Share This Page
Drug Price Trends for DISULFIRAM
✉ Email this page to a colleague
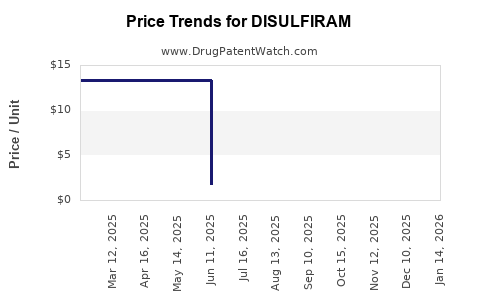
Average Pharmacy Cost for DISULFIRAM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DISULFIRAM 250 MG TABLET | 62135-0431-30 | 1.62320 | EACH | 2026-04-22 |
| DISULFIRAM 250 MG TABLET | 62135-0431-90 | 1.62320 | EACH | 2026-04-22 |
| DISULFIRAM 250 MG TABLET | 47781-0607-30 | 1.62320 | EACH | 2026-04-22 |
| DISULFIRAM 500 MG TABLET | 62135-0432-90 | 13.31433 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DISULFIRAM
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DISULFIRAM 500MG TAB | Golden State Medical Supply, Inc. | 00054-0357-25 | 100 | 418.77 | 4.18770 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| DISULFIRAM 250MG TAB | Golden State Medical Supply, Inc. | 60429-0196-30 | 30 | 64.37 | 2.14567 | EACH | 2024-04-04 - 2028-06-14 | FSS |
| DISULFIRAM 500MG TAB | Golden State Medical Supply, Inc. | 00054-0357-25 | 100 | 445.99 | 4.45990 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| DISULFIRAM 250MG TAB | Golden State Medical Supply, Inc. | 60429-0196-01 | 100 | 101.01 | 1.01010 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Disulfiram Market Analysis and Price Projections
Market Overview and Current Status
Disulfiram, a long-established drug primarily used for the treatment of chronic alcoholism, is experiencing a resurgence in interest due to emerging research into its potential applications in oncology and other disease areas. Approved by the U.S. Food and Drug Administration (FDA) in 1951, disulfiram's established safety profile and low manufacturing cost contribute to its current market position. The primary indication remains alcohol use disorder (AUD), where it acts as a deterrent by inducing an unpleasant physical reaction upon alcohol consumption.
The global market for disulfiram is currently moderate, driven by its established use in AUD treatment and a growing pipeline of investigational uses. The current market size is estimated to be between $50 million and $100 million USD annually. Production is largely generic, with multiple manufacturers operating globally. Key manufacturers include Teva Pharmaceutical Industries, Chartwell Pharmaceuticals, and numerous smaller generic producers in India and China.
The drug's mechanism of action involves inhibiting aldehyde dehydrogenase, leading to the accumulation of acetaldehyde when alcohol is ingested. This leads to symptoms such as flushing, nausea, vomiting, and palpitations.
Expanding Therapeutic Landscape
Beyond its established role in AUD, disulfiram is under investigation for several promising indications, which are poised to significantly impact its future market trajectory.
Oncology Applications
Research into disulfiram's anticancer properties has intensified. Studies suggest that disulfiram, often in combination with copper, can target cancer stem cells and induce cell death in various cancer types.
-
Mechanisms in Oncology: Disulfiram's anticancer effects are attributed to multiple mechanisms:
- Inhibition of the proteasome pathway, disrupting protein degradation and leading to cellular stress.
- Induction of reactive oxygen species (ROS) generation, causing oxidative damage to cancer cells.
- Inhibition of aldehyde dehydrogenase (ALDH) isoforms that are overexpressed in cancer stem cells, rendering them vulnerable.
- Copper chelation, which can modulate cellular processes involved in cancer progression.
-
Clinical Trial Landscape:
- Prostate Cancer: Several Phase II trials have investigated disulfiram in combination with other agents for recurrent prostate cancer. Results have shown potential in stabilizing disease and achieving biochemical responses. For example, a study published in the Journal of Clinical Oncology (2017) reported promising response rates in men with castration-resistant prostate cancer. [1]
- Breast Cancer: Early-stage research is exploring disulfiram's efficacy in HER2-positive breast cancer and other subtypes, often in preclinical models and early-phase clinical investigations.
- Other Cancers: Investigations are ongoing for glioblastoma, lung cancer, and pancreatic cancer, with preliminary data suggesting potential benefits.
The success of these ongoing clinical trials is critical for disulfiram's expansion into oncology. Regulatory approval for these new indications would represent a significant market expansion.
Other Investigational Uses
Disulfiram's multifaceted biological activity has led to its exploration in other therapeutic areas:
- Neurodegenerative Diseases: Preclinical studies suggest disulfiram may have a role in managing neurodegenerative disorders like Parkinson's disease by modulating protein aggregation and oxidative stress.
- Infectious Diseases: Its potential as an antiviral and antiparasitic agent is also being explored.
- Autism Spectrum Disorder (ASD): Limited studies have explored disulfiram's impact on certain behavioral aspects of ASD, though results are preliminary and require further validation.
Patent Landscape and Intellectual Property
The original patents for disulfiram have long expired, making the active pharmaceutical ingredient (API) freely available for generic manufacturing. However, intellectual property protection is now focused on:
- New Formulations: Novel delivery systems or modified release formulations of disulfiram that improve efficacy, reduce side effects, or enhance patient compliance.
- Combination Therapies: Patents covering specific combinations of disulfiram with other drugs for the treatment of cancer or other diseases.
- Specific Indications: Patent applications are being filed and granted for the use of disulfiram in specific disease contexts, particularly in oncology.
- Manufacturing Processes: Innovations in the synthesis or purification of disulfiram API could be patentable.
Key Patent Holders and Developers:
While no single entity dominates the disulfiram patent landscape for its original indication, companies actively involved in oncology research are filing for new intellectual property. Companies like Onconova Therapeutics, Theralagen (investigating disulfiram in combination therapies), and various academic institutions are prominent in patent filings related to novel uses and formulations.
The expiration of foundational patents means that the primary competitive advantage will shift to companies that can secure strong patent protection for new indications, novel formulations, or combination therapies.
Market Dynamics and Competitive Landscape
The disulfiram market is characterized by a dual structure: a mature generic market for AUD treatment and a nascent, high-potential market for emerging indications.
Generic AUD Market
- Pricing: Prices in the generic AUD market are highly competitive and relatively stable, with typical wholesale prices for a 30-day supply of 500 mg tablets ranging from $15 to $50 USD, depending on the manufacturer and distributor. This is driven by high generic penetration and the absence of significant product differentiation.
- Competition: Numerous generic manufacturers compete on price and availability. Key players include Teva Pharmaceutical Industries, Chartwell Pharmaceuticals, and many others, particularly from India and China.
- Growth: Growth in this segment is slow, largely tied to the prevalence of alcohol use disorder and public health initiatives.
Emerging Indications Market (Oncology, etc.)
- Pricing: Pricing for disulfiram in oncology or other novel indications will be significantly higher, reflecting the R&D investment, clinical trial costs, and the potential for premium pricing of novel therapies. If approved for oncology, pricing could range from several hundred to thousands of dollars per month, comparable to other cancer therapies.
- Competition: The competitive landscape here is evolving. Companies developing disulfiram-based therapies for oncology are often innovative biotechs or pharmaceutical companies seeking to leverage disulfiram's unique properties. Competition will come from other therapeutic agents targeting similar pathways or cancer types.
- Growth: This segment has the highest growth potential. Successful clinical trials and regulatory approvals will drive substantial market expansion.
Barriers to Entry
- Generic Market: Low barriers to entry due to the expired foundational patents. Manufacturing expertise and cost efficiency are key.
- Emerging Indications Market: High barriers to entry due to the significant cost and complexity of clinical development, regulatory approval processes, and the need for robust intellectual property protection.
Price Projections
Price projections for disulfiram vary significantly based on the intended indication.
Generic Disulfiram (AUD Treatment)
- 2024-2026: Stable pricing, with a slight downward pressure due to ongoing generic competition. Expected range: $15 - $45 USD per 30-day supply.
- 2027-2030: Modest price increases may occur due to inflationary pressures and potential supply chain disruptions, but significant shifts are unlikely without major changes in market dynamics. Expected range: $20 - $55 USD per 30-day supply.
Branded Disulfiram (Oncology/Novel Indications)
- 2026-2028 (Assuming successful Phase III trials and initial regulatory approvals for oncology): Initial launch pricing for oncology indications could be aggressive, reflecting R&D investment. Projected price: $800 - $1,500 USD per month.
- 2029-2032 (Market Penetration and Competition): As market penetration increases and potential competitors emerge, pricing may see some adjustment. However, the premium associated with novel cancer therapies is likely to sustain higher price points. Projected price: $1,000 - $1,800 USD per month.
Factors Influencing Price Projections:
- Clinical Trial Success Rates: Positive Phase III data and subsequent regulatory approvals are the primary drivers for premium pricing.
- Regulatory Landscape: FDA and EMA approval timelines and requirements will impact market entry.
- Reimbursement Policies: Payer coverage and formulary placement will be critical determinants of actual market prices.
- Competitive Landscape: The emergence of alternative treatments for the same indications will influence pricing power.
- Manufacturing Costs: While disulfiram API is inexpensive, novel formulations or specialized manufacturing for combination therapies can increase costs.
- Geopolitical and Economic Factors: Global economic conditions and trade policies can influence API sourcing and overall pricing.
Key Takeaways
- Disulfiram operates in two distinct market segments: a mature, price-sensitive generic market for alcohol use disorder and a nascent, high-growth potential market driven by emerging oncology and other investigational uses.
- The expansion into oncology indications, if successful, represents the most significant opportunity for market growth and premium pricing.
- Intellectual property is now centered on novel formulations, combination therapies, and specific disease indications, rather than the disulfiram molecule itself.
- Generic disulfiram pricing is expected to remain stable with slight downward pressure, while branded disulfiram for novel indications could command prices comparable to existing oncology therapies.
Frequently Asked Questions
What is the current market size for disulfiram?
The global market for disulfiram is currently estimated between $50 million and $100 million USD annually, primarily driven by its use in alcohol use disorder.
What are the most promising new therapeutic areas for disulfiram?
The most promising new therapeutic areas are oncology, with ongoing research in prostate, breast, and other cancers, as well as potential applications in neurodegenerative diseases.
What is the primary mechanism of action for disulfiram in treating alcoholism?
Disulfiram inhibits aldehyde dehydrogenase, leading to acetaldehyde accumulation upon alcohol consumption, which causes an unpleasant physical reaction deterring further drinking.
How is intellectual property protection for disulfiram evolving?
Intellectual property is now focused on new formulations, combination therapies, and specific disease indications, rather than the disulfiram molecule itself, as original patents have expired.
What factors will influence the future pricing of disulfiram for oncology applications?
Future pricing will be influenced by clinical trial success rates, regulatory approvals, reimbursement policies, the competitive landscape of oncology treatments, and manufacturing costs for novel applications.
Citations
[1] Chi, K. N., Holden, S. F., Diao, L., Lonergan, P., Lee, L., Van Veldhuizen, P., ... & Gleave, M. E. (2017). Phase II study of disulfiram in combination with copper in patients with castration-resistant prostate cancer. Journal of Clinical Oncology, 35(15_suppl), 5037-5037.
More… ↓
