Share This Page
Drug Price Trends for DIBUCAINE
✉ Email this page to a colleague
Average Pharmacy Cost for DIBUCAINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
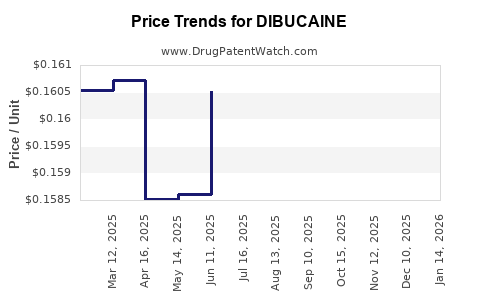
| DIBUCAINE 1% OINTMENT | 00536-1211-95 | 0.15020 | GM | 2026-03-18 |
| DIBUCAINE 1% OINTMENT | 00536-1211-95 | 0.15213 | GM | 2026-02-18 |
| DIBUCAINE 1% OINTMENT | 00536-1211-95 | 0.15677 | GM | 2026-01-21 |
| DIBUCAINE 1% OINTMENT | 00536-1211-95 | 0.15836 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Dibucaine: Market Dynamics and Price Forecast 2024-2030
Dibucaine, a potent local anesthetic, is positioned for stable market demand driven by its established therapeutic applications and the consistent need for pain management in surgical and dental procedures. Price projections indicate a moderate upward trend, influenced by manufacturing costs, regulatory environments, and supply chain dynamics.
What is Dibucaine and its Primary Applications?
Dibucaine is a synthetic ester-type local anesthetic. Its primary mechanism of action involves reversibly blocking nerve impulse conduction by inhibiting sodium ion influx across the neuronal membrane. This blockade is achieved by binding to voltage-gated sodium channels.
Key applications of dibucaine include:
- Local Anesthesia: Used for infiltration anesthesia, nerve blocks, and topical anesthesia. It is particularly effective in procedures requiring prolonged anesthesia due to its slower onset and longer duration of action compared to some other local anesthetics.
- Spinal Anesthesia: Formulations of dibucaine are utilized in spinal anesthesia for surgical procedures below the umbilicus. The concentration and volume injected determine the extent and duration of the block.
- Ophthalmology: Employed in ophthalmic procedures for topical anesthesia of the eye.
- Dermatology: Used in dermatological procedures and for the relief of itching associated with various skin conditions, often in suppository or cream formulations for hemorrhoids and pruritus ani.
- Research: Serves as a research tool in neurophysiology to study sodium channel function and nerve conduction.
A significant characteristic is its higher potency and longer duration of action compared to lidocaine or procaine, making it suitable for more extensive or prolonged procedures. However, it also carries a greater risk of systemic toxicity if administered in excessive doses.
What is the Current Market Size and Growth Trajectory for Dibucaine?
The global market for dibucaine is not segmented into a standalone category by most market research firms. Instead, it is typically included within broader segments such as "local anesthetics" or "anesthetics." This makes precise market sizing for dibucaine alone challenging. However, based on its established use, the market is characterized by steady demand rather than rapid expansion.
Estimates for the broader local anesthetic market, which includes dibucaine, suggest a Compound Annual Growth Rate (CAGR) of approximately 4% to 6% over the next five to seven years. This growth is underpinned by:
- Aging Global Population: Increased incidence of age-related conditions requiring medical interventions, including pain management.
- Rising Surgical Procedures: Growth in elective and non-elective surgeries across various specialties globally.
- Advancements in Pain Management: Continued research and development in anesthesia and analgesia, with dibucaine maintaining its niche due to its specific properties.
- Prevalence of Chronic Pain: A growing global burden of chronic pain conditions that require effective anesthetic interventions.
While dibucaine may not be experiencing the explosive growth of novel therapeutics, its consistent application in well-defined medical scenarios ensures a stable and predictable market presence. The market size for dibucaine is estimated to be in the tens of millions of U.S. dollars annually, a fraction of the multi-billion dollar overall local anesthetic market.
What are the Key Market Drivers for Dibucaine?
The demand for dibucaine is primarily driven by several factors inherent to its therapeutic profile and the healthcare landscape:
- Established Clinical Efficacy and Safety Profile: Dibucaine has been in use for decades, with a well-documented efficacy and safety profile when used within recommended guidelines. Healthcare professionals are familiar with its properties, leading to continued adoption.
- Demand in Specific Procedural Niches: Its longer duration of action and high potency make it the preferred choice for certain lengthy surgical procedures, nerve blocks requiring sustained anesthesia, and spinal anesthesia applications where prolonged pain relief is critical.
- Cost-Effectiveness in Certain Applications: Compared to some newer or more complex anesthetic agents, dibucaine can offer a cost-effective solution for specific clinical needs, particularly in regions with budget constraints.
- Growth in Outpatient Surgery Centers: The increasing trend towards outpatient surgical procedures, which often require local or regional anesthesia, contributes to the demand for anesthetics like dibucaine.
- Geriatric Population Growth: The expanding elderly population globally necessitates more medical interventions, including surgeries and pain management, thereby increasing the demand for effective local anesthetics.
- Prevalence of Chronic Pain and Palliative Care: Dibucaine's use in managing pain associated with conditions like hemorrhoids and in palliative care settings contributes to consistent demand.
What are the Key Market Restraints for Dibucaine?
Despite its established role, several factors constrain the market growth of dibucaine:
- Availability of Newer Anesthetics with Improved Safety Profiles: The development of newer local anesthetics with faster onset, shorter duration, and potentially lower systemic toxicity (e.g., ropivacaine, levobupivacaine) provides alternatives that may be preferred in certain clinical scenarios.
- Systemic Toxicity Risks: Dibucaine has a narrower therapeutic index and a higher risk of systemic toxicity (e.g., cardiovascular and central nervous system depression) compared to some amide-type anesthetics. This necessitates careful dosing and monitoring.
- Allergic Reactions: As with all local anesthetics, allergic reactions, though rare, can occur.
- Limited Novel Drug Development: There is limited ongoing research and development for novel dibucaine formulations or significant new indications, which limits its potential for market expansion through innovation.
- Regulatory Scrutiny and Pharmacovigilance: Like all pharmaceutical products, dibucaine is subject to stringent regulatory oversight and pharmacovigilance, which can impact manufacturing, labeling, and market access.
- Competition from Generics: The generic nature of dibucaine means that price competition among manufacturers is intense, limiting profit margins.
What are the Key Geographies for Dibucaine Consumption?
Dibucaine consumption is globally distributed, with higher concentrations in regions with well-developed healthcare infrastructures and a high volume of surgical procedures.
- North America (United States, Canada): This region represents a significant market due to the high number of surgical procedures, advanced healthcare systems, and established pain management protocols. The presence of numerous hospitals, surgical centers, and dental clinics drives demand.
- Europe (Germany, UK, France, Italy, Spain): Similar to North America, Europe has a substantial demand driven by advanced medical facilities, an aging population, and a high volume of elective surgeries. National health services and private healthcare providers contribute to consumption.
- Asia-Pacific (China, India, Japan, South Korea): This region is experiencing rapid growth in its healthcare sector. Increasing disposable incomes, improving healthcare access, and a rising prevalence of lifestyle-related diseases requiring surgical intervention are key drivers. China and India, with their large populations, are increasingly important markets.
- Latin America (Brazil, Mexico): The growing economies and improving healthcare infrastructure in countries like Brazil and Mexico are leading to increased demand for medical supplies, including anesthetics.
- Middle East and Africa: While currently smaller markets, these regions are projected to see growth as healthcare investments increase and access to medical services expands.
What are the Key Players in the Dibucaine Market?
The market for dibucaine is characterized by a mix of large pharmaceutical companies that may produce dibucaine as part of a broader anesthesia portfolio and smaller, specialized chemical manufacturers that focus on active pharmaceutical ingredients (APIs).
Key types of players include:
- API Manufacturers: Companies that synthesize and supply bulk dibucaine hydrochloride for pharmaceutical formulation. Examples of companies that may be involved in API production or have historically supplied it include:
- Hubei Jusheng Technology Co., Ltd.
- Anhui Haoyun Biotechnology Co., Ltd.
- Xi'an Sost Biological Technology Co., Ltd.
- Shandong Xinhua Pharmaceutical Co., Ltd. (though their primary focus may be broader)
- Several other fine chemical and pharmaceutical intermediate suppliers, particularly in China and India.
- Pharmaceutical Formulators: Companies that purchase dibucaine API and formulate it into finished dosage forms (injections, creams, suppositories). These are often regional or global pharmaceutical companies with anesthesia or pain management product lines. Examples of brands that may contain dibucaine include:
- Nupercainal (a brand historically associated with dibucaine ointment for hemorrhoids)
- Local anesthetic preparations by major pharmaceutical companies.
Due to the generic nature of dibucaine, specific market share data for individual manufacturers of the API or finished product is not consistently tracked or publicly disclosed by market research firms in the same way as patented drugs. Competition is primarily on price and supply reliability.
What are the Price Projections for Dibucaine Hydrochloride (API) from 2024 to 2030?
The price of Dibucaine Hydrochloride (API) is influenced by manufacturing costs, raw material availability, regulatory compliance, and global supply-demand dynamics. Projections indicate a stable to moderate upward trend.
| Year | Estimated Price Range (USD/kg) | Year-over-Year Change (%) | Key Influencing Factors |
|---|---|---|---|
| 2024 | $250 - $400 | N/A | Current manufacturing costs, raw material prices, existing supply chain |
| 2025 | $260 - $420 | +2.0% to +5.0% | Incremental increases in energy and labor costs, minor supply fluctuations |
| 2026 | $270 - $440 | +2.0% to +5.0% | Continued cost inflation, steady demand, potential minor supply chain adjustments |
| 2027 | $280 - $460 | +2.0% to +5.0% | Persistent manufacturing cost pressures, stable demand from end-use markets |
| 2028 | $290 - $480 | +2.0% to +5.0% | Global economic conditions impacting manufacturing, consistent procedural volumes |
| 2029 | $300 - $500 | +2.0% to +5.0% | Anticipated moderate inflation in raw materials and production, ongoing demand |
| 2030 | $310 - $520 | +2.0% to +5.0% | Long-term cost trends, supply chain stability, and sustained use in established applications |
Notes on Projections:
- API Pricing: These figures represent the price for bulk Dibucaine Hydrochloride API, not finished pharmaceutical products.
- Manufacturing Costs: Primary drivers include the cost of precursor chemicals, energy, labor, and compliance with Good Manufacturing Practices (GMP).
- Supply Chain: Disruptions or efficiencies in the supply chain, particularly from key manufacturing regions like China and India, can influence prices.
- Regulatory Compliance: Increasing stringency in environmental and safety regulations can add to manufacturing costs.
- Demand Stability: Dibucaine's primary uses are in well-established medical procedures, leading to a relatively stable demand curve that supports gradual price increases rather than volatile fluctuations.
- Currency Fluctuations: As a global commodity, prices can be influenced by currency exchange rates between major manufacturing and consuming regions.
What are the Price Projections for Finished Dibucaine Products (e.g., Injections) from 2024 to 2030?
Pricing for finished dibucaine products is more complex, incorporating API costs, formulation, packaging, distribution, marketing, and regulatory approval expenses.
| Product Type | Year | Estimated Price Range (USD per unit) | Year-over-Year Change (%) | Key Influencing Factors |
|---|---|---|---|---|
| Injection (10mL, 1% solution) | 2024 | $15 - $35 | N/A | API cost, formulation, packaging, distribution, competition |
| 2025 | $16 - $37 | +3.0% to +7.0% | Modest API cost increase, general inflation, supply chain costs | |
| 2026 | $17 - $39 | +3.0% to +7.0% | Continued cost pressures, R&D for improved formulations (if any), market access | |
| 2027 | $18 - $41 | +3.0% to +7.0% | Inflation, operational costs, competition among formulators | |
| 2028 | $19 - $43 | +3.0% to +7.0% | Global economic factors, healthcare reimbursement policies | |
| 2029 | $20 - $45 | +3.0% to +7.0% | Sustained demand, manufacturing efficiency, regulatory compliance | |
| 2030 | $21 - $47 | +3.0% to +7.0% | Long-term cost trends, market penetration in emerging economies | |
| Topical Ointment (e.g., 0.5%, 1 oz tube) | 2024 | $10 - $25 | N/A | API cost, formulation, packaging, retail markups, competitive landscape |
| 2025 | $11 - $27 | +4.0% to +8.0% | Higher relative impact of formulation and marketing costs, stable API price growth | |
| 2026 | $12 - $29 | +4.0% to +8.0% | General inflation in consumer goods, brand positioning | |
| 2027 | $13 - $31 | +4.0% to +8.0% | Market competition, retail dynamics, distribution channels | |
| 2028 | $14 - $33 | +4.0% to +8.0% | Economic conditions, consumer demand for OTC or prescription topical relief | |
| 2029 | $15 - $35 | +4.0% to +8.0% | Continued market stability, brand loyalty | |
| 2030 | $16 - $37 | +4.0% to +8.0% | Long-term market trends in dermatological care |
Notes on Projections:
- Formulation Costs: The cost of excipients, preservatives, sterile manufacturing processes, and quality control for injectable products significantly impacts pricing.
- Packaging: Sterility requirements for injectables and specific packaging for topical formulations contribute to costs.
- Distribution and Logistics: Maintaining cold chain (if applicable) and efficient distribution networks adds to the final price.
- Regulatory Approval and Maintenance: Costs associated with obtaining and maintaining regulatory approval in different markets are factored in.
- Market Competition: The presence of multiple generic manufacturers and competing anesthetic agents limits significant price increases for finished products.
- Reimbursement Policies: Healthcare provider reimbursement rates can influence pricing strategies for prescription products.
- OTC vs. Prescription: Pricing for over-the-counter (OTC) topical formulations will be influenced by different market dynamics and consumer price sensitivity compared to prescription injectables.
What is the Competitive Landscape for Dibucaine?
The competitive landscape for dibucaine is characterized by several layers of competition:
- Within Dibucaine Products: Competition exists among manufacturers of dibucaine API and among formulators of finished dibucaine products. This competition is primarily price-driven due to its generic status. Key differentiators are reliability of supply, quality control, and cost-effectiveness.
- Against Other Local Anesthetics: Dibucaine competes directly with a broad range of other local anesthetics. The choice of anesthetic is often dictated by the specific clinical procedure, desired onset and duration of action, patient factors, and clinician preference. This includes:
- Amide-type anesthetics: Lidocaine, bupivacaine, ropivacaine, mepivacaine, prilocaine. Ropivacaine and bupivacaine are often favored for their longer duration and potential for reduced cardiotoxicity compared to older agents.
- Ester-type anesthetics: Procaine, tetracaine. These are generally less potent and shorter-acting than dibucaine, and some have higher allergenic potential.
- Against Non-Anesthetic Pain Management Modalities: In some contexts, particularly for chronic pain or post-operative pain management, dibucaine may compete with other pain management strategies, including opioids, non-steroidal anti-inflammatory drugs (NSAIDs), acetaminophen, and non-pharmacological interventions.
The established efficacy and long duration of action of dibucaine secure its position for specific procedural needs, but it faces increasing competition from newer agents with potentially more favorable safety profiles or specific clinical advantages.
Key Takeaways
- Dibucaine maintains a stable market position within the broader local anesthetic sector, driven by its proven efficacy and utility in specific surgical, spinal, and topical applications.
- Market growth for dibucaine is projected to align with the overall local anesthetic market, estimated at 4%-6% CAGR, fueled by an aging population and an increase in surgical procedures.
- Key drivers include its established clinical profile, long duration of action, and cost-effectiveness in certain niches.
- Restraints stem from the availability of newer anesthetics with potentially improved safety profiles and a narrower therapeutic index for dibucaine.
- API pricing for Dibucaine Hydrochloride is projected to increase moderately between 2% and 5% annually, reaching $310-$520/kg by 2030, due to manufacturing and raw material costs.
- Finished dibucaine product pricing is expected to see higher annual increases of 3%-8% for injectables and 4%-8% for topical formulations, influenced by formulation, distribution, and market competition.
- The competitive landscape is characterized by generic price competition and direct rivalry with a spectrum of other local anesthetics, with choice often dependent on procedural requirements.
Frequently Asked Questions
-
What is the primary difference between dibucaine and lidocaine in terms of clinical use? Dibucaine is characterized by a slower onset and longer duration of action compared to lidocaine, making it suitable for procedures requiring prolonged anesthesia, whereas lidocaine offers a faster onset and shorter duration, making it versatile for many common procedures.
-
Are there significant risks associated with the use of dibucaine? Yes, dibucaine carries a risk of systemic toxicity, including central nervous system depression and cardiovascular effects, especially if administered in excessive doses or injected intravascularly. It also has a narrower therapeutic index than some newer anesthetics.
-
Will dibucaine be superseded by newer anesthetic agents in the near future? While newer agents with potentially improved safety profiles and different pharmacokinetic properties offer alternatives, dibucaine's unique potency and long duration of action ensure its continued use in specific clinical niches where these characteristics are paramount.
-
What factors are most likely to impact the cost of Dibucaine Hydrochloride API in the coming years? The primary factors influencing API cost will be fluctuations in raw material prices, energy costs for synthesis, labor expenses in manufacturing regions, and the ongoing costs associated with maintaining Good Manufacturing Practice (GMP) compliance and environmental regulations.
-
Is dibucaine used in any over-the-counter (OTC) products, and how does this affect its market? Dibucaine is available in some OTC topical preparations, such as ointments for hemorrhoid relief. This OTC availability contributes to a consistent demand from the consumer healthcare segment, separate from its prescription use in clinical settings.
More… ↓
