Share This Page
Drug Price Trends for DORZOLAMIDE-TIMOLOL
✉ Email this page to a colleague
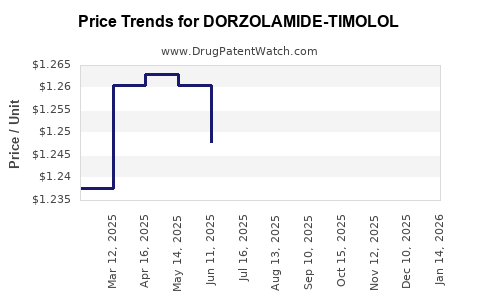
Average Pharmacy Cost for DORZOLAMIDE-TIMOLOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DORZOLAMIDE-TIMOLOL EYE DROPS | 24208-0486-10 | 0.94345 | ML | 2026-03-18 |
| DORZOLAMIDE-TIMOLOL 2-0.5%(PF) | 50742-0323-60 | 1.05706 | EACH | 2026-03-18 |
| DORZOLAMIDE-TIMOLOL EYE DROPS | 61314-0030-02 | 0.94345 | ML | 2026-03-18 |
| DORZOLAMIDE-TIMOLOL EYE DROPS | 42571-0147-26 | 0.94345 | ML | 2026-03-18 |
| DORZOLAMIDE-TIMOLOL 2-0.5%(PF) | 65862-0947-60 | 1.05706 | EACH | 2026-03-18 |
| DORZOLAMIDE-TIMOLOL EYE DROPS | 71921-0226-10 | 0.94345 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DORZOLAMIDE-TIMOLOL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DORZOLAMIDE-TIMOLOL (PF) | Thea Pharma, Inc. | 50383-0261-61 | 60X0.2ML | 45.91 | 2023-02-01 - 2028-01-31 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Dorzolamide-Timolol Ophthalmic Solution: Patent Landscape and Market Forecast
Dorzolamide-timolol ophthalmic solution, a fixed-dose combination for lowering intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension, faces a mature market with significant generic competition. Key patents for the original New Drug Application (NDA) have expired, allowing for widespread generic availability. The market's trajectory is influenced by the availability and pricing of generics, the emergence of newer treatment modalities, and evolving clinical guidelines.
What is the patent status of dorzolamide-timolol ophthalmic solution?
The primary patents covering the original NDA for dorzolamide-timolol ophthalmic solution, including formulation and method of use patents, have expired. For instance, U.S. Patent No. 5,451,605, which claimed a topical carbonic anhydrase inhibitor, expired in 2013. Similarly, patents related to the combination therapy and specific formulations have also lapsed.
The Orange Book lists the original NDA 020670 for dorzolamide-timolol ophthalmic solution (Cosopt® by Merck & Co.) with multiple patent expirations preceding the present. The last listed patent expiry for the originator product was in 2015, based on its 2007 patent extension [1].
Generic manufacturers have successfully launched their versions of dorzolamide-timolol ophthalmic solution following these patent expirations. The landscape is characterized by a significant number of approved generic products from various pharmaceutical companies.
Newer patent filings related to this combination, if any, are likely to focus on novel delivery systems, specific patient populations, or modified formulations (e.g., preservative-free versions, sustained-release formulations), rather than the core active pharmaceutical ingredients (APIs) themselves. For example, patents may address improved stability, enhanced patient compliance, or reduced side effects.
What is the current market size and segmentation for dorzolamide-timolol ophthalmic solution?
The global market for dorzolamide-timolol ophthalmic solution is substantial, driven by the high prevalence of glaucoma and ocular hypertension worldwide. Quantifying the exact market size for this specific fixed-dose combination is challenging as it is often reported within broader categories of glaucoma treatment or topical ophthalmic medications.
However, the market is segmented by:
- Brand vs. Generic: The market is dominated by generic products due to patent expirations. The original branded product (Cosopt®) holds a diminishing market share as generic alternatives offer significant cost savings.
- Prescription Channels: Sales are primarily through retail pharmacies, hospital pharmacies, and direct sales to ophthalmologists.
- Geographic Regions: North America and Europe represent significant markets due to high healthcare expenditure and established diagnostic infrastructure. Asia-Pacific is a growing market with increasing awareness and access to treatment.
- Formulations: The standard ophthalmic solution is the primary formulation. Preservative-free versions are available and cater to specific patient needs, often commanding a premium price.
Estimates from market research reports indicate that the global glaucoma treatment market, which includes dorzolamide-timolol, is valued in the billions of dollars and is projected to grow. While specific figures for dorzolamide-timolol alone are not always disaggregated, its position as a widely prescribed first- and second-line therapy contributes significantly to this market. For instance, the global glaucoma treatment market was valued at approximately $5.5 billion in 2022 and is projected to reach over $7.9 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.6% [2]. Dorzolamide-timolol represents a substantial portion of this market due to its established efficacy and accessibility.
Who are the key players in the dorzolamide-timolol ophthalmic solution market?
The market for dorzolamide-timolol ophthalmic solution is highly competitive, with a large number of generic manufacturers. The originator, Merck & Co. (with Cosopt®), is a key player but faces intense competition from generic alternatives.
Key generic manufacturers include, but are not limited to:
- Sun Pharmaceutical Industries Ltd.: A major global pharmaceutical company with a broad portfolio of generic ophthalmic products.
- Teva Pharmaceutical Industries Ltd.: A leading generic drug manufacturer with a significant presence in the ophthalmic space.
- Allergan (now part of AbbVie): While historically a significant player, their role may have shifted with the integration into AbbVie.
- Apotex Inc.: A Canadian pharmaceutical company with a global reach in generics.
- Hikma Pharmaceuticals PLC: A multinational pharmaceutical group with generic and branded products.
- Bausch Health Companies Inc.: Offers a range of ophthalmic solutions, including glaucoma treatments.
- Laboratories Valeant (now Bausch Health): Historically a player in this segment.
- Lupin Limited: Another major Indian pharmaceutical company with a growing generics business.
- Viatem, Inc.: Known for its generic ophthalmic products.
- Matrix Laboratories (part of Mylan/Viatris): A significant API and finished dosage form manufacturer.
These companies compete based on price, product availability, and distribution networks. The market is characterized by constant price adjustments and promotional activities to gain and maintain market share.
What are the primary therapeutic benefits and clinical indications of dorzolamide-timolol?
Dorzolamide-timolol ophthalmic solution is a combination medication that reduces intraocular pressure (IOP). Elevated IOP is a primary risk factor for optic nerve damage and vision loss in glaucoma.
- Dorzolamide: A topical carbonic anhydrase inhibitor. It works by decreasing the production of aqueous humor within the eye.
- Timolol: A beta-adrenergic blocker. It also reduces aqueous humor production and may slightly increase uveoscleral outflow.
The combination of these two mechanisms of action provides a synergistic effect, leading to a greater reduction in IOP compared to either agent alone [3].
Primary Clinical Indications:
- Open-Angle Glaucoma: A common form of glaucoma characterized by a slow blockage in the drainage angle of the eye, leading to gradual IOP elevation.
- Ocular Hypertension: A condition where IOP is elevated, but without detectable optic nerve damage or visual field loss. This condition carries an increased risk of developing glaucoma.
The drug is typically prescribed as a first-line treatment or when monotherapy with a prostaglandin analog, beta-blocker, or carbonic anhydrase inhibitor is insufficient to control IOP. Its efficacy in lowering IOP has been well-established in numerous clinical trials [4, 5].
What factors influence the price of dorzolamide-timolol ophthalmic solution?
The pricing of dorzolamide-timolol ophthalmic solution is primarily influenced by the following factors:
- Generic Competition: With numerous generic manufacturers in the market, competition is fierce, driving prices down significantly from the original branded product. The presence of multiple suppliers for the same API also contributes to price erosion.
- Manufacturing Costs: The cost of raw materials (APIs, excipients), manufacturing processes, quality control, and packaging contribute to the overall production cost.
- Supply Chain Dynamics: Global supply chain disruptions, raw material availability, and transportation costs can impact pricing.
- Regulatory Approvals and Compliance: Adhering to strict FDA (and other regulatory bodies') manufacturing standards and obtaining approval for generic versions involves significant investment.
- Formulation Specifics: Preservative-free formulations, while offering benefits for certain patients (e.g., those with dry eye or contact lens wearers), typically command a higher price due to more complex manufacturing and packaging requirements.
- Reimbursement Policies: Payer policies, formularies, and reimbursement rates set by government and private insurers influence what healthcare providers and patients are willing to pay.
- Volume Discounts and Contracts: Large purchasers, such as hospital systems or pharmacy benefit managers (PBMs), can negotiate significant volume discounts.
- Competition from Alternative Therapies: The availability and pricing of other glaucoma medications, including newer classes of drugs and surgical options, can indirectly influence pricing strategies.
Price Comparison Example:
As of late 2023/early 2024, a 5 mL bottle of generic dorzolamide-timolol ophthalmic solution (e.g., 2% / 0.5%) can range from approximately $10 to $30 USD at retail pharmacies, depending on the manufacturer, pharmacy, and insurance coverage. This is a stark contrast to the cost of the branded product before its patent expiry, which could be upwards of $150-$200 USD per bottle. Preservative-free versions may be priced 30-50% higher than their preserved counterparts.
What are the projected market trends and future outlook for dorzolamide-timolol ophthalmic solution?
The market for dorzolamide-timolol ophthalmic solution is expected to remain relatively stable in terms of volume, driven by its established efficacy and cost-effectiveness as a generic treatment option. However, significant growth in terms of revenue for this specific combination is unlikely, and potential price erosion may continue.
Projected Market Trends:
- Continued Generic Dominance: Generic versions will continue to be the primary driver of the market, offering a cost-effective solution for patients.
- Price Competition: Intense price competition among generic manufacturers will persist, potentially leading to further price decreases.
- Rise of Newer Therapies: The development and adoption of novel glaucoma treatments, including sustained-release drug delivery systems (e.g., Ring Designs, injectable implants), prostaglandin analogs with improved profiles, and minimally invasive glaucoma surgery (MIGS), may gradually impact the market share of traditional topical agents like dorzolamide-timolol, particularly in patients requiring more advanced or differentiated treatment options.
- Focus on Preservative-Free Formulations: Demand for preservative-free versions is likely to increase, driven by patient and physician preference for improved ocular surface health and reduced adverse effects. This segment may offer higher profit margins for manufacturers.
- Geographic Market Expansion: Growth in emerging markets in Asia and Latin America, as access to healthcare and diagnosis improves, will contribute to volume increases.
- Combination Therapies: While dorzolamide-timolol is a fixed-dose combination, there may be continued research into optimized combinations or novel delivery methods for existing APIs to improve patient adherence and efficacy.
- Clinical Practice Guidelines: Evolving clinical guidelines for glaucoma management may influence prescribing patterns, potentially favoring newer agents or treatment algorithms that incorporate a wider array of therapeutic options.
Future Outlook:
The outlook for dorzolamide-timolol ophthalmic solution is one of maturity and sustained, albeit modest, utility. It will remain a cornerstone therapy for many patients with open-angle glaucoma and ocular hypertension due to its proven track record, affordability, and broad availability. However, its growth trajectory will be constrained by the aforementioned factors, particularly the continuous innovation in the glaucoma therapeutic landscape. Manufacturers focusing on cost-efficient production, robust distribution, and potentially niche formulations (preservative-free) will be best positioned to succeed in this competitive environment.
How does dorzolamide-timolol compare to other topical glaucoma medications?
Dorzolamide-timolol offers a dual-mechanism approach to IOP lowering, differentiating it from monotherapies and some other combination products.
| Medication Class | Primary Mechanism | Example(s) | Typical IOP Reduction (Approximate %) | Key Advantages | Key Disadvantages |
|---|---|---|---|---|---|
| Dorzolamide-Timolol | Decreases aqueous humor production | Cosopt® (Merck), Generic versions | 25-35% | Dual mechanism, synergistic effect, well-established efficacy, cost-effective (generic) | Potential for systemic side effects of timolol (e.g., bradycardia), ocular side effects |
| Prostaglandin Analogs | Increases uveoscleral outflow | Latanoprost (Xalatan®), Travoprost, Bimatoprost | 25-35% | Once-daily dosing, generally well-tolerated ocularly, significant IOP lowering | Ocular side effects (iris pigmentation, eyelash growth, conjunctival hyperemia) |
| Beta-Blockers (Monotherapy) | Decreases aqueous humor production | Timolol (Timoptic®), Betaxolol | 15-25% | Well-established, cost-effective (generic) | Systemic side effects (respiratory, cardiovascular), ocular irritation |
| Alpha-Adrenergic Agonists | Decreases aqueous humor production, increases outflow | Brimonidine (Alphagan®) | 15-25% | Can be used as adjunctive therapy, alternative mechanism for patients intolerant to beta-blockers | Ocular allergy, dry eye, systemic side effects (fatigue, dry mouth) |
| Carbonic Anhydrase Inhibitors (Topical) | Decreases aqueous humor production | Dorzolamide (Trusopt®), Brinzolamide | 15-25% | Alternative mechanism for patients intolerant to beta-blockers | Ocular irritation, systemic side effects (less common than oral CAIs) |
| Rho Kinase Inhibitors | Increases uveoscleral outflow, reduces episcleral venous pressure | Netarsudil (Rhopressa®) | 15-25% | Novel mechanism, potential for IOP reduction at specific times of day | Ocular hyperemia, corneal verticillata, higher cost |
Dorzolamide-timolol is often favored for its proven efficacy and convenience as a single drop. Its generic availability makes it a cost-effective choice for a large patient population. However, for patients who cannot tolerate beta-blockers due to systemic contraindications (e.g., asthma, bradycardia) or experience significant ocular side effects, alternative monotherapies or different combination products are preferred. Newer agents like rho kinase inhibitors offer novel mechanisms but are typically more expensive and may be reserved for specific clinical situations or as adjunctive therapy.
What are the regulatory considerations for dorzolamide-timolol ophthalmic solution?
The regulatory landscape for dorzolamide-timolol ophthalmic solution is primarily governed by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- ANDA (Abbreviated New Drug Application): Generic manufacturers must submit an ANDA to the FDA, demonstrating that their product is bioequivalent to the reference listed drug (RLD), in this case, Cosopt®. This involves proving sameness in API, dosage form, strength, route of administration, and therapeutic equivalence.
- Patent Certifications (Hatch-Waxman Act): Generic applicants must certify their intent to market the drug, either by stating that relevant patents have expired, will expire, are invalid, or will not be infringed upon. This process can lead to patent litigation.
- cGMP Compliance: All manufacturing facilities must adhere to current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and consistency. Regular inspections by regulatory authorities are standard.
- Labeling Requirements: Generic labels must be consistent with the RLD's labeling, including indications, contraindications, warnings, precautions, and adverse reactions.
- Post-Marketing Surveillance: Manufacturers are required to monitor and report adverse events that occur after the drug is on the market.
- Preservative-Free Formulations: Approval for preservative-free versions involves demonstrating sterility and stability without the use of preservatives, often requiring specialized packaging and manufacturing processes.
- International Harmonization: Manufacturers seeking approval in multiple regions must navigate varying regulatory requirements, though efforts towards international harmonization exist.
The regulatory pathway for generics, while stringent, allows for rapid market entry once primary patents have expired, fostering competition and driving down prices.
Key Takeaways
- Dorzolamide-timolol ophthalmic solution is a mature market segment with expired originator patents, leading to widespread generic availability and intense price competition.
- The drug is a cornerstone therapy for open-angle glaucoma and ocular hypertension, valued for its dual-mechanism IOP reduction and cost-effectiveness in generic form.
- Key market players are predominantly generic manufacturers, with Sun Pharma, Teva, and Apotex among the prominent suppliers.
- Pricing is driven by generic competition, manufacturing costs, and supply chain factors, with significant price drops from the original branded product.
- Future market trends indicate continued generic dominance, potential for further price erosion, and increasing demand for preservative-free formulations, alongside competition from newer glaucoma treatment modalities.
Frequently Asked Questions
-
What is the expected price trajectory for generic dorzolamide-timolol ophthalmic solution in the next five years? The price trajectory is expected to be characterized by continued gradual erosion due to ongoing competition among a large number of generic manufacturers. Significant price increases are unlikely unless there are substantial shifts in manufacturing costs or raw material availability.
-
Are there any significant pipeline developments or new patent filings for dorzolamide-timolol? New patent filings are unlikely for the core API combination or its standard formulation. Any new patents would likely focus on novel delivery systems (e.g., sustained-release implants, improved topical formulations), specific patient subpopulations, or unique manufacturing processes. Research is more focused on entirely new therapeutic classes or novel mechanisms of action for glaucoma.
-
How does the efficacy of generic dorzolamide-timolol compare to the branded Cosopt®? Generic dorzolamide-timolol ophthalmic solutions are required to be bioequivalent to the branded Cosopt®. This means they demonstrate the same rate and extent of absorption, resulting in comparable therapeutic effects and safety profiles when used as directed.
-
What are the most common adverse events associated with dorzolamide-timolol ophthalmic solution? Common ocular adverse events include stinging, burning, transient blurred vision, and foreign body sensation. Less common but more serious side effects can include allergic reactions, changes in taste, and, due to the timolol component, systemic effects such as bradycardia, bronchospasm, and central nervous system depression in susceptible individuals.
-
In which patient populations might dorzolamide-timolol be contraindicated or require special caution? Dorzolamide-timolol is contraindicated in patients with bronchial asthma, severe COPD, sinus bradycardia, second- or third-degree atrioventricular block, overt cardiac failure, or cardiogenic shock. Caution is advised in patients with myasthenia gravis, certain renal or hepatic impairments, and those undergoing surgery with general anesthesia.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from FDA Orange Book Database (Note: Specific patent expiry dates are derived from entries within this database for NDA 020670).
[2] Global Market Insights. (2023). Glaucoma Treatment Market Size, Share & Trends Analysis Report By Drug Class, By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030.
[3] Noecker, R. J., et al. (2002). A comparison of dorzolamide/timolol combination and latanoprost in patients with elevated intraocular pressure. Ophthalmology, 109(11), 2049-2057.
[4] World Glaucoma Association. (2015). World Glaucoma Association Consensus Portfolio.
[5] Day, S. E., et al. (2007). Dorzolamide/timolol combination therapy versus latanoprost in patients with open-angle glaucoma or ocular hypertension. Journal of Glaucoma, 16(4), 340-345.
More… ↓
