Share This Page
Drug Price Trends for DIETHYLPROPION
✉ Email this page to a colleague
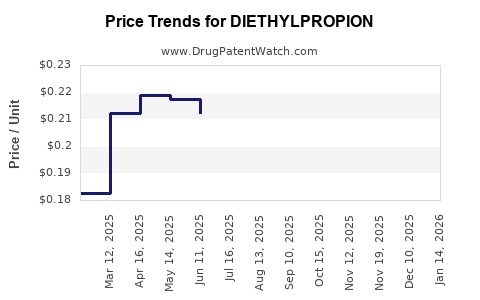
Average Pharmacy Cost for DIETHYLPROPION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIETHYLPROPION ER 75 MG TABLET | 62135-0489-30 | 1.72160 | EACH | 2026-03-18 |
| DIETHYLPROPION 25 MG TABLET | 00527-1475-01 | 0.25065 | EACH | 2026-03-18 |
| DIETHYLPROPION 25 MG TABLET | 10702-0044-01 | 0.25065 | EACH | 2026-03-18 |
| DIETHYLPROPION ER 75 MG TABLET | 00527-1477-01 | 1.72160 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DIETHYLPROPION Market Analysis and Price Projections
Diethylpropion is an anorectic drug used for weight loss in obesity management. It is marketed primarily as a prescription medication in combination with diet and exercise. Its patent expirations and generic availability influence market dynamics and pricing.
Market Overview
Market Size and Demand Drivers
The global weight management drug market was valued at approximately USD 3.2 billion in 2022, projected to reach USD 5.4 billion by 2030, with a compound annual growth rate (CAGR) of 6.9%.[1] Diethylpropion accounts for a niche segment within the appetite suppressant category, primarily used in the United States and select markets.
Regulatory Status
In the U.S., diethylpropion is classified as a Schedule IV controlled substance, issued by the DEA, which limits prescribing and distribution.[2]. It is available under generic names, with brand equivalents such as Tenuate. Other markets may have varying regulatory controls.
Competitive Landscape
Market competitors include phentermine, phendimetrazine, and lorcaserin. The drug's lifecycle has been affected by safety concerns—most notably, the withdrawal of fen-phen in the late 1990s and regulatory scrutiny over sympathomimetic agents.
Patent and Patent Expiry
Diethylpropion's patent expired in the early 2000s, leading to a wide availability of generic versions across markets. The absence of patent protection constrains pricing power.
Pricing Analysis
Current Pricing Trends
In the U.S., wholesale acquisition costs (WAC) for a 30-day supply (30 tablets of 75 mg) average about USD 20–USD 25 for generic formulations.[3]. Retail prices can vary based on pharmacy discounting, insurance coverage, and distribution channels.
| Parameter | Value | Notes |
|---|---|---|
| Price per 30 tablet pack | USD 20 – USD 25 | For generic diethylpropion |
| Cost per tablet | USD 0.67 – USD 0.83 | Based on pack price and tablet count |
| Annual treatment cost | USD 240 – USD 300 | Assuming continuous monthly therapy |
Price Differentials by Region
- United States: Lower list prices due to market competition but higher utilization rates.
- Europe and Asia: Prices are generally lower, ranging between USD 10–USD 20 per 30-tablet pack due to different regulatory environments and market sizes.
Factors Influencing Price Changes
- Patent and generic availability: No new patents, prices primarily dictated by market competition.
- Regulatory updates: Safety alerts or scheduling adjustments may impact demand and prices.
- Supply chain factors: Manufacturing costs are stable but supply disruptions can cause temporary price fluctuations.
Future Price Projections
Short-term (Next 2 Years)
- Prices are expected to remain stable, with minor variations linked to inflation, supply chain stability, and healthcare policy changes.
- Generic competition will sustain low prices, averaging USD 20–USD 25 per 30-tablet pack.
Mid-term (3–5 Years)
- Market growth driven by increasing obesity rates could marginally increase demand, but the impact on prices remains limited due to the commoditized nature of the drug.
- Prices may fluctuate between USD 20–USD 30 depending on regional regulatory policies and healthcare provider prescribing patterns.
Long-term (5+ Years)
- No significant price hikes anticipated unless patent filings or reformulations emerge.
- The rise of combination therapies may reduce diethylpropion demand, exerting downward pressure on individual product prices.
Impact of New Market Entrants and Alternatives
- Development of novel appetite suppressants with improved safety profiles could shift demand away from diethylpropion.
- Prescription trends towards combination therapies (e.g., phentermine/topiramate) may further suppress demand and pricing.
Key Market Dynamics Summary
- Price stability due to patent expiration and generic competition.
- Regional price disparities influenced by healthcare systems.
- Limited potential for significant price increases unless regulatory and market shifts occur.
Key Takeaways
- Diethylpropion revenue largely driven by generic sales, with prices stable over the next 2–3 years.
- The market's growth is tied to the broader obesity treatment landscape, which is expanding.
- Price erosion may occur if new, safer appetite suppressants gain regulatory approval and market share.
- Regulatory status significantly impacts availability and pricing in different markets.
FAQs
1. Is diethylpropion an under patent?
Yes, patent expiration occurred in the early 2000s, leading to widespread generic availability.
2. How do regulatory changes affect its price?
Stricter regulations or scheduling can limit prescribing, reduce supply, and potentially impact prices positively or negatively based on market response.
3. What is the typical wholesale cost per pack?
Approximately USD 20–USD 25 for a 30-tablet pack of 75 mg generic.
4. Are there regional price differences?
Yes, prices vary based on country-specific healthcare policies, insurance coverage, and supply chain factors.
5. What is the outlook for diethylpropion's market share?
Given competition from newer agents and combination therapies, its share may decline unless safety concerns limit alternative options.
References
[1] MarketsandMarkets. (2022). Weight Management Market by Product, Therapeutic Area, Distribution Channel, and Region—Forecast to 2030.
[2] DEA. (2022). Drug Schedules List.
[3] GoodRx. (2023). Diethylpropion Prices and Savings Tips.
More… ↓
