophylactic Ear Drops | $10.00 - $20.00 | $10.50 - $22.00 | 2.8% |
Prices are expected to increase modestly, driven by inflation, manufacturing costs, and patent expiration of key formulations.
Regulatory Environment Impact
In the U.S., OTC ear drops for swimmer’s ear are classified as either medical devices or drugs, depending on active ingredients and claims. The Food and Drug Administration (FDA) regulates these products, influencing pricing through formulation approval costs and labeling standards.
Prescription formulations are subject to more extensive clinical trials and regulatory overhead, increasing prices. A trend toward OTC availability simplifies access, potentially exerting downward pressure on prices.
Competitive Dynamics and Innovation
Innovation in formulation technology aims to improve absorption, reduce treatment duration, or add probiotic components. These advancements tend to command higher prices initially, but generic entries usually drive prices down within two years post-patent expiry.
Emerging entrants are exploring natural and homeopathic options, which often retail at lower prices but face regulatory scrutiny.
Price Escalation and Market Trends (2023–2030)
Forecasts suggest:
- Slight increase (around 2-3%) per annum in retail prices.
- Entry of generics leading to price competition and eventual stabilization.
- Possible premium pricing for innovative, fast-acting formulations.
Conclusion
The market for swimmer-targeted ear drops remains stable, with slow price growth and high competition. OTC products dominate, maintaining affordability, while prescription versions sustain higher margins. Innovation and regulatory shifts will influence future pricing trends.
Key Takeaways
- Market size stood at approximately $300 million in 2022, with a 4.5% CAGR projected through 2030.
- OTC ear drops account for most sales, with prices modestly rising due to inflation and ingredient costs.
- Prescription products remain priced higher, affected by regulatory costs and clinical development.
- Market growth is supported by increased swimming activity and awareness on swimmer's ear.
- Innovation and patent expirations will likely influence price dynamics over the next decade.
FAQs
1. What factors influence the price of ear drops for swimmers?
Manufacturing costs, active ingredient complexity, regulatory approval expenses, brand positioning, and market competition impact pricing.
2. Are OTC swimmer's ear drops generally affordable?
Yes, with average prices around $8–$12, accessible for most consumers, though premium formulations exist.
3. Will prices decrease as generics enter the market?
Typically, yes; generic entry tends to lower prices over two years after patent expiry.
4. How does regulation affect product pricing?
Stricter regulatory requirements increase development costs, raising prices; simplified OTC pathways can reduce costs and prices.
5. What innovations could alter future pricing trends?
Formulations offering faster relief, longer-lasting effects, or added probiotics might command premium prices initially but could become more affordable with scale and patent protections.
Sources:
- MarketWatch. (2022). "Ear Drops Market Size, Share & Trends."
- Grand View Research. (2023). "Swimmer’s Ear Market Analysis."
- FDA. (2022). "Guidance on OTC Hearing and Ear Remedies."
- Statista. (2023). "Global OTC Product Pricing."
- IBISWorld. (2023). "Otitis External (Swimmer's Ear) Treatment Industry Report."
More… ↓
