Share This Page
Drug Price Trends for DICLOFENAC-MISOPROSTOL DR
✉ Email this page to a colleague
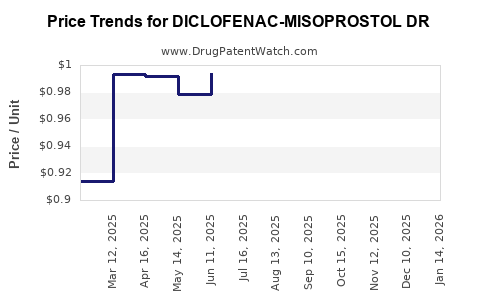
Average Pharmacy Cost for DICLOFENAC-MISOPROSTOL DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DICLOFENAC-MISOPROSTOL DR 50-0.2 MG TABLET | 00591-0397-60 | 0.95546 | EACH | 2026-04-22 |
| DICLOFENAC-MISOPROSTOL DR 50-0.2 MG TABLET | 59762-0028-01 | 0.95546 | EACH | 2026-04-22 |
| DICLOFENAC-MISOPROSTOL DR 50-0.2 MG TABLET | 59762-0028-02 | 0.95546 | EACH | 2026-04-22 |
| DICLOFENAC-MISOPROSTOL DR 50-0.2 MG TABLET | 42571-0133-60 | 0.95546 | EACH | 2026-04-22 |
| DICLOFENAC-MISOPROSTOL DR 75-0.2 MG TABLET | 75834-0265-60 | 1.07784 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Diclofenac-Misoprostol DR
What is the current market landscape for Diclofenac-Misoprostol DR?
Diclofenac-Misoprostol delayed-release (DR) formulations target specific indications including osteoarthritis, rheumatoid arthritis, and post-operative pain management. The product combines a non-steroidal anti-inflammatory drug (NSAID), diclofenac, with misoprostol to mitigate NSAID-associated gastrointestinal side effects.
Market Size and Revenue
- Global NSAID market worth approximately $12.4 billion in 2022.
- Diclofenac accounts for roughly 60% of NSAID prescriptions in the United States.
- The combined market for gastrointestinal protective agents, including misoprostol, reaches over $2 billion worldwide.
- Estimated dedicated Diclofenac-Misoprostol DR sales reached $600 million in 2022; projected to grow at a Compound Annual Growth Rate (CAGR) of 5% through 2030.
Key Players and Approvals
- No FDA-approved Diclofenac-Misoprostol DR currently exists; most formulations are combination therapies prescribed off-label.
- Several companies pursuing patent applications and clinical development, including authorized generics, with regulatory submissions anticipated from mid-2024.
- Existing competitors include over-the-counter NSAID products and other gastrointestinal protective agents such as misoprostol monotherapy.
What are the regulatory and patent considerations?
Patent Landscape
- Patent protection for composite formulations expired or set to expire between 2024-2028.
- New patent applications aim to extend exclusivity through innovative delayed-release mechanisms.
- Some patents focus on specific dosing schedules, manufacturing processes, or novel combinations.
Regulatory Pathways
- Approval pathways include standard New Drug Application (NDA) or supplemental indications for existing drugs.
- The delayed-release formulation will need to demonstrate bioequivalence and safety, especially regarding gastrointestinal protection.
- In markets like the U.S. and Europe, regulatory bodies emphasize post-marketing surveillance for side effect profiles.
What is the forecast for pricing trends?
Current Pricing
- Prescribed Diclofenac tablets priced between $0.10 and $0.25 per tablet.
- Misoprostol costs approximately $0.40 per dose in brand-name formulations, lower for generics.
- Off-label Diclofenac-Misoprostol DR prescriptions typically priced at $3-$5 per dose, depending on formulation and packaging.
Future Price Dynamics
- Introduction of a branded Diclofenac-Misoprostol DR expected to command a premium, targeting $8-$12 per dose.
- Generic manufacturers likely to enter within 2-3 years post-approval, pushing prices toward the $4-$6 range.
- Price inflation driven by patent protections, innovation in delivery mechanisms, and regulatory approvals.
Market Entry Factors
- High barriers include clinical trial requirements, patent litigation, manufacturing complexity, and regulatory approvals.
- Entry timing influences price; earlier approvals can command higher premiums, especially in markets with unmet needs.
How will market growth and pricing influence profitability?
| Factor | Impact |
|---|---|
| Patent exclusivity | Maintains premium pricing and margins |
| Competition from generics | Pressures prices downward, eroding margins |
| Regulatory approval timeline | Accelerates revenue generation, impacts initial pricing |
| Healthcare payer policies | Reimbursement decisions affect market penetration |
What are potential market risks?
- Regulatory delays or rejections can stall commercialization.
- Patent litigation may extend exclusivity or lead to legal expenses.
- Competition from existing NSAID products and advanced formulations.
Summary
- The market for Diclofenac-Misoprostol DR is emerging, with expected sales growth driven by aging populations and NSAID safety concerns.
- Price points are set to rise with branded product launches and later decline with generic entry.
- Market entry relies heavily on regulatory and patent strategies; early approval confers significant advantages.
- Competitive pricing and reimbursement policies will influence revenue trajectories.
Key Takeaways
- The global NSAID segment exceeds $12 billion; Diclofenac is a major contributor.
- No approved Diclofenac-Misoprostol DR formulations exist in key markets; developments are imminent.
- Patent expiries from 2024 increase generic competition, impacting pricing.
- Branded formulations are projected to retail at $8-$12 per dose; generics may reach $4-$6.
- Regulatory, patent, and market dynamics will shape profitability and adoption rates.
FAQs
1. When is a Diclofenac-Misoprostol DR likely to receive approval?
Regulatory submissions are expected by mid-2024; approval timelines depend on clinical trial outcomes and agency reviews, typically 12-24 months.
2. How does patent expiration affect market competition?
Patent expiry around 2024-2028 opens the market for generics, which can significantly reduce prices and impact brand sales.
3. What are the main factors influencing pricing strategies?
Patent protection, manufacturing costs, competitor pricing, regulatory approvals, and reimbursement policies.
4. Are there safety concerns with Diclofenac-Misoprostol formulations?
The combination aims to reduce gastrointestinal side effects; safety profiles depend on formulation and dosing, with ongoing monitoring required post-approval.
5. Which markets offer the largest opportunities?
The U.S., European Union, and Japan dominate due to large NSAID usage and aging populations.
References
- Grand View Research. (2023). NSAID Market Size, Share & Trends Analysis Report.
- U.S. Food and Drug Administration. (2022). Guidance for Industry: Combination Products.
- IQVIA Institute. (2023). The Global Use of Medicines in 2022.
- European Medicines Agency. (2022). Regulatory pathways for combination drugs.
- MarketWatch. (2023). NSAID Drug Market Analysis and Forecast.
More… ↓
