Share This Page
Drug Price Trends for DEXTROAMPH-AMPHET ER
✉ Email this page to a colleague
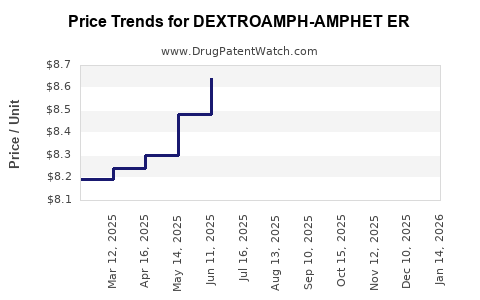
Average Pharmacy Cost for DEXTROAMPH-AMPHET ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROAMPH-AMPHET ER 25 MG CAP | 00406-0803-01 | 8.58582 | EACH | 2026-05-20 |
| DEXTROAMPH-AMPHET ER 12.5 MG CP | 57664-0950-88 | 8.16778 | EACH | 2026-05-20 |
| DEXTROAMPH-AMPHET ER 50 MG CAP | 57664-0953-88 | 8.51217 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Dextroamphet-Amph ER?
Dextroamphetamine-amphetamine extended-release (Dextroamphet-Amph ER) is a central nervous system stimulant approved for treating attention deficit hyperactivity disorder (ADHD) and narcolepsy. It combines dextroamphetamine and amphetamine in a formulation designed for gradual absorption, providing therapeutic effects over an extended period.
How is the current market landscape for Dextroamphet-Amph ER?
The market for long-acting stimulant medications for ADHD and narcolepsy remains robust. Key players include Shire (now part of Takeda), Teva Pharmaceutical Industries, and Novelty and Emerging Generic Players. The global ADHD medication market was valued at approximately $13.7 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of around 4.2% (2022-2030).
The market is affected by increased diagnosis rates, growing awareness, and expanding treatment in adult populations. Generics account for nearly 75% of prescriptions in the US, highlighting pressure on branded drugs' pricing. The US remains the dominant market, accounting for approximately 60% of global ADHD medication sales.
What’s the patent status and regulatory outlook?
The original patent for Dextroamphet-Amph ER expired around 2018, opening the market for generic manufacturers. Several generics have entered the US market since then. Regulatory agencies like the FDA have approved multiple generic versions, with variability in bioequivalence and release profiles.
FDA approvals permit multiple manufacturers, intensifying price competition. Manufacturers must submit Abbreviated New Drug Applications (ANDAs) demonstrating bioequivalence. Patent litigation and exclusivity periods influence market entry timelines and pricing.
What are the price trends for Dextroamphet-Amph ER?
The brand-name Dextroamphet-Amph ER currently retails at approximately $300–$350 for a month’s supply (30 tablets, 20mg each) in the US. Generic versions are priced around $50–$80 for the same quantity, reflecting significant price erosion post-generic entry.
In 2018, the average retail price for the brand was approximately $500 per month, which declined to current levels following generic competition. The price difference between the brand and generics can reach 80%–85%.
What are future price projections?
Multiple factors influence future pricing:
- Market Competition: As more generics enter, price erosion continues. If 10 or more generic producers market the drug, prices could decrease further to below $20 per month.
- Regulatory Changes: Patent extensions or new formulations could temporarily sustain higher prices.
- Pricing Trends: Historical trends in stimulant medications suggest a steady decline post-generic approval, with annual reductions of 15–25% observed in comparable drugs.
- Reimbursement Policies: Payers seek lower-cost alternatives, pressuring manufacturers to lower prices further.
- Global Expansion: Emerging markets typically see lower prices but represent growing sales opportunities.
Given these factors, estimates for the next 5 years project a sustained decline in generic prices, potentially stabilizing around $20–$30 per month by 2028, assuming no significant regulatory or market disruptions.
How do regulatory and market forces shape market dynamics?
Patent expiration facilitated generic entry, creating price competition. Regulatory agencies enforce bioequivalence standards but may also approve unique formulations or abuse-deterrent versions, potentially maintaining higher prices.
Market forces like insurance reimbursement policies influence prescription volume and profitability. Some payers prefer generics, further accelerating price reductions.
Emerging treatment modalities, such as non-stimulant medications or behavioral therapies, could also impact demand for Dextroamphet-Amph ER, influencing long-term sales and pricing.
Who are the key competitors and potential disruptors?
- Teva: Generic manufacturer with significant market share.
- Mallinckrodt: Previously licensed generic versions.
- Novel entrants: Small biotech firms exploring alternative formulations or delivery systems.
- Innovative therapies: Non-stimulant medications like atomoxetine or behavioral interventions could reduce reliance on stimulants.
Synthetic drug innovations or reformulated products with abuse-deterrent features are potential disruptors, potentially maintaining higher prices during development phases.
What are the key takeaways?
- The current US retail price for Dextroamphet-Amph ER is approximately $300–$350 for brand, with generics available at $50–$80.
- Patent expiry (~2018) spurred multiple generics, leading to steep price declines.
- Future pricing likely hinges on the number of generic competitors, regulatory policies, and payer behavior.
- Prices for generics may fall to below $20 per month within 5 years if market conditions persist.
- Market growth remains driven by increased diagnosis, expanding adult ADHD treatment, and global expansion opportunities.
FAQs
1. When did Dextroamphet-Amph ER become available as a generic?
Generic versions were approved around 2018, following patent expiration.
2. What factors could delay price declines?
Patent disputes, formulation modifications, or regulatory hurdles could extend higher prices temporarily.
3. How does insurance coverage influence pricing?
Payers favor generics, leading to higher utilization and lower out-of-pocket costs, driving prices downward.
4. Are there global markets beyond the US?
Yes, countries like Europe, Canada, and Australia are expanding use, often at lower prices, but face regulatory and reimbursement landscape differences.
5. What is the outlook for new formulations?
Developments like abuse-deterrent formulations or long-acting meds could sustain higher prices and market share for innovative products.
References
[1] IQVIA, "Medicap," 2022.
[2] MarketWatch, "ADHD medication market forecast," 2022.
[3] US Food and Drug Administration, "ANDA approvals," 2018–2022.
[4] Centers for Medicare & Medicaid Services, "Reimbursement trends," 2022.
More… ↓
