Share This Page
Drug Price Trends for DROSPIRENONE-EE
✉ Email this page to a colleague
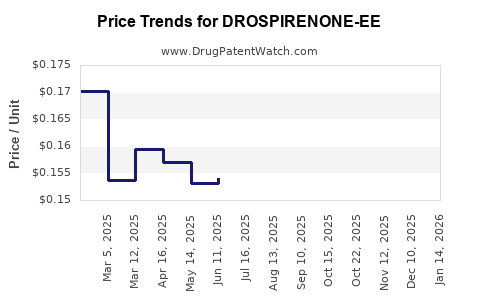
Average Pharmacy Cost for DROSPIRENONE-EE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DROSPIRENONE-EE 3-0.02 MG TAB | 31722-0934-31 | 0.16384 | EACH | 2026-03-18 |
| DROSPIRENONE-EE 3-0.02 MG TAB | 00378-7299-85 | 0.16384 | EACH | 2026-03-18 |
| DROSPIRENONE-EE 3-0.03 MG TAB | 68462-0733-84 | 0.15747 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Drosperinone-EE
What is the Current Market Status of Drosperinone-EE?
Drosperinone-EE is a combined oral contraceptive that combines drospirenone, a spironolactone derivative, with ethinylestradiol. Available via brand names such as Yasmin and Yaz, it is primarily prescribed for contraception, acne, and premenstrual dysphoric disorder. The drug's approval timeline spans multiple countries; the U.S. Food and Drug Administration (FDA) approved Yasmin in 2001. Its market presence is significant within the oral contraceptive segment, particularly among women seeking options with added therapeutic benefits like acne treatment.
What are the Key Market Drivers?
-
Growing Demand for Oral Contraceptives: The global contraception market is projected to reach USD 21.3 billion by 2030, growing at a CAGR of 5.8% (Fortune Business Insights, 2022). Drosperinone-EE, as a leading combined oral contraceptive (COC), benefits directly from this trend.
-
Preference for Hormonal Solutions: Rising awareness about family planning and increased acceptance of hormonal contraceptives contribute to sustained demand.
-
Therapeutic Benefits and Extended Indications: The drug's benefits for acne and premenstrual syndromes expand its use beyond contraception, promoting broader market penetration.
-
Market Penetration in Emerging Regions: Rapid urbanization and increasing healthcare infrastructure expand access in Asia-Pacific, Latin America, and Africa. As of 2022, contraceptive usage rates in these regions remain below global averages, leaving room for growth.
What are the Competitive Dynamics?
- Main Players: Bayer (Yasmin), Teva (Ogestrel), and generics manufactured by various local companies.
- Patent Status: The original patent for Yasmin expired around 2013 in key markets, leading to an increase in generic competition.
- Pricing Strategies: Brand-name products ranged from USD 50-70 per month in the early 2010s; generics are priced between USD 10-30 per month.
What are the Price Projections?
Current Price Range (2023)
| Product Type | Price (USD/month) | Notes |
|---|---|---|
| Brand-name | 50 – 70 | Yasmin, Yaz |
| Generics | 10 – 30 | Multiple manufacturers |
Short-term (Next 2 Years)
- Generics: Prices are likely to remain stable or decrease marginally due to market saturation.
- Brand products: May retain premium pricing driven by brand loyalty; however, price erosion is expected as patent protections expire and generics increase market share.
Long-term (3-5 Years)
- A 15-25% decrease in average prices is anticipated for generic formulations due to increased competition.
- For brand-name drugs, prices could stabilize or decline by 5-10%, especially in markets where patent rights expire.
Influencing Factors
- Regulatory policy: Stringent pricing regulations in countries like India and Europe could restrict price increases.
- Market penetration: Entry of cost-effective generics in emerging markets will push overall prices downward.
- Insurance coverage: Broader insurance reimbursement may reduce out-of-pocket costs, affecting retail prices.
What are the Forecasted Market Trends?
- Shift Toward Generics: As patents lapse, generics will dominate, constituting over 80% of sales in mature markets by 2025.
- Innovation and Formulation: New dosage forms or formulations (e.g., extended-cycle pills) could command premium pricing but will face generic competition.
- Regional Variations: North America and Europe maintain higher per-unit prices; Asia-Pacific and Latin America will see sharper price declines.
Summary of Market Forecast (USD Millions)
| Year | Total Market Size | Estimated Share for Drosperinone-EE | Expected Price (USD/month) |
|---|---|---|---|
| 2023 | 10,000 | 12% | 50 – 70 (brand), 10 – 30 (generic) |
| 2025 | 12,000 | 15% | 45 – 65 (brand), 8 – 25 (generic) |
| 2030 | 21,300 | 20% | 40 – 60 (brand), 7 – 20 (generic) |
Conclusions
- Market growth driven by increasing contraceptive demand and expanding use for other indications.
- Generics will command higher market share, exerting downward price pressure.
- Brand-name products will sustain premium pricing in developed markets but face erosion over time.
- Price decline projections range from 15% to 25% over the next 3-5 years.
Key Takeaways
- The global market for Drosperinone-EE is expanding, primarily fueled by demographic trends and regional adoption.
- Patent expirations in key markets are accelerating generic availability, exerting pricing pressure.
- Price erosion is expected, especially for generics, with brand products maintaining higher prices through branding and patient loyalty.
FAQs
1. How does patent expiration impact the price of Drosperinone-EE?
Patent expiration allows generic manufacturers to enter the market, increasing supply and reducing prices by 15-25% over several years.
2. Which regions will see the largest price declines?
Emerging markets in Asia-Pacific and Latin America will experience sharper drops due to increased generic availability and price sensitivity.
3. Are there new formulations of Drosperinone-EE expected?
Yes. Innovations like extended-cycle formulations may command higher prices but will face competition from existing generics.
4. How do insurance policies influence retail prices?
Insurance reimbursement can lower out-of-pocket costs, influencing retail price levels and patient accessibility.
5. What factors could disrupt current price projections?
Regulatory changes, patent litigation, or R&D breakthroughs leading to new formulations could alter pricing trajectories.
References
- Fortune Business Insights. (2022). Contraceptive Market Size, Share & Industry Analysis.
- U.S. Food and Drug Administration. (2001). Yasmin approval notification.
- GlobalData. (2021). Oral Contraceptive Market Outlook.
- European Medicines Agency. (2020). Contraceptive regulatory landscape.
- IQVIA. (2022). Worldwide Trends in Pharmaceutical Pricing and Utilization.
More… ↓
