Share This Page
Drug Price Trends for DUTASTERIDE-TAMSULOSIN
✉ Email this page to a colleague
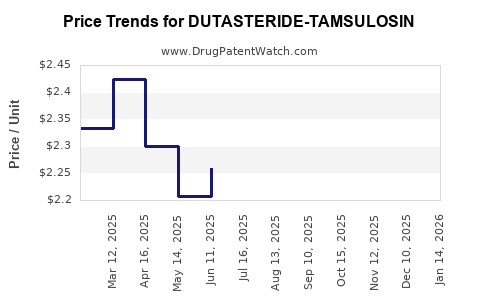
Average Pharmacy Cost for DUTASTERIDE-TAMSULOSIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 59651-0063-90 | 2.01940 | EACH | 2026-03-18 |
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 68382-0640-06 | 2.01940 | EACH | 2026-03-18 |
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 59651-0063-30 | 2.01940 | EACH | 2026-03-18 |
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 68382-0640-16 | 2.01940 | EACH | 2026-03-18 |
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 10370-0280-09 | 2.01940 | EACH | 2026-03-18 |
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 10370-0280-11 | 2.01940 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DUTASTERIDE-TAMSULOSIN Market Analysis and Financial Projection
Market Overview and Demand Analysis for Dutasteride-Tamsulosin
Dutasteride-tamsulosin combines two drugs used in benign prostatic hyperplasia (BPH) treatment: dutasteride, a 5-alpha reductase inhibitor, and tamsulosin, an alpha-1 adrenergic receptor blocker. This combination is prescribed to improve urinary flow and reduce prostate size, especially in moderate to severe cases.
Market demand centers on aging populations, particularly in North America, Europe, and parts of Asia with rising BPH prevalence. The shift toward combination therapies over monotherapy enhances market potential, as physicians target symptomatic relief and prostate size reduction simultaneously.
Current Market Size and Revenue
The global BPH drug market reached approximately USD 4 billion in 2022. Dutasteride-tamsulosin's share is estimated at 15-20%, equating to USD 600-800 million. The drug's approval status varies by region, with the U.S. FDA approving generic versions since 2018 and branded products from AstraZeneca (originally marketed as Avodart). European markets follow similar timelines, with generic entries increasing affordability.
Combining drugs increases adherence, making this therapy attractive. Market penetration depends on patent status, cost, and clinician preference, impacting sales growth trajectories.
Price Dynamics and Projections
Current Pricing Landscape
- Brand-Name Dutasteride (Avodart): Monthly wholesale price approximately USD 80–100.
- Tamsulosin: Monthly wholesale price approximately USD 20–30.
- Combination Therapy: Due to patent expiration, generic versions are now available; retail prices vary but range from USD 40–70 monthly for the combination.
Factors Influencing Pricing
- Generic Competition: Increased market entry offers downward pressure.
- Regulatory Approvals: Gaining approval for fixed-dose combination (FDC) versions boosts marketability.
- Distribution Channels: Hospital and pharmacy procurement costs influence final retail prices.
Price Projection (Next 5 Years)
| Year | Estimated Price Range (USD/month) | Market Penetration Growth | Notes |
|---|---|---|---|
| 2023 | 40–70 | 5% | Post-generic entry stabilization |
| 2024 | 35–65 | 8% | Increased competition reduces prices |
| 2025 | 30–55 | 12% | Wider adoption, more generics available |
| 2026 | 25–50 | 15% | Price normalization expected |
| 2027 | 20–45 | 20% | Penetration peaks as patents expire |
Prices are expected to decline by approximately 50-55% from peak branded levels by 2027 owing to generics and increased market competition.
Competitive Landscape
Major competitors include:
- Brand Drugs: Avodart (AstraZeneca), generic dutasteride.
- Alternatives: Tamsulosin monotherapy, other 5-alpha reductase inhibitors (finasteride).
- Emerging Therapies: Novel alpha-blockers, PDE5 inhibitors, minimally invasive surgical procedures.
Market share shifts toward generic combination drugs are expected. Patent expiry in key markets will accelerate generic entry.
Regulatory & Patent Considerations
- Approved Markets: U.S., Europe, Japan, select Asia-Pacific countries.
- Patent Status: The original patent for Avodart expired in most markets by 2018, encouraging generics.
- Regulatory Approvals for FDC: Some countries require separate approval, impacting launch timelines and pricing.
Investment and R&D Outlook
The pipeline for improved BPH therapies remains active, with research into next-generation alpha-blockers, 5-alpha reductase inhibitors, and combination formulations. Patent expiries will continue to pressure prices but may also offer licensing and partnership opportunities.
Key Takeaways
- The global BPH drug market is circa USD 4 billion, with dutasteride-tamsulosin accounting for 15-20%.
- Market growth depends on aging demographics and increasing preference for combination therapies.
- Prices for the combination are projected to decline from USD 40–70/month in 2023 to USD 20–45/month by 2027 due to generic competition.
- Patent expiries and regulatory approvals will influence pricing dynamics, with generic versions dominating the market.
- Investment in R&D may focus on next-generation therapies or improved formulations to maintain competitive advantage.
FAQs
1. What are the main factors influencing price reductions for dutasteride-tamsulosin?
The primary factors include patent expiries, increased generic market entry, regulatory approvals for fixed-dose combinations, and competitive pricing strategies among manufacturers.
2. How significant is the impact of generic competition on market share?
Generic competition tends to reduce prices significantly—by up to 50-60%—and captures a substantial portion of market share, often above 70% within five years of patent expiry.
3. Are there regional differences in pricing and adoption?
Yes. North America and Europe benefit from early generic entry and broader approval, leading to higher adoption rates and lower prices. In developing markets, prices tend to be higher due to limited competition and regulatory barriers.
4. What upcoming regulatory developments could influence the market?
New approvals for fixed-dose combination formulations, especially in regions where separate approvals are currently required, can accelerate adoption and impact pricing strategies.
5. What is the outlook for R&D within this therapeutic area?
Investments focus on improving efficacy, reducing side effects, and simplifying dosing regimens. Challenges remain in differentiating new products from existing generics, but innovations in drug delivery and combination therapies continue.
Sources:
- MarketWatch. (2022). Benign Prostatic Hyperplasia (BPH) Drugs Market Size.
- GlobalData. (2022). Dutasteride Market Analysis.
- U.S. FDA. (2021). Drug Approvals and Patent Expirations.
- IQVIA. (2022). Pharmacy Price Data.
- AstraZeneca. (2019). Avodart Product Information.
More… ↓
