Share This Page
Drug Price Trends for DEXTROAMP-AMPHETAMINE
✉ Email this page to a colleague
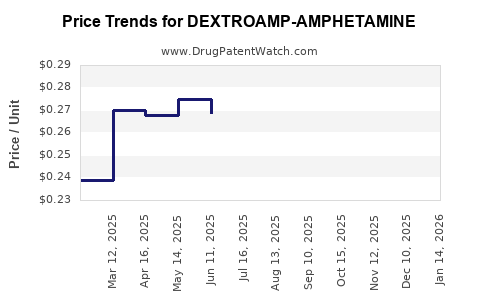
Average Pharmacy Cost for DEXTROAMP-AMPHETAMINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 00406-8891-01 | 0.25619 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 00555-0971-02 | 0.25619 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 11534-0190-01 | 0.25619 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 00527-0760-37 | 0.25619 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 72516-0016-01 | 0.25619 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for Dextroamp-Amphetamine
Dextroamp-Amphetamine is a central nervous system stimulant primarily used for attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is marketed under various brand names, including Dexedrine and Adderall, in formulations of different dosages. The drug's market is influenced by prescription trends, regulatory policies, and competitive dynamics.
Market Size and Demand Drivers
Global Prescription Market
The global ADHD drugs market, dominated by stimulants such as Dextroamp-Amphetamine, was valued at approximately $11.2 billion in 2022. This market is projected to grow at a compound annual growth rate (CAGR) of 6.1% through 2028 [1].
Regional Breakdown
- North America holds nearly 70% of the global stimulant market, with the U.S. accounting for approximately 80% of prescriptions.
- Europe is the second-largest market, driven by increased diagnosis and treatment adoption.
- Asia-Pacific shows rising demand due to growing awareness and healthcare infrastructure development.
Prescription Trends
- Increased diagnosis of ADHD in both children and adults escalates demand.
- Growing acceptance of pharmacological treatments over behavioral therapies sustains high prescription rates.
- Off-label uses and misuse potential influence demand dynamics.
Regulatory Environment Impact
The regulation of Dextroamp-Amphetamine varies:
- In the U.S., it is classified as a Schedule II controlled substance, restricting prescription quantities and dispensing processes [2].
- Stringent controls lead to supply limitations and influence pricing strategies.
Patent and Exclusivity Status
- Several formulations are patent-protected, extending market exclusivity until 2030.
- Generic formulations, once approved, increase competitive pressure and reduce prices.
Competition and Market Share
Key Competitors
| Product | Manufacturer | Brand Name | Formulation Types | Patent Status |
|---|---|---|---|---|
| Dextroamp-Amphetamine | Various | Adderall, Dexedrine | Immediate-release, extended-release | Patent until 2030 (varies) |
| LisDexamfetamine | Takeda Pharmaceuticals | Vyvanse | Prodrug formulation | Patent through 2030 |
Impact on Pricing
- Brand-name drugs typically retail between $200–$400 per month without insurance.
- Generic versions price between $80–$150 per month, depending on dosage and pharmacy.
Price Projections
Short to Mid-term Outlook (Next 3-5 Years)
- Prices for branded Dextroamp-Amphetamine products are expected to decline marginally due to patent expirations and increased generic competition.
- The average retail price for generic formulations could fall by approximately 15–25% over the forecast period.
- Wholesale and insurance reimbursement rates are likely to decrease as formulary preferences shift toward generics.
Long-term Price Trends (Beyond 5 Years)
- Market saturation with generics may stabilize prices at lower levels.
- The introduction of biosimilars or novel formulations could disrupt current pricing structures.
- Regulatory changes or shifts in prescribing guidelines might further influence market prices.
Impact of New Entrants and Formulation Innovations
- New extended-release or implant formulations may command higher prices initially but face generic competition within 3–5 years.
- Digital therapeutics and non-stimulant alternatives pose potential threats reducing stimulant demand and prices.
Key Factors Influencing Future Market and Prices
- Regulatory modifications altering classification or scheduling could restrict or expand market access.
- Patent expirations open pathways for generic entrants, reducing prices.
- Changes in clinical guidelines and healthcare provider preferences influence prescription volumes.
- Mental health awareness campaigns could increase diagnosis rates, boosting demand.
Summary of Price Estimates
| Year | Estimated Average Price (USD) | Scenario |
|---|---|---|
| 2023 | $200–$400 (brand) | Current market |
| 2024 | $180–$360 | Slight price decline |
| 2025 | $150–$300 | Increased generic competition |
| 2028 | $120–$250 | Post-patent expiration impact |
Key Takeaways
- The Dextroamp-Amphetamine market remains robust, driven by increasing diagnosis rates and approval of formulations.
- Patent expirations and generic availability will result in significant price reductions over the next five years.
- Regulatory and clinical guideline changes will influence prescription dynamics and market size.
- Pricing projections account for current trends; unexpected regulatory or scientific breakthroughs could alter forecasts.
- Investment in formulations or delivery methods offering differentiation may sustain higher margins.
FAQs
1. What is the primary driver of Dextroamp-Amphetamine market growth?
Increased diagnosis and acceptance of stimulant treatments for ADHD and narcolepsy.
2. How do patent expirations influence price trends?
They allow generics to enter the market, leading to price reductions of approximately 15–25%.
3. What regulatory factors could impact future prices?
Changes in scheduling status, prescription restrictions, or approval of non-stimulant alternatives.
4. Are biosimilars relevant for Dextroamp-Amphetamine?
Currently, biosimilars are not applicable; however, novel formulations or digital therapeutics might influence the market.
5. How does insurance coverage affect patient out-of-pocket costs?
Insurance preferences for generics can significantly lower patient expenses, influencing demand and pricing.
Citations
- Statista, "Global ADHD Drugs Market Size," 2022.
- U.S. Drug Enforcement Administration, "Controlled Substances Act," 2022.
More… ↓
