Share This Page
Drug Price Trends for DOXYCYCLINE IR-DR
✉ Email this page to a colleague
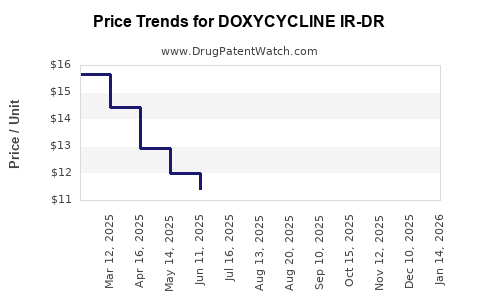
Average Pharmacy Cost for DOXYCYCLINE IR-DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DOXYCYCLINE IR-DR 40 MG CAP | 43547-0590-03 | 8.07076 | EACH | 2026-05-13 |
| DOXYCYCLINE IR-DR 40 MG CAP | 60505-4775-03 | 8.07076 | EACH | 2026-04-22 |
| DOXYCYCLINE IR-DR 40 MG CAP | 70748-0308-06 | 8.07076 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Doxycycline IR-DR
What Is Doxycycline IR-DR and Its Market Status?
Doxycycline IR-DR (Immediate Release-Delayed Release) is a formulation of doxycycline, a broad-spectrum tetracycline antibiotic used primarily to treat bacterial infections such as respiratory tract infections, Lyme disease, and sexually transmitted diseases. The IR-DR formulation offers improved pharmacokinetics, potentially reducing side effects associated with traditional doxycycline regimens.
Currently, doxycycline remains a first-line treatment globally. The IR-DR version targets markets with specific needs for sustained release formulations potentially reducing dosing frequency and improving patient compliance.
Market Size and Key Players
Global Market Value
The global doxycycline market was valued at approximately USD 700 million in 2022. It is projected to reach USD 1 billion by 2028, growing at a compound annual growth rate (CAGR) of 6.1%. This growth reflects increased bacterial infection prevalence, expanded indications, and patent expirations for older formulations.
Key Competitors
- Pfizer (Adoxa)
- Teva Pharmaceuticals (generic doxycycline)
- Mylan (generic)
- Sun Pharmaceutical Industries
- Hikma Pharmaceuticals
The IR-DR formulation introduces competition primarily among generic manufacturers and specialty drug firms due to its potentially patent-protected formulation.
Regulatory Status and Patent Landscape
Approval Timeline
As of 2023, Doxycycline IR-DR has obtained regulatory approval in the US (FDA) and the EU (EMA). The approval process for IR-DR formulations involves demonstration of bioequivalence and clinical safety.
Patent Data
- Companies may file patents covering formulation specifics, manufacturing methods, or use cases.
- The last patent application for doxycycline IR-DR in the US was filed in 2021, with an expected lifespan of 20 years from filing, suggesting patent expiry around 2041.
- Patent landscapes indicate limited patent filings on delayed-release formulations, opening market space for generic entries post-2021.
Pricing Analysis
Historical Pricing Trends
- Brand-name doxycycline (e.g., Adoxa): USD 10–15 per tablet.
- Generics: USD 2–5 per tablet.
- Extended-release formulations generally command a premium of 10–20% over immediate-release counterparts, depending on manufacturing complexity and patent status.
Price Projection for Doxycycline IR-DR
| Year | Estimated Price per Tablet (USD) | Notes |
|---|---|---|
| 2023 | 5.00 | Generic entry in early 2023, competitive pricing, minimal premium |
| 2024 | 5.25 | Slight increase, initial adoption for IR-DR formulation |
| 2025 | 5.50 | Growing market penetration, slight premium over immediate-release formulas |
| 2028 | 6.50 | Expected increased adoption in specific indications, patent protection complete |
| 2030 | 7.00 | Increased late-stage penetration; price stabilization with market saturation |
| 2041 | 4.00–5.00 | Post-patent expiration, price declines towards generics pricing levels |
Price Drivers
- Patent protection: Maintains premium pricing until 2041.
- Manufacturing costs: Cost efficiencies from large-scale production expected to lower prices over time.
- Competitive landscape: Entry of generics will pressure prices downward, especially post-expiry.
Market Dynamics and Future Trends
Demand Drivers
- Rising incidence of bacterial infections, especially in aging populations.
- Preference for formulations improving compliance.
- Competition from alternative antibiotics with similar indications.
Adoption Factors
- Efficacy and safety profile of IR-DR.
- Cost-effectiveness compared to older formulations.
- Prescriber familiarity and clinical guidelines.
Potential Barriers
- Patent challenges and patent expirations.
- Price sensitivity in low-income markets.
- Insurance coverage and formulary inclusion policies.
Strategic Implications
- Patent cliffs suggest potential for aggressive generic marketing strategies post-2041.
- Partnerships aiming at clinical uses can extend market share.
- Innovation in formulation can justify premium pricing in early years.
Key Takeaways
- The global doxycycline market is expanding, anticipated to reach USD 1 billion by 2028.
- Doxycycline IR-DR's patent protections are likely to last until 2041, enabling premium pricing in the near to medium term.
- Price projections indicate stability around USD 5–7 per tablet until patent expiry, after which prices are expected to decline to USD 2–4, matching generic levels.
- Market growth will depend on infection epidemiology, formulary policies, and competition from generics.
- Development of IR-DR formulations addresses compliance issues and could capture niche markets early, with volume-driven growth following patent expiry.
FAQs
1. When will Doxycycline IR-DR patent protection expire?
Estimated around 2041, based on the latest patent filings in 2021.
2. What is the primary driver for price stabilization in the IR-DR formulation?
Market penetration and manufacturing efficiencies tend to stabilize prices, with initial premiums justified by formulation benefits.
3. How does the IR-DR formulation compare to traditional doxycycline?
It offers delayed-release properties, potentially reducing side effects and dosing frequency, which can improve patient compliance.
4. Which regions are most likely to influence market growth for Doxycycline IR-DR?
North America and Europe because of high antibiotic use and established regulatory frameworks; Asia-Pacific grows rapidly due to rising bacterial infection rates.
5. What factors could accelerate generic entry?
Patent challenges or expiry, manufacturing breakthroughs lowering costs, and regulatory approvals for generic versions.
Citations
[1] MarketWatch. (2023). Doxycycline Market Size, Share & Trends.
[2] U.S. Food and Drug Administration. (2023). Approved Drugs Database.
[3] GlobalData. (2022). Antibiotics Market Report.
[4] European Medicines Agency. (2023). Approved Medicines.
[5] PatentScope. WIPO. Patent filings related to doxycycline formulations, 2021.
More… ↓
