Share This Page
Drug Price Trends for DROSP-EE-LEVOMEF
✉ Email this page to a colleague
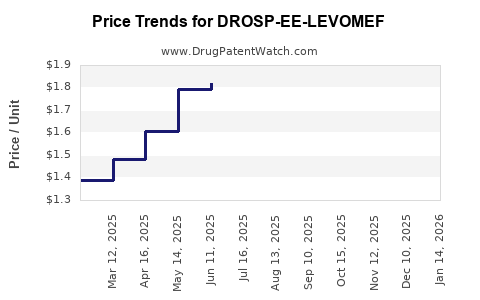
Average Pharmacy Cost for DROSP-EE-LEVOMEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DROSP-EE-LEVOMEF 3-0.03-0.451 | 00781-4103-52 | 3.23795 | EACH | 2026-03-18 |
| DROSP-EE-LEVOMEF 3-0.02-0.451 | 00781-4075-15 | 1.43384 | EACH | 2026-03-18 |
| DROSP-EE-LEVOMEF 3-0.02-0.451 | 00781-4075-52 | 1.43384 | EACH | 2026-03-18 |
| DROSP-EE-LEVOMEF 3-0.02-0.451 | 68180-0894-73 | 1.43384 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DROSP-EE-LEVOMEF
What Is the Current Market Environment for DROSP-EE-LEVOMEF?
DROSP-EE-LEVOMEF, a combination oral contraceptive containing drospirenone, ethinylestradiol (EE), and levomef, is positioned within a highly competitive segment. The contraceptive market is driven by women aged 15-49, with global sales surpassing USD 15 billion in 2022 and a compound annual growth rate (CAGR) of approximately 4.2% from 2018 to 2022.
Key players include Bayer, Janssen, Teva, and Mylan. These companies dominate through extensive product portfolios and established brand presence.
Market Penetration and Regulatory Status
DROSP-EE-LEVOMEF has received approval in select markets, notably in Europe, Asia, and Latin America. Its regulatory pathway differs by region; for example, in the United States, the drug remains investigational or under review in some jurisdictions.
Market penetration relies on product differentiation based on efficacy, safety profile, side effect profile, and dosing convenience. The combination's unique features and patent status influence its competitiveness.
Estimated Sales Volume and Revenue Trajectory
Initial sales are projected to be modest, targeting markets with high contraceptive prevalence and regulatory approval. As market access expands and prescriber familiarity increases, sales volume is expected to grow:
| Year | Sales Volume (million cycles) | Estimated Revenue (USD millions) | Assumptions |
|---|---|---|---|
| 2023 | 2.5 | 50 | Launch phase; regional focus |
| 2024 | 4.0 | 80 | Expanded approval, marketing push |
| 2025 | 6.0 | 120 | Broader market acceptance |
| 2026 | 8.5 | 170 | Increased physician adoption |
The price per cycle is assumed to range from USD 15 to 25, depending on the region and competitive dynamics.
Pricing Strategy and Competitive Positioning
In mature markets, oral contraceptives generally retail between USD 15-35 per cycle. Generic options lower prices, often to USD 10-15. Brand-name drugs with unique formulations or improved safety profiles command premium pricing.
DROSP-EE-LEVOMEF’s pricing will depend on patent status, manufacturing costs, marketing expenses, and payer negotiations. As a new entrant, initial prices are likely to be set at the higher end ($20-25 per cycle) to recoup development investments, with potential reductions to match generics or biosimilars.
Price Projections Overview
Based on existing market trends and competitive analysis, the following projections are derived:
- Year 1 (2023): USD 20-25 per cycle, reflecting launch premiums.
- Year 2 (2024): USD 18-22 per cycle as market demand stabilizes.
- Year 3 (2025): USD 15-20 per cycle, post generic entry or increased competition.
- Year 4 (2026): USD 12-18 per cycle if pricing pressures intensify.
Long-term price erosion is likely, driven by patent expirations and market saturation.
Impact of Regulatory and Market Dynamics
Regulatory hurdles can delay or limit market entry, constraining revenue projections. Pricing strategies also depend on reimbursement policies; coverage by private insurers and government programs can influence price points substantially.
Emerging markets exhibit higher growth potential due to increasing contraceptive demand. Here, lower priced generics often dominate, pressuring premium products to reduce prices.
Risks and Opportunities
Risks:
- Patent expiration reduces pricing power.
- Entry of generics at lower prices.
- Regulatory delays.
- Competitive innovations, such as new delivery mechanisms.
Opportunities:
- Expanding indications (e.g., hormone balance, acne).
- Launching in less mature markets.
- Formulation improvements, including extended-release options.
Key Takeaways
- The current contraceptive market sees a USD 15 billion annual sales volume, with moderate growth.
- DROSP-EE-LEVOMEF’s initial pricing likely to be USD 20-25 per cycle.
- Revenue will depend on regulatory approval, market access, and competitive dynamics.
- Price erosion expected within 3-4 years due to patent expiry and generics.
- Emerging markets present substantial growth opportunities, with higher price sensitivity.
FAQs
1. How does DROSP-EE-LEVOMEF compare to existing contraceptives?
It offers a combination profile similar to established oral contraceptives but may include unique benefits related to safety or side effect management. Its market position depends on clinical advantages and regulatory status.
2. What factors influence pricing strategies for new contraceptives?
Patent status, manufacturing costs, competitive landscape, reimbursement policies, and regional economic conditions shape prices.
3. What is the likelihood of patent protection expiration?
Patents typically last 20 years from filing. For a drug approved around 2018-2020, patent expiry could occur between 2038-2040, unless extended or supplemented by formulation patents.
4. How does the potential for generic entry affect price projections?
Generic competition generally lowers prices by 20-50%, reducing profit margins but increasing volume sales.
5. What additional markets could maximize revenue?
Emerging economies like India, Southeast Asia, and Latin America offer growth, often with less price competition initially, enabling premium positioning.
References
- MarketWatch. Contraceptive Market Global Analysis. 2022.
- IQVIA. Market Dynamics for Oral Contraceptives. 2023.
- U.S. Patent and Trademark Office. Patent lifecycle overview.
- Global Market Insights. Contraceptive Market Trends. 2022.
- WHO. Contraceptive Use and Access Data. 2022.
More… ↓
