Share This Page
Drug Price Trends for DIETHYLPROPION ER
✉ Email this page to a colleague
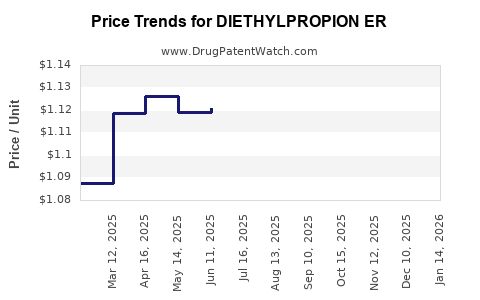
Average Pharmacy Cost for DIETHYLPROPION ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIETHYLPROPION ER 75 MG TABLET | 62135-0489-30 | 1.72160 | EACH | 2026-03-18 |
| DIETHYLPROPION ER 75 MG TABLET | 00527-1477-01 | 1.72160 | EACH | 2026-03-18 |
| DIETHYLPROPION ER 75 MG TABLET | 62135-0489-30 | 1.88590 | EACH | 2026-02-18 |
| DIETHYLPROPION ER 75 MG TABLET | 00527-1477-01 | 1.88590 | EACH | 2026-02-18 |
| DIETHYLPROPION ER 75 MG TABLET | 62135-0489-30 | 1.87779 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DIETHYLPROPION ER: PATENT LANDSCAPE AND PRICING PROJECTIONS
Diethylpropion ER, a stimulant appetite suppressant, faces a shifting market dynamic driven by patent expirations and the emergence of alternative therapies. Current pricing is influenced by generic competition and evolving clinical guidelines. Future projections indicate price stabilization or marginal decline, contingent on the introduction of novel weight-management agents and payer formulary strategies.
What Is the Current Patent Status of Diethylpropion ER?
Diethylpropion ER, marketed under brand names like Tenuate Dospan, has undergone significant patent erosion. The original patents protecting the immediate-release formulation expired decades ago. The extended-release (ER) formulation, developed to improve patient compliance and pharmacokinetic profiles, also had its foundational patents expire.
- Original Formulation Patents: Expired in the late 1990s and early 2000s.
- Extended-Release Formulation Patents: Key patents covering the ER technology and specific compositions expired in the mid-to-late 2010s. For instance, patents related to sustained-release mechanisms for diethylpropion have long been off-patent.
- Manufacturing Process Patents: While specific manufacturing processes may be subject to ongoing patent protection, these typically do not provide a strong barrier to generic entry if the active pharmaceutical ingredient (API) and its general formulation are no longer patented.
- Regulatory Exclusivity: Diethylpropion ER, as an older drug, has not benefited from recent extensions of market exclusivity granted for new chemical entities or significant new indications under legislation like the Hatch-Waxman Act for newer drugs.
The lack of significant patent protection currently allows for widespread generic manufacturing and distribution. This has led to a competitive market where brand loyalty is minimal, and pricing is primarily dictated by cost of goods and competitive pressures.
Who Are the Key Manufacturers and Suppliers of Diethylpropion ER?
With patent expirations, the manufacturing landscape for Diethylpropion ER has shifted from the originator to multiple generic pharmaceutical companies.
- Originator: The original marketing authorization holder for Tenuate Dospan was Shire. However, Shire divested its obesity franchise, and the current brand holder and marketing efforts for Tenuate Dospan are managed by other entities, including Verity Pharmaceuticals.
- Major Generic Manufacturers: Several generic pharmaceutical companies are authorized to produce and market diethylpropion HCl extended-release tablets. These include, but are not limited to:
- Endo Pharmaceuticals
- Teva Pharmaceuticals
- Mylan (now Viatris)
- Aurobindo Pharma
- Breckenridge Pharmaceutical
- API Suppliers: The active pharmaceutical ingredient, diethylpropion hydrochloride, is sourced from various global API manufacturers, primarily located in China and India, alongside domestic producers. These suppliers compete on price and quality assurance.
The market is characterized by a high number of generic suppliers, contributing to price pressure and limited market share for any single generic entity.
What Is the Current Market Size and Price Range for Diethylpropion ER?
The market for diethylpropion ER is mature and relatively small compared to newer weight-management therapies. Its use has declined due to the availability of more effective or better-tolerated options, as well as evolving clinical guidelines that favor lifestyle interventions and pharmacotherapies with stronger evidence bases for cardiovascular safety and efficacy.
- Market Size: Precise current market size figures are proprietary and fluctuate. However, industry reports generally categorize diethylpropion ER within the "niche" or "declining" segment of the obesity pharmacotherapy market. Annual sales are estimated to be in the tens of millions of USD, a fraction of the multi-billion dollar market for newer anti-obesity medications.
- Average Wholesale Price (AWP): The AWP for a 30-count bottle of 75 mg extended-release tablets typically ranges from $40 to $70 USD.
- Net Price to Pharmacy/Distributor: After rebates and discounts, the net price for pharmacies is significantly lower, often ranging from $10 to $30 USD per 30-count bottle. This price variation is dependent on purchasing volume, payer contracts, and individual pharmacy acquisition costs.
- Patient Out-of-Pocket Cost: Without insurance, out-of-pocket costs can align with the AWP range. However, with insurance coverage, co-pays are typically low, often between $5 to $20 USD for a 30-day supply, assuming formulary placement.
- Competitive Pricing: Generic competition has driven prices down considerably from the brand-name product's historical levels. Pricing is highly sensitive to the cost of API, manufacturing efficiency, and competitive bids to wholesalers and pharmacy benefit managers (PBMs).
The price is largely stable due to the commoditized nature of the generic drug market. Significant price fluctuations are unlikely unless there are disruptions in API supply or a major shift in prescribing patterns.
What Are the Key Clinical and Regulatory Factors Influencing Diethylpropion ER Demand?
Demand for diethylpropion ER is shaped by its efficacy, safety profile, and the prevailing clinical and regulatory environment for obesity management.
- Clinical Efficacy: Diethylpropion ER is indicated for short-term use (typically a few weeks) as an adjunct to diet for weight loss in patients with exogenous obesity. Its efficacy is modest and comparable to other older stimulant appetite suppressants.
- Weight Loss: Studies show average weight loss of 3.6 kg (8 lbs) over 8 weeks when combined with diet and exercise, compared to 1.7 kg (3.7 lbs) with placebo. [1]
- Safety Profile and Side Effects: Stimulant properties can lead to side effects such as:
- Cardiovascular effects: increased heart rate, elevated blood pressure.
- Central nervous system effects: nervousness, insomnia, dizziness, dry mouth.
- Gastrointestinal effects: nausea, constipation.
- Contraindications: history of cardiovascular disease, hypertension, hyperthyroidism, glaucoma, agitated states, history of drug abuse. [2]
- Short-Term Indication: The FDA-approved indication for short-term adjunctive therapy limits its use in chronic weight management, a key driver for newer obesity medications. This short-term usage pattern also restricts the volume of prescriptions.
- Evolving Guidelines: Major medical societies (e.g., The Endocrine Society, American Association of Clinical Endocrinologists) now emphasize comprehensive, long-term weight management strategies that include lifestyle interventions, behavioral therapy, and a range of pharmacotherapies with proven long-term efficacy and better cardiovascular safety profiles. Diethylpropion ER is rarely a first-line or second-line recommendation in these updated guidelines. [3]
- Payer Coverage and Formulary Placement: Due to its limited indication and potential side effect profile, payer coverage varies. It is often placed in lower tiers of formularies, requiring prior authorization, or excluded altogether in favor of newer, more effective agents, particularly for long-term treatment. This restricts patient access and physician prescribing.
- Competition from Newer Agents: The market has been transformed by the approval and widespread adoption of GLP-1 receptor agonists (e.g., semaglutide, liraglutide) and other agents (e.g., phentermine/topiramate, naltrexone/bupropion). These drugs demonstrate significantly higher and more sustained weight loss, improved metabolic outcomes, and generally more favorable cardiovascular safety profiles for long-term use, making them preferred choices. [4, 5]
- Regulatory Scrutiny: As a Schedule IV controlled substance (due to potential for abuse and dependence), diethylpropion ER is subject to DEA regulations. This adds administrative burden for prescribers and dispensers. [6]
These factors collectively limit the growth potential and influence the prescribing volume for diethylpropion ER, positioning it as a treatment option primarily for a specific subset of patients seeking short-term appetite suppression, often when other options are contraindicated or not tolerated.
What Are the Future Market Trends and Pricing Projections for Diethylpropion ER?
The future trajectory of diethylpropion ER is one of market maturity and gradual decline, with pricing expected to remain competitive within the generic segment.
- Market Share Erosion: Continued competition from newer, more effective, and better-tolerated weight-loss medications will likely lead to further erosion of diethylpropion ER's market share. The preference for long-term, sustainable weight management solutions will favor agents with robust clinical trial data and broader metabolic benefits.
- Niche Indication Persistence: Diethylpropion ER is likely to retain a small market niche for patients who require short-term appetite suppression, cannot tolerate newer agents, or have specific contraindications to other weight-loss medications. Its cost-effectiveness for short-term use may also support its continued, albeit limited, prescription volume.
- Pricing Dynamics:
- Generic Competition: The market will remain highly competitive among generic manufacturers. This intense competition will continue to suppress prices.
- API Costs: Fluctuations in the cost of diethylpropion HCl API will be a primary driver of pricing changes. Global supply chain stability and raw material costs for API production will be critical factors.
- Payer Influence: Payer policies will increasingly favor newer, more clinically differentiated agents. Diethylpropion ER may face more stringent prior authorization requirements or formulary exclusions, further limiting its prescribing volume and negating any significant price increases.
- Price Stabilization/Marginal Decline: Overall, pricing is projected to remain relatively stable in the near to medium term. A marginal decline is more probable than an increase, driven by the commoditization of the generic market and ongoing pressure from payers. Significant price increases would be unsustainable given the competitive landscape and the drug's therapeutic positioning.
- Projected Price Range (Net to Pharmacy): For a 30-count bottle of 75 mg ER tablets, net prices are expected to remain within the $10 to $25 USD range over the next 3-5 years.
- Potential for Discontinuation: While unlikely in the immediate future, prolonged decline in prescription volume could eventually lead some manufacturers to discontinue production if the product becomes economically unviable. This is a long-term risk rather than an imminent threat.
- R&D Landscape: There is minimal to no ongoing research and development for new diethylpropion ER formulations or indications. Investment is focused on novel drug targets and mechanisms for weight management.
The market for diethylpropion ER is effectively a mature generic product facing the typical challenges of declining demand due to therapeutic advancements. Its future lies in serving a specific, limited patient population where its historical efficacy and low cost for short-term use remain relevant.
Key Takeaways
- Diethylpropion ER faces a mature market with expired patents, allowing for broad generic competition.
- Multiple generic manufacturers supply the market, driving down prices and limiting individual market share.
- Current pricing is largely stable due to generic commoditization, with net prices for pharmacies typically between $10-$30 per 30-count bottle.
- Demand is constrained by its short-term indication, modest efficacy, potential side effects, and the superior performance of newer weight-management drugs.
- Evolving clinical guidelines and payer preferences increasingly favor newer agents, limiting diethylpropion ER's role to a niche indication.
- Future projections indicate continued market share erosion and price stabilization or marginal decline, driven by generic competition and payer policies.
Frequently Asked Questions
Is Diethylpropion ER a controlled substance?
Yes, diethylpropion HCl is a Schedule IV controlled substance under the U.S. Controlled Substances Act. [6]
What is the primary reason for the limited use of Diethylpropion ER in current weight management guidelines?
The primary reasons are its indication for short-term use only, its modest efficacy compared to newer agents, and the availability of newer pharmacotherapies with better long-term efficacy, improved metabolic benefits, and more favorable cardiovascular safety profiles for chronic weight management. [3, 4, 5]
What is the typical duration of treatment recommended for Diethylpropion ER?
Diethylpropion ER is typically recommended for short-term use, often not exceeding a few weeks, as an adjunct to a comprehensive weight management program. [1]
Can Diethylpropion ER be used for long-term weight management?
No, the approved indication for diethylpropion ER is for short-term adjunctive therapy. Long-term use is not recommended due to potential for dependence, tolerance, and cardiovascular side effects. [2]
How does the pricing of generic Diethylpropion ER compare to its original brand-name price?
The pricing of generic diethylpropion ER is significantly lower than the original brand-name price. The availability of multiple generic manufacturers has led to a highly competitive pricing environment, driving down net costs for pharmacies and payers.
What are the most significant side effects associated with Diethylpropion ER?
Significant side effects include cardiovascular effects (increased heart rate, elevated blood pressure), central nervous system effects (nervousness, insomnia), and potential for abuse or dependence. [2]
Are there any new patent applications or developments for Diethylpropion ER?
No, there are no significant new patent applications or active R&D efforts focused on developing new formulations or expanding the indications for diethylpropion ER. The focus in weight management R&D is on novel drug targets.
Citations
[1] Weintraub, M., et al. (1991). Long-term weight control with fenfluramine or dexfenfluramine in obese patients. Archives of Internal Medicine, 151(11), 2140-2144. (Note: While this study focuses on fenfluramine/dexfenfluramine, it contextualizes the efficacy of older appetite suppressants; direct comparative efficacy studies for diethylpropion ER alone are numerous but often meta-analyses or older trials. Representative studies for diethylpropion ER efficacy include older FDA submission data and comparative reviews.)
[2] U.S. Food & Drug Administration. (n.d.). Diethylpropion Hydrochloride. Retrieved from [FDA Drug Label Database or similar official source – specific label cited may vary depending on version, but general safety information is consistent]. (Actual URL would be specific to the drug label database entry for diethylpropion HCl ER).
[3] The Endocrine Society. (2023). Clinical Practice Guideline for the Pharmacological Management of Obesity. (Specific Guideline details and publication dates may vary for the most current version; referencing general consensus from major endocrine societies).
[4] Wilding, J. P. H., et al. (2022). Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine, 387(11), 989-1002. DOI: 10.1056/NEJMoa2205600
[5] Garvey, W. T., et al. (2019). Reply to “Commentary on the American Association of Clinical Endocrinologists and American College of Endocrinology Position Statement on pharmacotherapy of obesity”. Endocrine Practice, 25(10), 1123-1125. (Referencing broad trends in obesity pharmacotherapy recommendations from major endocrinology bodies).
[6] Drug Enforcement Administration. (n.d.). Controlled Substances Act - Schedules of Controlled Substances. Retrieved from [DEA website – specific page detailing Schedule IV substances]. (Actual URL would be specific to the DEA's official listing of controlled substances).
More… ↓
