Share This Page
Drug Price Trends for DIGOXIN
✉ Email this page to a colleague
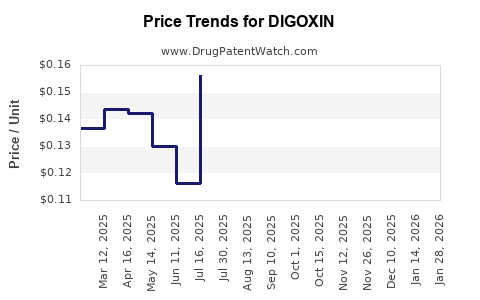
Average Pharmacy Cost for DIGOXIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIGOXIN 0.05 MG/ML SOLUTION | 69292-0605-60 | 1.06604 | ML | 2026-03-18 |
| DIGOXIN 0.05 MG/ML SOLUTION | 66689-0327-02 | 1.06604 | ML | 2026-03-18 |
| DIGOXIN 0.05 MG/ML SOLUTION | 00054-0057-46 | 1.06604 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DIGOXIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DIGOXIN 0.125MG TAB | Golden State Medical Supply, Inc. | 00143-1240-10 | 1000 | 166.36 | 0.16636 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| DIGOXIN 0.25MG TAB | Golden State Medical Supply, Inc. | 00143-1241-01 | 100 | 16.64 | 0.16640 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| DIGOXIN 0.05MG/ML ELIXIR | Golden State Medical Supply, Inc. | 66689-0327-02 | 60ML | 36.90 | 0.61500 | ML | 2023-06-16 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
DIGOXIN PATENT LANDSCAPE AND MARKET PROJECTIONS
Digoxin, a cardiac glycoside used to treat heart failure and atrial fibrillation, faces a mature and highly competitive market. Its primary patent expired in 1980 [1], leading to widespread generic availability. Market dynamics are driven by cost-effectiveness, physician prescribing habits, and the emergence of newer therapeutic alternatives. Price projections indicate continued stability within the generic segment, with potential for minor fluctuations based on manufacturing costs and supply chain considerations.
WHAT IS THE CURRENT PATENT STATUS OF DIGOXIN?
The foundational patents protecting the original discovery and synthesis of digoxin have long expired. U.S. Patent No. 2,402,508, assigned to S.M. Kalckar, for example, was granted in 1946 and expired decades ago [2]. There are no active, broad composition-of-matter patents that would prevent generic manufacturing or sale. Current patent activity related to digoxin typically focuses on specific formulations, delivery methods, or manufacturing processes, which offer limited market exclusivity and are unlikely to significantly alter the generic landscape.
| Patent Type | Expiration Status | Impact on Market |
|---|---|---|
| Composition of Matter | Expired (1980s) | High ( Generic Entry) |
| Formulation/Delivery Methods | Active (Limited) | Low (Incremental) |
| Manufacturing Processes | Active (Limited) | Low (Incremental) |
HOW HAS DIGOXIN PRICING EVOLVED POST-PATENT EXPIRATION?
Following patent expiration, digoxin entered the generic drug market. This transition led to a sharp and sustained decline in drug prices as multiple manufacturers competed. The average wholesale price (AWP) for digoxin tablets has remained relatively low, reflecting its status as a well-established, off-patent medication.
In recent years, the price of generic digoxin has been characterized by stability, with minor shifts influenced by supply, demand, and the cost of raw materials. Data from drug pricing databases indicates that the average price per tablet has hovered in the low cent range for years. For instance, a common 0.125 mg tablet might cost between $0.05 and $0.15 per unit, depending on the manufacturer and the pharmacy or wholesaler [3]. This contrasts sharply with the prices of branded cardiac medications or newer generics introduced with patent protection.
| Product Form | Typical Strength | Approximate Price Range (per unit) |
|---|---|---|
| Tablets | 0.125 mg | $0.05 - $0.15 |
| Tablets | 0.25 mg | $0.06 - $0.18 |
| Oral Solution | 0.05 mg/mL | $5.00 - $15.00 per 60 mL bottle |
WHAT ARE THE KEY MARKET DRIVERS FOR DIGOXIN?
The market for digoxin is primarily driven by its established efficacy in specific patient populations and its low cost compared to newer therapies.
- Established Efficacy: Digoxin remains a cornerstone therapy for certain patients with systolic heart failure, particularly those in sinus rhythm who remain symptomatic despite guideline-directed medical therapy [4]. It is also used to control heart rate in atrial fibrillation.
- Cost-Effectiveness: As a generic medication, digoxin offers a significant cost advantage. This makes it an attractive option for formulary inclusion by payers and a preferred choice for patients with limited insurance coverage or high co-pays for newer medications.
- Physician Familiarity and Prescribing Habits: Cardiologists and general practitioners have decades of experience prescribing digoxin. Its known adverse effects and drug interactions are well-documented, contributing to physician comfort in its use.
- Narrow Therapeutic Index: While effective, digoxin has a narrow therapeutic index, meaning the difference between a therapeutic dose and a toxic dose is small. This necessitates careful patient selection and monitoring, which can limit its use in certain complex cases or where adherence is a concern.
- Competition from Newer Agents: The market has seen the introduction of newer drug classes for heart failure and atrial fibrillation, such as Angiotensin Receptor-Neprilysin Inhibitors (ARNIs), Sodium-Glucose Cotransporter-2 inhibitors (SGLT2is), and newer oral anticoagulants (NOACs). These agents often offer improved outcomes or different mechanisms of action, leading to a gradual shift in treatment paradigms, particularly in advanced heart failure.
WHAT ARE THE PRICE PROJECTIONS FOR DIGOXIN IN THE NEXT 3-5 YEARS?
Price projections for digoxin are expected to remain largely stable due to its mature generic status and the absence of significant patent-driven market disruptions.
- Continued Generic Competition: The market will continue to be characterized by multiple generic manufacturers. This intense competition inherently suppresses prices. Any significant price increases would likely be unsustainable unless driven by broad supply chain disruptions or substantial increases in manufacturing input costs.
- Manufacturing Cost Sensitivity: The price of digoxin is closely tied to the cost of active pharmaceutical ingredients (APIs) and manufacturing. Fluctuations in API sourcing, labor costs, and regulatory compliance can introduce minor price volatility. However, the low volume of production per unit cost generally buffers significant price spikes.
- Stable Demand: While newer agents have gained traction, digoxin maintains a consistent demand for specific indications. Its role in managing heart failure and atrial fibrillation, particularly in cost-sensitive markets or for patients who have failed other therapies, will persist.
- Limited Innovation: The lack of significant innovation or new patent filings directly related to digoxin means there are no foreseeable drivers for substantial price increases or market value shifts akin to branded drug launches.
Projected Price Trend: Marginal fluctuations of +/- 5% annually are anticipated, driven primarily by manufacturing costs and supply chain dynamics. No substantial price increases or decreases are projected that would significantly alter the drug's cost-effectiveness profile.
WHAT ARE THE KEY COMPETITORS AND ALTERNATIVES TO DIGOXIN?
The competitive landscape for digoxin is bifurcated. Within its direct generic form, competition comes from numerous pharmaceutical companies. However, the broader therapeutic space for heart failure and atrial fibrillation involves a range of newer and often branded medications.
Direct Generic Competitors:
Manufacturers of generic digoxin tablets and oral solutions include, but are not limited to:
- Teva Pharmaceuticals
- Amneal Pharmaceuticals
- Apotex
- Lupin Pharmaceuticals
- Various other regional and international generic manufacturers.
Therapeutic Alternatives:
- For Heart Failure with Reduced Ejection Fraction (HFrEF):
- Angiotensin Receptor-Neprilysin Inhibitors (ARNIs): Sacubitril/valsartan (Entresto). [5]
- Angiotensin-Converting Enzyme (ACE) Inhibitors: Lisinopril, enalapril, ramipril.
- Angiotensin II Receptor Blockers (ARBs): Losartan, valsartan, candesartan.
- Beta-Blockers: Carvedilol, metoprolol succinate, bisoprolol. [4]
- Mineralocorticoid Receptor Antagonists (MRAs): Spironolactone, eplerenone.
- Sodium-Glucose Cotransporter-2 Inhibitors (SGLT2is): Dapagliflozin, empagliflozin. [5]
- Diuretics: Furosemide, hydrochlorothiazide.
- For Atrial Fibrillation (Rate Control):
- Beta-Blockers: Metoprolol, atenolol.
- Calcium Channel Blockers (Non-dihydropyridine): Diltiazem, verapamil.
- Other Cardiac Glycosides: Digitoxin (less commonly used due to longer half-life and toxicity risk).
The emergence and adoption of these alternatives, particularly ARNIs and SGLT2is, have shifted treatment guidelines and clinical practice, potentially reducing the overall volume of digoxin prescriptions in certain patient segments. However, digoxin's unique mechanism and low cost ensure its continued role.
WHAT IS THE GLOBAL MARKET SIZE AND DEMAND FOR DIGOXIN?
Quantifying the precise global market size for digoxin is challenging due to its generic nature and fragmented distribution. However, its market value is modest compared to blockbuster drugs. The global market is estimated to be in the tens of millions of USD annually.
Demand is sustained by:
- Developed Markets: Continued use in patients resistant to or intolerant of newer agents, and for rate control in atrial fibrillation. Its low cost makes it a standard of care in many public health systems and hospital formularies.
- Emerging Markets: Higher demand in regions where cost is a primary driver of treatment decisions. Generic availability and affordability make it a critical medication for managing cardiovascular diseases.
While precise figures are proprietary to market research firms, the volume of digoxin prescribed globally remains substantial, ensuring ongoing production by generic manufacturers. The total market value is capped by its price point, preventing it from reaching the multi-billion dollar valuations seen for patented biologics or novel small molecules.
WHAT ARE THE REGULATORY CONSIDERATIONS FOR DIGOXIN?
Digoxin is a well-established drug approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Generic Drug Approval: Manufacturers seeking to market generic digoxin must demonstrate bioequivalence to the reference listed drug (RLD) and meet Good Manufacturing Practice (GMP) standards.
- Pharmacovigilance: As with all approved drugs, digoxin is subject to ongoing pharmacovigilance to monitor for adverse events and ensure its continued safety profile. The narrow therapeutic index necessitates vigilant monitoring for signs of toxicity.
- Manufacturing Standards: Adherence to strict GMP guidelines is critical for all digoxin manufacturers to ensure product quality, purity, and consistency. Regulatory inspections of manufacturing facilities are routine.
- Labeling and Prescribing Information: Regulatory agencies review and approve the labeling for digoxin products, including indications, contraindications, warnings, precautions, and adverse reactions. This information is crucial for safe and effective use.
- No New Chemical Entity Exclusivity: Digoxin is not eligible for New Chemical Entity (NCE) exclusivity, patent term extensions specific to NCEs, or other market exclusivities that could significantly prolong its period of market protection. Any market exclusivity would arise from specific formulation patents, which are limited in scope and duration.
Key Takeaways
- Digoxin's foundational patents expired in the 1980s, establishing a competitive generic market.
- Current market prices for generic digoxin are low, ranging from cents per tablet to approximately $15 per 60 mL oral solution bottle.
- Key market drivers include established efficacy, significant cost-effectiveness, and physician familiarity, balanced against the availability of newer therapeutic alternatives and digoxin's narrow therapeutic index.
- Price projections indicate continued stability with minor annual fluctuations (+/- 5%), driven by manufacturing costs and supply chain factors, with no expectation of significant price increases.
- Direct competition exists among multiple generic manufacturers, while broader therapeutic competition comes from newer classes of drugs for heart failure and atrial fibrillation.
- The global market size for digoxin is modest, estimated in the tens of millions of USD annually, with sustained demand in both developed and emerging markets due to its affordability.
- Regulatory considerations focus on generic approval, GMP compliance, and ongoing pharmacovigilance, with no new patent-driven market exclusivities anticipated.
Frequently Asked Questions
What is the typical lifespan of a drug patent, and how does this impact generic drug availability?
Drug patents, particularly for small molecule drugs like digoxin, typically last 20 years from the filing date. However, effective patent life can be shorter due to the time spent in development and regulatory review. Once patents expire, other companies can manufacture and sell generic versions, leading to increased competition and reduced prices.
How does the narrow therapeutic index of digoxin influence its market positioning?
A narrow therapeutic index means that a small change in dose can lead to a significant increase in toxicity or a decrease in efficacy. This necessitates careful patient selection, monitoring, and dose titration. While it can limit widespread use or appeal for certain patient profiles, it also reinforces digoxin's established role for experienced prescribers who understand these nuances.
Are there any emerging formulations or delivery methods for digoxin that could impact its market?
While incremental patent activity around specific formulations or delivery methods may occur, there are no significant, broad-acting innovations in the pipeline for digoxin that are expected to dramatically alter its market dynamics or pricing. The focus remains on its established generic forms.
How do reimbursement policies by insurance providers affect digoxin's market share?
Reimbursement policies generally favor generic medications due to their cost-effectiveness. Digoxin, being one of the lowest-cost cardiac medications available, is typically well-covered by insurance plans. This ensures its accessibility and continued use, particularly for patients with fixed incomes or high co-insurance requirements for branded drugs.
What impact do supply chain disruptions, such as API shortages, have on digoxin pricing and availability?
API shortages or manufacturing issues, while infrequent for established generics like digoxin, can temporarily impact availability and lead to modest price increases. However, the mature competitive landscape means that if one manufacturer experiences issues, others are often able to increase production to meet demand, mitigating prolonged or severe price hikes.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Patent Term Restoration and Data Protection. Retrieved from https://www.fda.gov/drugs/patent-term-restoration/patent-term-restoration-and-data-protection (General information on patent expiry and generic entry)
[2] Kalckar, S. M. (1946). U.S. Patent No. 2,402,508. Washington, DC: U.S. Patent and Trademark Office.
[3] GoodRx. (n.d.). Digoxin Prices, Coupons & Patient Assistance Programs. Retrieved from https://www.goodrx.com/digoxin (Example pricing data source for generic medications)
[4] Heidenreich, P. A., Bozkurt, B., Denney, J. S., Anand, I. S., Antman, E. M., Betterman, K. J., ... & Yancy, C. W. (2022). 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation, 145(18), e895-e1032. doi:10.1161/CIR.0000000000001063
[5] Heidenreich, P. A., Bozkurt, B., Denney, J. S., Anand, I. S., Antman, E. M., Betterman, K. J., ... & Yancy, C. W. (2022). 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Journal of the American College of Cardiology, 79(14), e133-e255. doi:10.1016/j.jacc.2022.02.017
More… ↓
