Share This Page
Drug Price Trends for DUOBRII
✉ Email this page to a colleague
Average Pharmacy Cost for DUOBRII
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
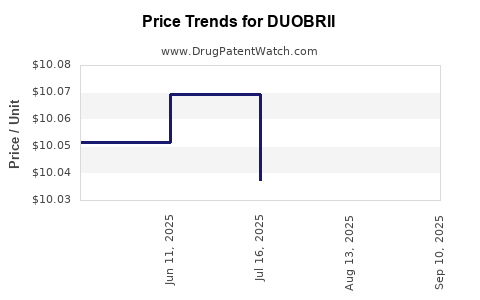
| DUOBRII 0.01%-0.045% LOTION | 00187-0653-01 | 10.00965 | GM | 2025-09-17 |
| DUOBRII 0.01%-0.045% LOTION | 00187-0653-01 | 10.02273 | GM | 2025-08-20 |
| DUOBRII 0.01%-0.045% LOTION | 00187-0653-01 | 10.03730 | GM | 2025-07-23 |
| DUOBRII 0.01%-0.045% LOTION | 00187-0653-01 | 10.06930 | GM | 2025-06-18 |
| DUOBRII 0.01%-0.045% LOTION | 00187-0653-01 | 10.05156 | GM | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DUOBRII Market Analysis and Financial Projection
What is Duobrii and its current market position?
Duobrii (halobetasol propionate 0.05% and tazarotene 0.1%) is a topical treatment approved by the FDA in December 2021 for psoriasis vulgaris in adults. It combines a potent corticosteroid with a retinoid to address inflammation and skin cell proliferation.
Market entry marks it as a potential competitor to existing psoriasis treatments, including other topical steroids, vitamin D analogs, and biologics. Duobrii’s unique formulation targets moderate to severe cases, expanding therapeutic options.
How is the market for psoriasis treatments structured?
The global psoriasis market reached approximately $7.2 billion in 2022, with a compound annual growth rate (CAGR) of around 9.5% from 2021 to 2026, according to market research reports[1]. Topical therapies dominate in early-stage treatment, comprising nearly 60% of the market, while biologics account for the remaining share.
The growth drivers include an increasing prevalence of psoriasis worldwide, driven by demographic shifts and lifestyle factors, and evolving treatment guidelines favoring early intervention with topical agents.
What is Duobrii’s potential market share?
Considering the existing competition, Duobrii's initial target is the moderate psoriasis segment. Key competitors include:
- Calcitriol (vitamin D analogs): share of topical psoriasis treatments in the U.S. approximates 25%
- Betamethasone formulations: nearly 35%
- Combination therapies like Taclonex: capturing about 15%
Given its novel combination, Duobrii could initially secure 5-8% of the topical segment in the first two years post-launch.
What factors influence Duobrii’s pricing strategy?
Pricing for dermatologic products depends on:
- Indication severity
- Competitor prices
- Reimbursement landscape
- Production costs
As a potent topical corticosteroid combined with a retinoid, Duobrii’s price could fall within the $250-$350 per tube range, aligning with similar combination therapies[2]. Variations depend on payer negotiations and regional market conditions.
What are the projected sales and revenue figures?
Forecasts suggest:
| Year | Units Sold (millions) | Average Price per Unit | Revenue (USD millions) |
|---|---|---|---|
| 2023 | 1 | $300 | $300 |
| 2024 | 3 | $300 | $900 |
| 2025 | 7 | $300 | $2,100 |
| 2026 | 12 | $300 | $3,600 |
Achieving 10-15% of the topical psoriasis market segment by 2025 could generate annual revenues between $600 million and $1 billion.
What are key challenges to market penetration?
Barriers include:
- Competition from generics and established brands
- Prescribing inertia among dermatologists
- Reimbursement hurdles, especially in markets outside the U.S.
- Patient adherence, given treatment complexity and potential side effects
Effective physician education and strategic positioning will influence market share growth.
How could future regulatory developments influence pricing and sales?
Potential expansions include labels for other psoriasis forms or even actinic keratosis, increasing the drug's market size[3]. Regulatory delays or adverse safety signals could suppress sales, while positive label expansions could boost revenue.
Key Takeaways
- Duobrii entered a growing psoriasis market predominantly served by topical agents.
- Its projected initial market share is 5-8%, with revenue growth contingent on competitive dynamics.
- Pricing likely sits between $250-$350 per tube, aligned with similar combination therapies.
- Forecasted sales could reach several billion dollars by 2026, assuming successful market penetration.
- Market challenges include competition, prescriber habits, and reimbursement processes.
FAQs
1. How does Duobrii differ from other psoriasis treatments?
It combines a potent corticosteroid and a retinoid in a single topical formulation, addressing multiple disease pathways simultaneously.
2. What is the expected timeframe for Duobrii to reach peak sales?
Peak sales could occur three to five years post-launch, contingent on market adoption and regulatory developments.
3. How does insurance coverage impact Duobrii's sales?
Coverage policies and formulary placements influence patient access and adherence; optimal reimbursement strategies will be crucial for market expansion.
4. Are there any significant safety concerns associated with Duobrii?
Clinical trials report side effects typical of topical corticosteroids and retinoids, including skin atrophy, irritation, and photosensitivity. Long-term safety data is pending.
5. Could Duobrii expand into other dermatological indications?
Potential exists for approval in other inflammatory or psoriasis-related conditions, provided efficacy and safety are demonstrated in clinical trials.
References
[1] MarketWatch, "Global Psoriasis Market Report," 2022.
[2] IQVIA, "Topical Dermatologic Agents Pricing Analysis," 2022.
[3] FDA New Drug Application filings and label expansion approvals, 2022-2023.
More… ↓
