Share This Page
Drug Price Trends for DICLOFENAC SOD EC
✉ Email this page to a colleague
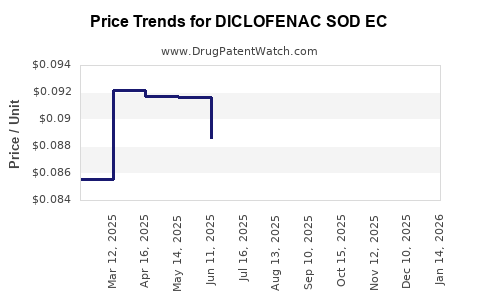
Average Pharmacy Cost for DICLOFENAC SOD EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DICLOFENAC SOD EC 50 MG TAB | 00228-2550-11 | 0.07036 | EACH | 2026-03-18 |
| DICLOFENAC SOD EC 50 MG TAB | 00228-2550-06 | 0.07036 | EACH | 2026-03-18 |
| DICLOFENAC SOD EC 75 MG TAB | 16571-0201-50 | 0.06933 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Diclofenac Sodium EC: Market Dynamics and Price Outlook
Diclofenac Sodium EC, a nonsteroidal anti-inflammatory drug (NSAID), is a mature but consistently utilized pharmaceutical ingredient. Its market is primarily driven by established indications for pain and inflammation management, particularly in osteoarthritis and rheumatoid arthritis. Price projections are influenced by raw material costs, generic competition, and manufacturing efficiencies.
What is Diclofenac Sodium EC?
Diclofenac sodium is a non-steroidal anti-inflammatory drug (NSAID) that works by inhibiting cyclooxygenase (COX) enzymes, thereby reducing the production of prostaglandins, which are key mediators of pain, inflammation, and fever [1]. The "EC" designation typically refers to enteric-coated formulations, designed to protect the stomach lining from irritation, a common side effect of NSAIDs, and to delay drug release until it reaches the small intestine for absorption [2].
Diclofenac sodium is available in various dosage forms, including oral tablets, capsules, suppositories, and topical gels and patches. The enteric-coated tablet is a prevalent oral formulation.
Current Market Landscape for Diclofenac Sodium EC
The market for diclofenac sodium EC is characterized by its long-standing presence and widespread therapeutic use.
Key Therapeutic Areas and Indications
Diclofenac sodium EC is primarily prescribed for the symptomatic treatment of:
- Osteoarthritis: Management of pain and inflammation associated with joint degeneration.
- Rheumatoid Arthritis: Reduction of pain, stiffness, and swelling in inflammatory joint disease.
- Ankylosing Spondylitis: Alleviation of pain and inflammatory symptoms.
- Other Musculoskeletal Conditions: Treatment of conditions such as acute gout and pain from soft tissue injuries [3].
Market Segmentation
The market can be segmented by:
- Formulation: Enteric-coated tablets, delayed-release capsules, extended-release formulations.
- Indication: Osteoarthritis, rheumatoid arthritis, pain management.
- Distribution Channel: Hospital pharmacies, retail pharmacies, online pharmacies.
- Geography: North America, Europe, Asia Pacific, Latin America, Middle East & Africa.
Competitive Landscape
The diclofenac sodium EC market is highly competitive, dominated by generic manufacturers. Key players focus on cost-effective production, supply chain reliability, and broad geographical reach. The intellectual property landscape for the active pharmaceutical ingredient (API) itself is largely expired, meaning innovation centers on formulation improvements or combination products.
Major generic manufacturers with significant presence include:
- Mylan N.V. (now Viatris): A substantial producer of generic NSAIDs, including diclofenac.
- Teva Pharmaceutical Industries Ltd.: A global leader in generic drug manufacturing with a comprehensive portfolio.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with a strong generics segment.
- Sun Pharmaceutical Industries Ltd.: Another major Indian pharmaceutical company with a significant global footprint in generics.
- Bayer AG: While historically a developer of branded NSAIDs, Bayer also participates in the generics market through its affiliates or legacy products.
These companies compete based on price, quality, regulatory compliance, and distribution networks.
Regulatory Environment
Diclofenac sodium EC is approved by regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Manufacturers must adhere to Good Manufacturing Practices (GMP) and maintain rigorous quality control standards. The safety profile of NSAIDs, including diclofenac, is subject to ongoing scrutiny, particularly regarding cardiovascular and gastrointestinal risks, which can influence prescribing patterns and market demand [4].
Factors Influencing Diclofenac Sodium EC Prices
The pricing of diclofenac sodium EC is influenced by a confluence of supply-side and demand-side factors.
Raw Material Costs
The primary raw material for diclofenac synthesis is typically 2,6-dichloroaniline and phenylacetic acid derivatives. Fluctuations in the prices of these petrochemical-derived intermediates, driven by global chemical market dynamics, crude oil prices, and supply chain disruptions, directly impact API production costs. For example, a sustained increase in petrochemical feedstock prices can lead to higher diclofenac API costs, which are then passed on to formulators and, ultimately, to end-users.
Manufacturing and Production Costs
Manufacturing diclofenac sodium EC involves several chemical synthesis steps and formulation processes. These include:
- API Synthesis: Multi-step chemical reactions.
- Purification: Ensuring high purity of the active ingredient.
- Enteric Coating: Application of polymer coatings to tablets to control drug release.
- Packaging: Blister packs, bottles, etc.
Energy costs for manufacturing facilities, labor expenses, and compliance with stringent environmental regulations add to the overall production cost. Economies of scale achieved by large-volume manufacturers can lead to lower per-unit production costs.
Generic Competition
The market for diclofenac sodium EC is saturated with generic alternatives. The expiration of patents for the original diclofenac formulations and the active ingredient has enabled numerous generic manufacturers to enter the market. This intense competition exerts downward pressure on prices. When new generic entrants receive regulatory approval, they often introduce products at lower price points to gain market share.
Supply Chain and Logistics
The global nature of pharmaceutical manufacturing means that supply chain efficiency and logistics play a critical role in pricing. Disruptions due to geopolitical events, trade disputes, or pandemics can affect the availability and cost of raw materials, intermediates, and finished products. Efficient logistics reduce transportation costs and minimize inventory holding periods, contributing to competitive pricing.
Regulatory Changes and Pharmacovigilance
Post-market surveillance and pharmacovigilance activities by regulatory bodies can impact the perceived safety of NSAIDs. If new safety concerns emerge regarding diclofenac (e.g., increased risk of cardiovascular events), this could lead to revised prescribing guidelines or warnings, potentially affecting demand. Regulatory requirements for manufacturing facility inspections and quality assurance also add to compliance costs for manufacturers.
Healthcare Policies and Reimbursement
Government healthcare policies, national drug formularies, and reimbursement rates significantly influence pricing. In countries with centralized healthcare systems, government agencies often negotiate drug prices directly with manufacturers or set price ceilings. Payer policies in private healthcare markets also dictate the extent to which diclofenac sodium EC is covered, impacting its market uptake and price sensitivity.
Demand from End-Users
The demand for diclofenac sodium EC is largely driven by the prevalence of inflammatory and pain-related conditions, particularly among aging populations. While new therapeutic agents for pain management are developed, diclofenac sodium EC remains a cost-effective first-line or adjunctive treatment option for many. The relative affordability and efficacy of generic diclofenac sodium EC contribute to its sustained demand.
Price Projections for Diclofenac Sodium EC
Predicting precise price trajectories for generic pharmaceuticals is challenging due to the dynamic nature of market forces. However, several trends suggest the likely direction of pricing for diclofenac sodium EC.
Historical Price Trends
Historically, prices for mature generic drugs like diclofenac sodium EC have tended to decrease over time, driven by increasing competition and optimization of manufacturing processes. Initial generic launches often occur at prices significantly lower than branded equivalents, and subsequent market entry by additional generics further erodes prices.
Current Average Selling Prices (ASPs)
Average Selling Prices (ASPs) for diclofenac sodium EC 50mg enteric-coated tablets (a common strength and formulation) in bulk quantities for pharmaceutical formulators can range from approximately $0.05 to $0.15 per tablet as of late 2023/early 2024, depending on volume, supplier, and geographical market. Retail prices for consumer packaging will be higher, reflecting distribution markups, pharmacy dispensing fees, and branded generics if available.
Projected Price Movement (2024-2028)
The outlook for diclofenac sodium EC pricing is expected to be characterized by continued gradual decline, with potential for stabilization.
- Short-Term (2024-2025): Expect a low single-digit percentage decrease in API and bulk formulation prices. This is driven by ongoing intense generic competition and efficient manufacturing. Raw material cost volatility might cause minor price fluctuations, but the overall trend will remain downward.
- Medium-Term (2026-2028): The rate of price decline is likely to slow. Prices may approach a floor dictated by production costs and a baseline profit margin for manufacturers. Significant further reductions would require major advancements in production technology or a substantial decrease in raw material costs. The market may see stabilization or very marginal decreases (less than 2% annually).
Factors Supporting Price Stability or Minor Decline
- Established Generic Market: The market is mature; no significant new competitive pressures are anticipated beyond existing players.
- Manufacturing Efficiency: Continuous improvements in synthesis and formulation processes by major manufacturers help maintain cost competitiveness.
- Cost of Goods Sold (COGS): The baseline cost of producing diclofenac sodium EC is well-understood, providing a floor for pricing.
Factors That Could Introduce Price Volatility
- Supply Chain Disruptions: Unexpected global events (e.g., geopolitical conflicts, major natural disasters affecting chemical production regions) could temporarily increase raw material and shipping costs, leading to short-term price hikes.
- Regulatory Scrutiny: Heightened regulatory concerns regarding NSAID safety could lead to shifts in prescribing patterns and demand, potentially impacting overall market volume and, consequently, pricing.
- Emergence of Novel Therapies: While diclofenac sodium EC is cost-effective, the development of genuinely disruptive, safer, or more effective pain management therapies could gradually erode its market share over the long term, though this is unlikely to cause immediate drastic price shifts.
Price Comparison with Other NSAIDs
Compared to newer, branded NSAIDs or COX-2 inhibitors (e.g., celecoxib), diclofenac sodium EC remains significantly more affordable. This price differential is a key driver of its continued widespread use, particularly in healthcare systems with budget constraints and for patients requiring long-term management of chronic inflammatory conditions. For example, a typical course of generic diclofenac sodium EC can cost a fraction of a similar course of a branded COX-2 inhibitor.
Key Takeaways
Diclofenac sodium EC is a staple NSAID with a stable demand profile driven by its efficacy in treating common inflammatory and pain conditions. The market is highly competitive, characterized by generic dominance. Prices are projected to continue a slow, incremental decline through 2028, driven by manufacturing efficiencies and ongoing generic competition. Potential price volatility exists, primarily due to supply chain risks and evolving regulatory assessments of NSAID safety. Its significant cost advantage over newer therapies ensures its continued relevance in the global pharmaceutical market.
Frequently Asked Questions
-
What is the typical API cost range for diclofenac sodium? API costs for diclofenac sodium vary by purity, supplier, and volume. Bulk purchases for pharmaceutical manufacturers can range from approximately $20 to $60 per kilogram.
-
Are there significant differences in price between enteric-coated and immediate-release diclofenac sodium tablets? The enteric coating process adds a manufacturing step and material cost, typically resulting in slightly higher prices for enteric-coated formulations compared to immediate-release versions of the same API strength.
-
What is the primary impact of patent expiry on diclofenac sodium EC pricing? Patent expiry allows multiple generic manufacturers to produce and sell the drug, leading to increased competition and a substantial decrease in prices from branded levels to generic levels.
-
How do raw material price fluctuations in China impact diclofenac sodium EC pricing globally? As China is a major producer of pharmaceutical intermediates and APIs, significant price swings for raw materials used in diclofenac synthesis originating from China can directly influence global API pricing and subsequently the finished product costs.
-
Will new combination therapies including diclofenac sodium affect its standalone pricing? The introduction of combination therapies may not directly impact the pricing of standalone diclofenac sodium EC in the short term. However, if these combinations offer superior efficacy or safety profiles, they could gradually reduce demand for the standalone product, potentially leading to price adjustments in the long run.
Citations
[1] Rainsford, K. D. (2003). Diclofenac: The first 20 years. Birkhäuser Basel.
[2] Sostres, C., & Lanas, A. (2014). NSAIDs and the gut. Current Opinion in Gastroenterology, 30(6), 673–679.
[3] National Institute for Health and Care Excellence. (2016). Rheumatoid arthritis in adults: management. NICE guideline [NG11].
[4] Hippisley-Cox, J., & Coupland, C. (2017). Risk of cardiovascular events in users of selected NSAIDs: nested case-control studies. BMJ, 357, j1759.
More… ↓
