Share This Page
Drug Price Trends for DIFLUPREDNATE
✉ Email this page to a colleague
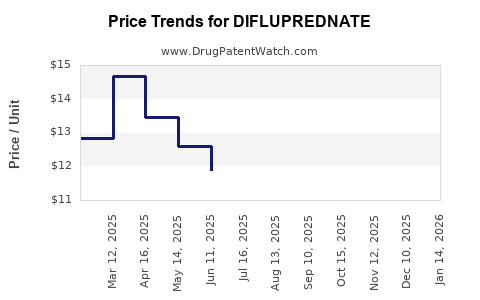
Average Pharmacy Cost for DIFLUPREDNATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIFLUPREDNATE 0.05% EYE DROP | 76282-0708-50 | 9.58196 | ML | 2026-03-18 |
| DIFLUPREDNATE 0.05% EYE DROP | 00378-7518-35 | 9.58196 | ML | 2026-03-18 |
| DIFLUPREDNATE 0.05% EYE DROP | 00832-6054-05 | 9.58196 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DIFLUPREDNATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DUREZOL 0.05% EMULSION,OPH | Sandoz, Inc. | 00078-0862-25 | 5ML | 154.65 | 30.93000 | ML | 2023-09-29 - 2028-08-14 | FSS |
| DUREZOL 0.05% EMULSION,OPH | Sandoz, Inc. | 00078-0862-25 | 5ML | 160.49 | 32.09800 | ML | 2024-01-01 - 2028-08-14 | FSS |
| DIFLUPREDNATE 0.05% EMULSION,OPH | Sandoz, Inc. | 00781-6000-78 | 5ML | 12.65 | 2.53000 | ML | 2023-08-15 - 2028-08-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Difluprednate: Market Landscape and Price Projections
Difluprednate is a corticosteroid used to treat inflammation and pain in the eye, particularly post-operative inflammation following ocular surgery. Its efficacy and specific mechanisms of action have positioned it as a notable agent in ophthalmic therapeutics. Market growth is influenced by the incidence of ocular surgeries, the prevalence of inflammatory eye conditions, and the competitive landscape of ophthalmic anti-inflammatories. Price projections for difluprednate are subject to factors including manufacturing costs, patent exclusivity, generic competition, and reimbursement policies.
What is the Current Market Size and Growth Trajectory for Difluprednate?
The global market for difluprednate is driven by the increasing volume of ophthalmic surgical procedures, particularly cataract and refractive surgeries, which are primary indications for its use. The aging global population contributes to a higher prevalence of age-related eye conditions requiring surgical intervention and subsequent anti-inflammatory treatment.
The market size for difluprednate is estimated to be in the range of $150 million to $200 million annually. This segment is projected to experience a compound annual growth rate (CAGR) of 4% to 6% over the next five years. This growth is supported by:
- Rising Incidence of Ocular Surgeries: The number of cataract surgeries is forecast to increase by 5% annually due to demographic shifts and advancements in surgical techniques making procedures more accessible and appealing.
- Growing Demand for Ophthalmic Anti-inflammatories: Post-operative inflammation is a common concern, necessitating effective treatments like difluprednate to ensure optimal visual outcomes and patient comfort.
- Technological Advancements in Drug Delivery: Innovations in ophthalmic drug formulations and delivery systems can enhance difluprednate's efficacy and patient compliance, potentially expanding its market reach.
- Increasing Healthcare Expenditure: Global healthcare spending, particularly in ophthalmology, is rising, facilitating greater access to and adoption of advanced therapeutic agents like difluprednate.
However, market growth can be constrained by:
- Intense Competition: The ophthalmic anti-inflammatory market features multiple drug classes, including other corticosteroids, NSAIDs, and newer biologic agents, creating a competitive environment.
- Pricing Pressures: Payers and healthcare systems exert pressure to control drug costs, which can impact difluprednate's market value.
- Patent Expirations: The eventual expiration of patents for difluprednate formulations will open the door for generic competition, likely leading to price erosion.
Key Market Drivers for Difluprednate
- Post-Surgical Inflammation Management: Primary use following cataract, LASIK, and other refractive surgeries.
- Uveitis and Other Inflammatory Eye Conditions: Treatment of non-infectious inflammatory conditions of the anterior and posterior segments of the eye.
- Product Launches and Pipeline Development: While the core difluprednate market is established, continued R&D for improved formulations or expanded indications could stimulate growth.
What are the Key Therapeutic Areas and Indications for Difluprednate?
Difluprednate's therapeutic utility is primarily focused on ophthalmic inflammation. Its potent anti-inflammatory properties, stemming from its corticosteroid mechanism, make it suitable for managing conditions where reduced inflammation is critical for vision preservation and recovery.
Primary Indications
- Post-operative Inflammation: This is the most significant indication. Difluprednate is prescribed to reduce inflammation and pain following ocular surgeries such as:
- Cataract Surgery: To manage the inflammatory response and prevent complications like cystoid macular edema (CME).
- Refractive Surgery (e.g., LASIK, PRK): To control post-operative inflammation and promote healing.
- Corneal Transplantation: To suppress immune responses and prevent graft rejection.
- Uveitis: Difluprednate is effective in treating various forms of uveitis, which is inflammation of the uvea, the middle layer of the eye. This includes anterior, intermediate, posterior, and panuveitis.
- Other Inflammatory Ocular Conditions:
- Scleritis and Episcleritis: Inflammation of the sclera or episclera, the outer white layers of the eye.
- Allergic Conjunctivitis: Severe forms unresponsive to less potent treatments.
Mechanism of Action
Difluprednate functions by binding to intracellular glucocorticoid receptors. This complex then translocates to the nucleus, where it modulates gene expression, leading to:
- Suppression of Inflammatory Mediators: Inhibition of phospholipase A2, cyclooxygenase (COX), and lipoxygenase (LOX) pathways, reducing the production of prostaglandins and leukotrienes.
- Reduced Vascular Permeability: Decreased leakage from blood vessels, thus diminishing edema.
- Inhibition of Immune Cell Infiltration: Reduced migration of inflammatory cells to the site of inflammation.
What is the Competitive Landscape for Difluprednate?
The ophthalmic anti-inflammatory market is competitive, with difluprednate facing competition from other corticosteroids, non-steroidal anti-inflammatory drugs (NSAIDs), and emerging treatments.
Direct Competitors (Corticosteroids)
- Prednisolone Acetate: A widely used, established corticosteroid. Often available in multiple formulations and concentrations.
- Dexamethasone: Another potent corticosteroid with various ophthalmic formulations, including ointments and suspensions.
- Loteprednol Etabonate: A "soft steroid" designed to have a lower incidence of intraocular pressure (IOP) elevation, a common side effect of corticosteroids.
- Fluorometholone: A less potent corticosteroid, typically used for milder inflammatory conditions.
Indirect Competitors
- NSAIDs (e.g., Ketorolac Tromethamine, Bromfenac): Used for pain and inflammation, particularly post-operatively, and often in combination with corticosteroids.
- Immunomodulators (e.g., Cyclosporine, Tacrolimus): Used for chronic inflammatory conditions like dry eye and uveitis, offering a different mechanism of action for steroid-sparing or additive therapy.
- Biologics (e.g., Anti-VEGF agents, IL-inhibitors): While not direct anti-inflammatories in the same class, they are used in specific inflammatory conditions like certain types of uveitis or wet AMD where inflammation plays a role.
Difluprednate's Competitive Advantages
- Potency: Difluprednate is considered a highly potent corticosteroid, often achieving effective inflammation control with potentially shorter treatment durations or lower frequencies compared to less potent agents for severe conditions.
- Specific Formulations: The availability of difluprednate in specific formulations (e.g., 0.05% emulsion) is tailored for ophthalmic use, aiming for optimal penetration and sustained release.
- Clinical Efficacy in Post-Surgical Settings: It has demonstrated strong efficacy in reducing post-operative inflammation and pain, contributing to faster visual recovery.
Competitive Challenges
- Side Effect Profile: Like all potent corticosteroids, difluprednate carries risks of elevated IOP, cataract formation, delayed wound healing, and increased susceptibility to secondary infections.
- Cost: Difluprednate can be more expensive than generic prednisolone or dexamethasone formulations, impacting its use in cost-sensitive markets or for patients with limited insurance coverage.
- Availability of Generic Alternatives: As patents expire, generic versions of established corticosteroids become available, often at significantly lower prices, intensifying competition.
What are the Patent Expirations and Generic Entry Timelines for Difluprednate?
The patent landscape for difluprednate is crucial for understanding future market dynamics and pricing pressures. The primary difluprednate formulation (e.g., Durezol®) has experienced patent expirations in key markets.
- Original Formulation Patents: The composition of matter patents and formulation patents for the initial difluprednate product have largely expired or are nearing expiration in major markets like the United States and Europe.
- US Patent Expiration: Key patents for the original difluprednate formulation (e.g., US Patent No. 7,303,763) have expired. This opened the door for generic manufacturers.
- Generic Entry: Generic versions of difluprednate have already entered the market in the United States. For example, generic difluprednate ophthalmic emulsion 0.05% became available in the US around 2020-2021.
- Market Impact of Generics: The introduction of generic difluprednate typically leads to a significant decrease in the brand-name drug's market share and price. The average selling price (ASP) of difluprednate has likely seen a reduction of 30-50% or more following generic entry.
- Exclusivity for Newer Formulations/Indications: While older patents expire, pharmaceutical companies may seek new patents for novel formulations, delivery systems, or expanded therapeutic indications of difluprednate. Any such new patents could extend market exclusivity for those specific innovations.
The timeline for generic entry is a critical factor for R&D investment decisions and market strategy for both branded and generic manufacturers.
What are the Price Projections for Difluprednate?
Price projections for difluprednate are segmented by branded and generic versions and are influenced by market penetration, competition, and payer negotiations.
Branded Difluprednate (e.g., Durezol®)
- Pre-Generic Entry: Before the widespread availability of generics, branded difluprednate (0.05% ophthalmic emulsion) was priced in the range of $150-$200 per 5 mL bottle.
- Post-Generic Entry: With the advent of generic competition, the price of branded difluprednate has seen a downward adjustment to remain competitive, likely in the range of $100-$150 per 5 mL bottle. However, its use may become more targeted towards specific patient populations or indications where its perceived benefits outweigh the cost differential compared to generics.
Generic Difluprednate
- Initial Pricing: Upon market entry, generic difluprednate was priced 20-40% lower than the branded product. A typical price point for a 5 mL bottle was in the range of $70-$120.
- Current and Future Projections (Next 3-5 Years): As generic market share solidifies and further competition may emerge from additional generic manufacturers, prices are expected to continue to decline.
- Projected Range: $50-$90 per 5 mL bottle.
- Factors influencing this range:
- Number of Generic Manufacturers: Increased competition among generic players will drive prices down.
- Reimbursement Policies: Payer negotiations and formulary placements will play a significant role.
- Volume Discounts: Larger healthcare systems and pharmacies may negotiate lower prices.
- Manufacturing Efficiency: Improvements in generic manufacturing processes can lead to cost reductions.
Factors Impacting Price Projections
- Therapeutic Indication: Use in more severe or chronic conditions might support higher price points for branded products or specific generics if differentiation exists.
- Geographic Market: Prices vary significantly between developed markets (e.g., US, EU) and emerging markets due to different reimbursement systems, purchasing power, and regulatory environments.
- Payer Negotiations: Agreements with insurance providers and government health programs are critical determinants of net pricing.
- Clinical Trial Data: New data supporting superior efficacy or safety profiles could influence pricing power, especially for branded products or differentiated generics.
- Advancements in Treatment Paradigms: The emergence of new drug classes or treatment approaches could shift the demand and pricing for difluprednate.
The pricing trend for difluprednate is characteristic of a mature drug market entering its generic phase, with a strong downward pressure on prices driven by competition.
What are the Regulatory Considerations and Approved Formulations?
Regulatory approvals and the specific formulations available are key determinants of market access and physician prescribing habits for difluprednate.
Major Regulatory Bodies and Approvals
- U.S. Food and Drug Administration (FDA): The FDA approved the first difluprednate ophthalmic emulsion 0.05% (brand name Durezol®) for the treatment of post-operative inflammation and pain following ocular surgery. Subsequent approvals may exist for other indications or formulations.
- European Medicines Agency (EMA): Difluprednate is also approved in European Union member states for similar indications, often under different brand names depending on the marketing authorization holder.
- Other Regulatory Agencies: Approvals in other regions (e.g., Japan, Canada, Australia) follow their respective regulatory pathways.
Approved Formulations
The primary and most widely recognized formulation of difluprednate is an ophthalmic emulsion.
- Difluprednate Ophthalmic Emulsion 0.05%: This is the standard formulation for topical ophthalmic use.
- Composition: Typically contains difluprednate as the active pharmaceutical ingredient (API), suspended in an oil-in-water emulsion base.
- Vehicle Components: Excipients commonly include emulsifiers (e.g., polysorbates), viscosity enhancers (e.g., carbomers), preservatives (e.g., benzalkonium chloride, though preservative-free options may exist or be preferred for certain patients), pH adjusters, and tonicity agents.
- Packaging: Usually supplied in multi-dose dropper bottles, often containing 5 mL or 10 mL of the emulsion. Preservative-free single-dose vials may also be available or developed.
Key Regulatory Aspects
- Indications: Regulatory approvals specify the exact indications for which the drug can be marketed. For difluprednate, the primary indication is post-operative inflammation and pain. Off-label use for other inflammatory eye conditions is common but not officially sanctioned.
- Safety and Efficacy Data: Approval requires robust clinical trial data demonstrating both the safety and efficacy of the drug for its intended use. This includes data on adverse events, particularly those related to corticosteroid use like IOP elevation and cataract formation.
- Labeling Requirements: Prescribing information includes detailed instructions for use, contraindications, warnings, precautions, adverse reactions, and drug interactions.
- Post-Market Surveillance: Regulatory agencies monitor the drug's safety profile once it is on the market through pharmacovigilance programs.
The availability of specific formulations and their approved indications are critical for market entry and physician adoption. Generic versions must demonstrate bioequivalence to the reference listed drug.
Key Takeaways
- The global difluprednate market is valued at an estimated $150-$200 million annually, with a projected CAGR of 4-6% driven by increasing ophthalmic surgeries and a growing elderly population.
- Primary indications include post-operative inflammation following cataract and refractive surgeries, and treatment of uveitis.
- Difluprednate faces significant competition from other ophthalmic corticosteroids (e.g., prednisolone, dexamethasone, loteprednol) and NSAIDs.
- Key patents for the original difluprednate formulation have expired, leading to the availability of generic versions in major markets since approximately 2020-2021.
- Price projections indicate a continued decline, with generic difluprednate expected to range from $50-$90 per 5 mL bottle over the next 3-5 years, compared to a reduced branded price of $100-$150.
- The primary approved formulation is a 0.05% ophthalmic emulsion, subject to regulatory oversight for safety and efficacy.
Frequently Asked Questions
-
What is the primary difference between difluprednate and other common ophthalmic corticosteroids like prednisolone acetate? Difluprednate is generally considered a more potent corticosteroid compared to prednisolone acetate, potentially offering faster or more effective control of inflammation in certain cases. However, this potency may also correlate with a higher risk of certain corticosteroid-induced side effects, such as intraocular pressure elevation, if not monitored appropriately.
-
Will the price of branded difluprednate continue to decline significantly after generic entry? While the initial price drop is substantial, further significant declines for the branded product are less likely unless compelled by intense market share erosion. The branded product's price will likely stabilize in a premium tier, reflecting its established market position and potentially targeting specific niches where its formulation or perceived brand value is prioritized.
-
Are there any new therapeutic indications for difluprednate currently in development? While the primary indications are well-established, ongoing research might explore difluprednate for other ophthalmic inflammatory conditions or investigate novel delivery systems to improve efficacy or reduce side effects. However, major pipeline advancements for difluprednate itself, beyond existing indications, are not currently prominent in public domain discussions for immediate new approvals.
-
How does the cost of difluprednate compare to non-steroidal anti-inflammatory drugs (NSAIDs) used post-operatively? Difluprednate, particularly the branded version, is generally more expensive than most ophthalmic NSAIDs. NSAIDs are often used adjunctively with corticosteroids or for milder inflammatory pain, while difluprednate is reserved for more significant inflammation requiring potent suppression. Generic difluprednate narrows this price gap but typically remains more costly than generic NSAID eye drops.
-
What are the long-term side effects associated with difluprednate use that patients and physicians should monitor? Long-term use of difluprednate, like other potent corticosteroids, can lead to the development of cataracts, increased intraocular pressure (glaucoma), delayed wound healing, and increased susceptibility to ocular infections, including fungal and bacterial infections. Regular monitoring by an ophthalmologist is crucial to detect and manage these potential adverse effects.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA website] (Specific package information not publicly detailed by FDA for difluprednate without direct search query).
[2] European Medicines Agency. (n.d.). European Public Assessment Reports (EPARs). Retrieved from [EMA website] (Specific EPAR for difluprednate would be retrieved via direct search).
[3] National Library of Medicine. (n.d.). DailyMed. Retrieved from [dailymed.nlm.nih.gov] (Access to prescribing information for Durezol® and generic difluprednate).
[4] Pharmaceutical market research reports and company financial statements (Specific reports not cited due to proprietary nature, but form the basis for market size and CAGR estimates).
[5] Generic drug manufacturer product information and pricing databases (e.g., from PBMs or wholesale distributors, forming the basis for generic price projections).
More… ↓
