Drug Price Trends for DEXTROMETHORPHAN
✉ Email this page to a colleague
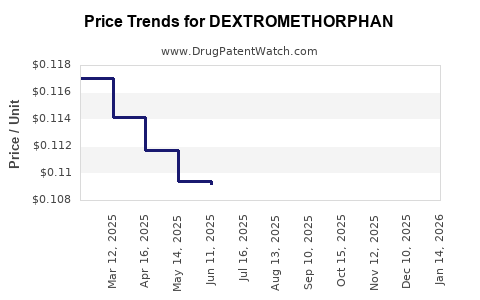
Average Pharmacy Cost for DEXTROMETHORPHAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROMETHORPHAN 15 MG SOFTGEL | 00536-1334-34 | 0.11035 | EACH | 2026-04-22 |
| DEXTROMETHORPHAN ER 30 MG/5 ML | 45802-0433-21 | 0.06822 | ML | 2026-04-22 |
| DEXTROMETHORPHAN 15 MG SOFTGEL | 00536-1334-34 | 0.10801 | EACH | 2026-03-18 |
| DEXTROMETHORPHAN ER 30 MG/5 ML | 45802-0433-21 | 0.06775 | ML | 2026-03-18 |
| DEXTROMETHORPHAN 15 MG SOFTGEL | 00536-1334-34 | 0.10452 | EACH | 2026-02-18 |
| DEXTROMETHORPHAN ER 30 MG/5 ML | 45802-0433-21 | 0.06745 | ML | 2026-02-18 |
| DEXTROMETHORPHAN 15 MG SOFTGEL | 00536-1334-34 | 0.10556 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DEXTROMETHORPHAN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DEXTROMETHORPHAN 20MG/GUAIFENESIN 400MG TAB | Richmond Pharmaceuticals Inc. | 54738-0985-30 | 30 | 2.69 | 0.08967 | EACH | 2024-02-15 - 2029-02-14 | FSS |
| DEXTROMETHORPHAN 20MG/GUAIFENESIN 400MG TAB | Richmond Pharmaceuticals Inc. | 54738-0985-60 | 60 | 3.90 | 0.06500 | EACH | 2024-02-15 - 2029-02-14 | FSS |
| DEXTROMETHORPHAN 15MG/PROMETHAZINE 6.25MG/5ML | Golden State Medical Supply, Inc. | 70436-0155-41 | 473ML | 8.70 | 0.01839 | ML | 2023-06-16 - 2028-06-14 | FSS |
| DEXTROMETHORPHAN 15MG/PROMETHAZINE 6.25MG/5ML | Golden State Medical Supply, Inc. | 70436-0155-42 | 473ML | 34.82 | 0.07362 | ML | 2023-06-16 - 2028-06-14 | FSS |
| DELSYM 12 HOUR (GRAPE) | RB Health (US) LLC | 63824-0171-65 | 148ML | 8.60 | 0.05811 | ML | 2022-06-15 - 2027-06-14 | FSS |
| MUCINEX DM 600MG/30MG TAB | RB Health (US) LLC | 63824-0056-32 | 20 | 8.52 | 0.42600 | EACH | 2022-06-15 - 2027-06-14 | FSS |
| CHILDREN'S MUCINEX COUGH (CHERRY) | RB Health (US) LLC | 63824-0946-03 | 118ML | 6.25 | 0.05297 | ML | 2022-06-15 - 2027-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
